Extractions Mixture of benzoic acid anthracene and pnitroaniline

Extractions Mixture of benzoic acid, anthracene, and p-nitroaniline
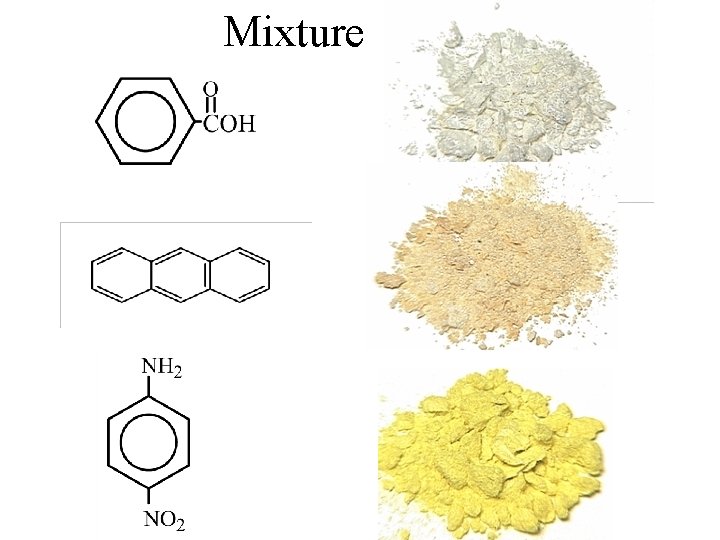
Mixture
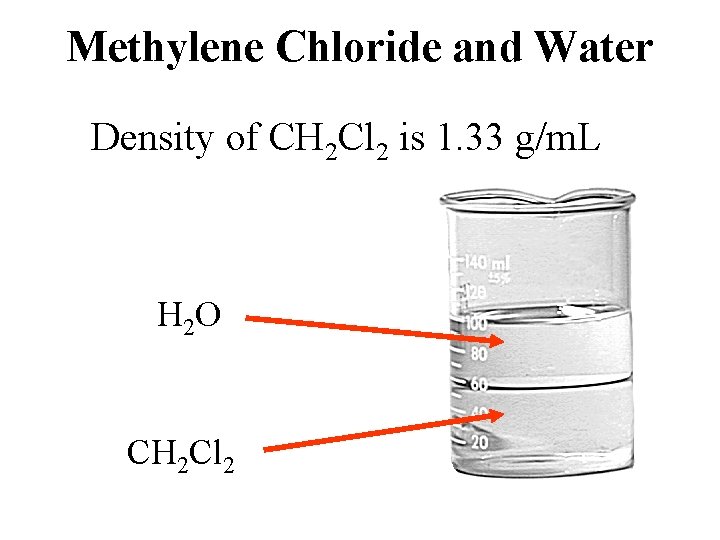
Methylene Chloride and Water Density of CH 2 Cl 2 is 1. 33 g/m. L H 2 O CH 2 Cl 2

Add Salt If we dissolve Na. Cl in this mixture which phase has the highest salt concentration? Salts concentrate in the water layer

