Extramural Intellectual Property 11 th Annual NIH SBIRSTTR

Extramural Intellectual Property 11 th Annual NIH SBIR/STTR Conference June 30, 2009 – Omaha, Nebraska Protecting Your Intellectual Property under your SBIR/STTR through i. Edison Invention: Why, When, & How - i. Edison J. P. Kim, J. D. , M. B. A. , M. Sc. , M. A. L. S. , Directory & Policy Officer, Division of Extramural Inventions & Technology Resources (DEITR), OPERA, NIH Office of Extramural Research (OER), OD, National Institutes of Health (NIH), U. S. Department of Health & Human Services (HHS) Email: jpkim@nih. gov 1

Why report inventions? n The Bayh-Dole Act n 35 U. S. C. 200 et al. 2

What Are the Main Provisions of the Bayh-Dole Act (1980)? n n n Provides title of Federally supported inventions to vest with the grantee/contractor organization Requires all grantee/contractors to demonstrate progress toward transfer of the technology Requires acknowledgment of the government’s involvement and license n n Government use license Federal support clause in the patent Provides incentive to inventors and more rapid development and commercialization Applicable to ALL U. S. Government-funded grantees and contractors: Domestic and Foreign 3

What is an Invention (per Bayh-Dole)? n 37 CFR Section 401. 14 Standard patent rights clauses. n n n (a) Definitions n (1) Invention means any invention or discovery which is or may be patentable or otherwise protectable under Title 35 of the United States Code, … n (2) Subject invention means any invention of the contractor conceived or first actually reduced to practice in the performance of work under this grant or contract, … Does not include solely copyrightable materials, e. g. , books Includes software; deemed patentable as of late ’ 90 s 4

Noted Misconceptions The government owns my invention. No, rights vest with the SBIR/STTR organization n If I report an invention, NIH (the government) will take my invention. No, NIH is not in competition with recipients. n By not reporting my invention to NIH, I will secure rights to an invention. No, there is a greater risk of loss of rights by not reporting. n 5

Stewardship of Federal Funds Practical Applications are an Important Part of Stewardship n How funds will be expended n n n How research will be conducted n n n Fiscal management Programmatic assessment Research integrity Conflict of interests How research outcomes will benefit the public n n Share outcomes to spawn new research ideas Encourage application of intellectual property 6

The Importance of Invention Reporting: Constant Scrutiny of NIH and Grantees n n n 1994 Congressional Inquiry 1995 Follow up compliance study 1997 GAO Audit n n 1998 GAO Audits n n n n E. O. 12591 Confirmatory licenses/Gov’t support clauses 1999 IG and GAO Audits n n Bayh-Dole Administration SBIRs Grantee compliance 5 year follow up to 1994 Report 2000 2002 2003 2005 – 2001 OIG: Further Revision of 1994 Report GAO - Use of Confirmatory Licenses GAO – Government Involvement in Taxol IP Congressional Inquiry 7

When do your report inventions? n The Bayh-Dole Act and its Implementing Regulations n n https: //s-edison. info. nih. gov/i. Edison/37 CFR 401. jsp Extramural Invention Reporting Compliance Responsibilities n https: //s-edison. info. nih. gov/i. Edison/timeline. jsp 8

Invention Reporting Requirements of SBIR Grantee/Contractors Administrative Requirements (per 37 CFR, 401. 14) n n n n n Implement Employee Agreements as needed Disclose Each Invention within 60 days Resolve Election or Waive of Title within 2 years File Patent within 1 yr. of election Provide License to the Govt. upon title election Indicate Govt. Support on Patent with patent appl. Product Manufacturing in U. S. required Report on Invention Utilization annually Final Invention Report at award close out 9

Annual Invention Utilization Reporting is Required for NIH n NIH Grants Policy Statement, 12/03 Revision, Page 119: “Grantees (and, in some cases, employee inventors) have rights to inventions (“subject inventions”) conceived or first actually reduced to practice in the performance of work under an NIH award. Grantee organizations must fulfill the following requirements: n Submit an annual utilization report for all inventions where election of title is made and for unpatented, yet licensed, inventions (37 CFR 401. 14(h)); …” 10