Add Benzoic Acid If we dissolve benzoic acid in this mixture in which phase would it concentrate? Neutral organic compounds concentrate in the organic phase Salts
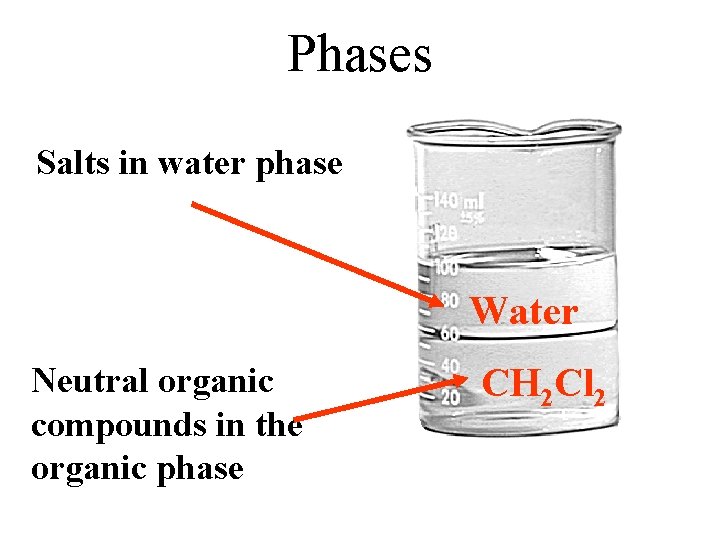
Phases Salts in water phase Water Neutral organic compounds in the organic phase CH 2 Cl 2
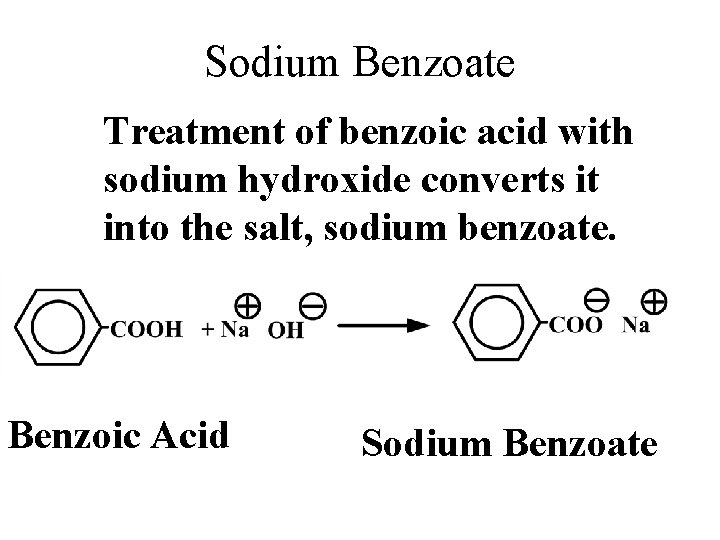
Sodium Benzoate Treatment of benzoic acid with sodium hydroxide converts it into the salt, sodium benzoate. Benzoic Acid Sodium Benzoate
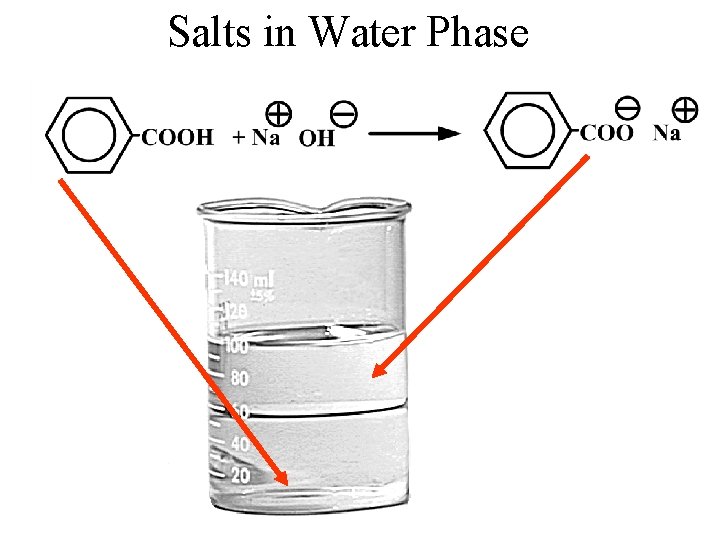
Salts in Water Phase
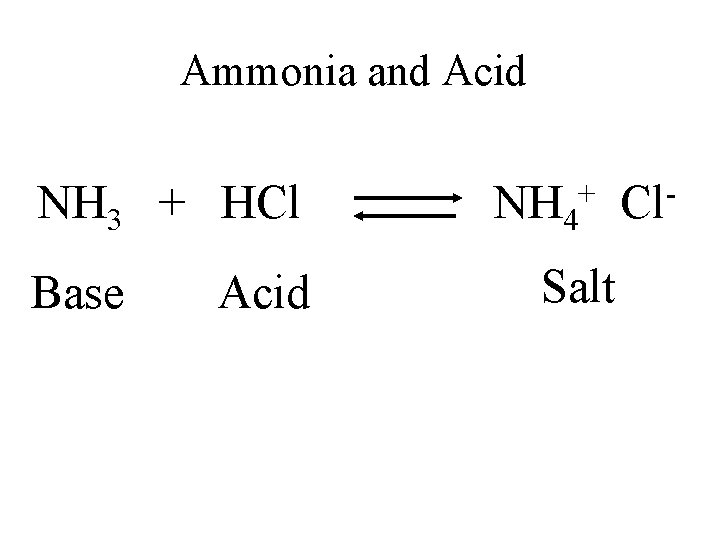
Ammonia and Acid NH 3 + HCl Base Acid + NH 4 Salt Cl
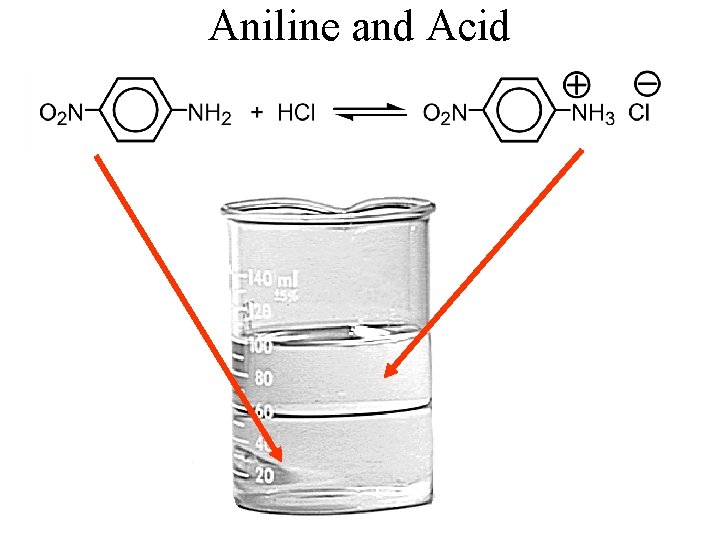
Aniline and Acid