Utilization Report Requirements https: //s-edison. info. nih. gov/i. Edison/utilization_reporting. jsp Report according to grantee/contractor-defined 12 -month reporting cycle 1. Please indicate the latest stage of development of any product arising from this invention according to the following categories: Not Licensed/Commercialized 2. In the designated reporting period, what was the total income received as a result of license or option agreements? Do not include specific patent costs reimbursement. If any product arising from this invention has reached the market, what was the calendar year of the first commercial sale? In the designated reporting period, did the grantee organization/contractor or any of the exclusive licensees request a waiver of the U. S. manufacturing requirements? If yes, how many such waivers were obtained? In the designated reporting period, how many licenses and/or options of any type were awarded to small businesses (< 500 employees)? In the designated reporting period, how many exclusive licenses and/or options have been awarded? In the designated reporting period, how many non-exclusive licenses and/or options have been awarded? Please provide the commercial name of any FDA-approved products, utilizing this invention, that have first reached the market during the designated reporting period. 3. 4. 5. 6. 7. 8. 11
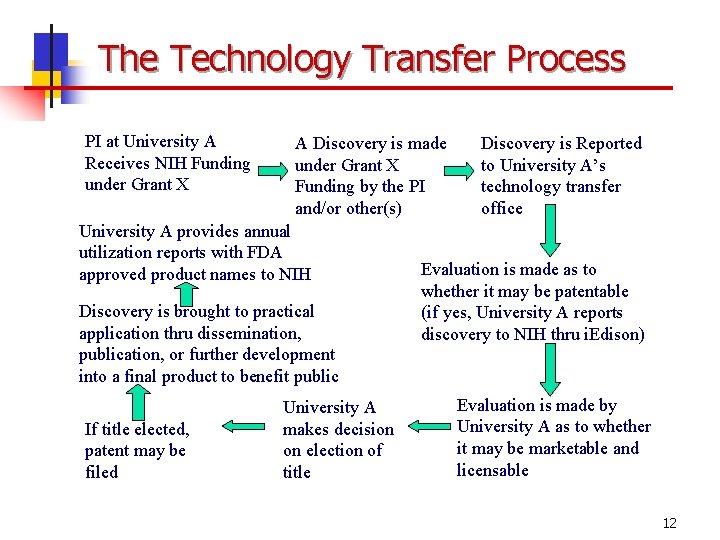
The Technology Transfer Process PI at University A Receives NIH Funding under Grant X A Discovery is made under Grant X Funding by the PI and/or other(s) University A provides annual utilization reports with FDA approved product names to NIH Discovery is brought to practical application thru dissemination, publication, or further development into a final product to benefit public If title elected, patent may be filed University A makes decision on election of title Discovery is Reported to University A’s technology transfer office Evaluation is made as to whether it may be patentable (if yes, University A reports discovery to NIH thru i. Edison) Evaluation is made by University A as to whether it may be marketable and licensable 12

How do you report inventions? n i. Edison (http: /i. Edison. gov) 13

http: //iedison. gov n Secure, Interactive Web-Based System for Bayh-Dole Policy and Reporting Compliance n n Didactic Content to inform investigators & grantee/contractor organizations n 37 CFR 401 n Invention Policy Issues n Invention Reporting Procedures/forms Current submission of Invention Report Records to any of 25 participating Federal Agencies including: n n n n NSF HHS/FDA HHS/CDC USAID HHS/ATSDR HHS/IHS AFRL/IF n n n n NAVY/ONR USAF/AFOSR DOC/NOAA EPA HHS/AHRQ (AHCPR) Army/SSC Army/SMDC n n n n USDA/CSREES USDA/Forest Service USDA/ARS USAF/ESC Army/MRMC NIH DOD/DMEA 14

http: //iedison. gov n Submission via browser or computer-to-computer datastream n n i. Edison Tutorial n n Used by ~1300 grantee/contractor organizations http: //era. nih. gov/Project. Mgmt/iedison 2/index. cfm i. Edison Registration n https: //sedison. info. nih. gov/i. Edison/Registration. Request. Form. jsp Help Line: (301) 435 -1986 Email: Edison@od. nih. gov 15

Why Use i. Edison? n Secure, relational, real time database with all invention reporting business rules built in n Requires minimal hardware/software n n Offers reminders of time critical reporting obligations i. Edison supports full electronic reporting: no paper is needed if you have the information electronically Internet connection Browser Meets invention reporting requirements for diverse population of interagency grantees/contractors Provides single gateway for invention reporting for over 25 government agencies 16

NIH Extramural Intellectual Property Resources for SBIR/STTR Awardees n Interagency Edison http: //i. Edison. gov n Extramural Inventions n n J. P. Kim, (jpkim@nih. gov), Director & Policy Officer, Division of Extramural Inventions & Technology Resources, OPERA, NIH Office of Extramural Research, OD, NIH, HHS John Salzman, (js 99 a@nih. gov), Assistant Extramural Inventions Policy Officer, Division of Extramural Inventions & Technology Resources, OPERA, NIH Office of Extramural Research, OD, NIH, HHS (301) 435 -1986 n n i. Edison User Support n edison@od. nih. gov n (301) 435 -1986 Websites: n http: //i. Edison. gov n http: //inventions. nih. gov 17
- Slides: 17