Separatory Funnel Use separatory funnel for separating liquid phases.

Shake to be sure phases equilibrate Hold stopper and stopcock
Vent frequently
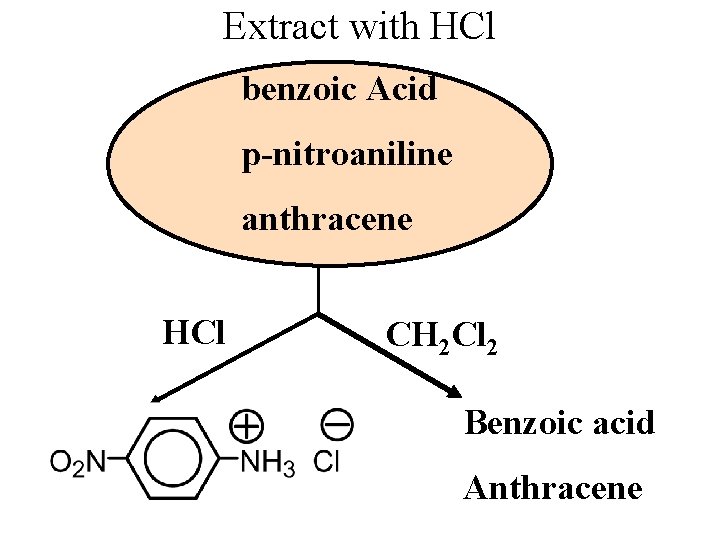
Extract with HCl benzoic Acid p-nitroaniline anthracene HCl CH 2 Cl 2 Benzoic acid Anthracene
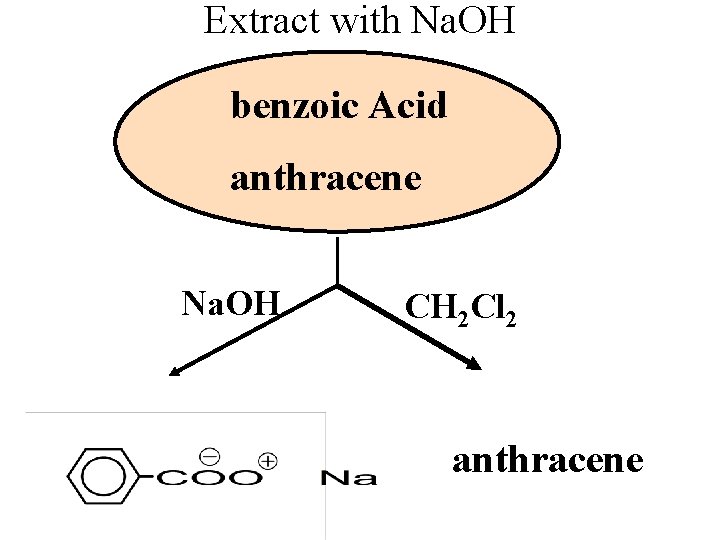
Extract with Na. OH benzoic Acid anthracene Na. OH CH 2 Cl 2 anthracene
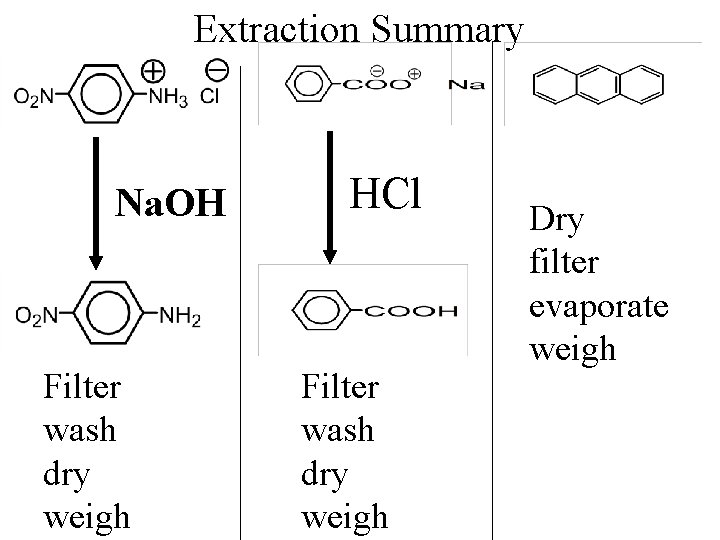
Extraction Summary Na. OH Filter wash dry weigh HCl Filter wash dry weigh Dry filter evaporate weigh

Precipitate Benzoic Acid Addition of acid to the sodium benzoate solution causes benzoic acid to precipitate.

Purify Benzoic Acid The benzoic acid is filtered, washed, and dried.

Weight, m. p. Weigh the benzoic acid and measure its melting point.
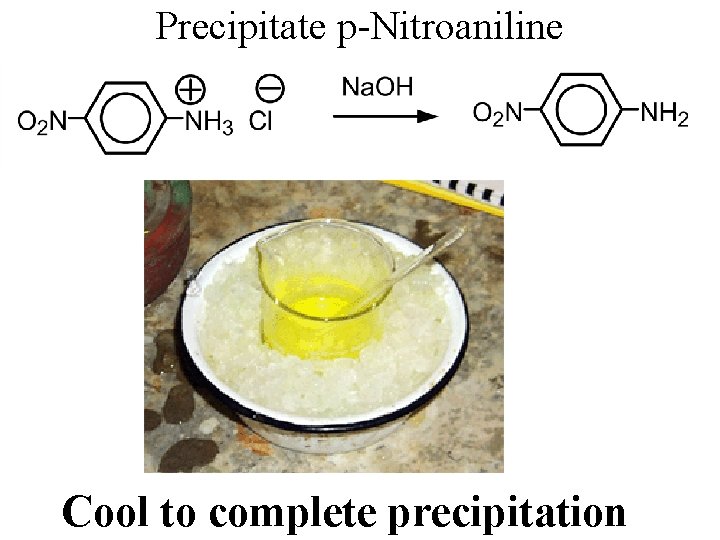
Precipitate p-Nitroaniline Cool to complete precipitation

Dry Methylene Chloride Dry the methylene chloride solution of anthracene with a small amount of sodium sulfate.

Filter Remove the drying agent by filtration through a fluted filter paper.

Remove Methylene Chloride Evaporate the methylene chloride on a steam bath. Use an inverted funnel connected to the vacuum line to collect the vapor.

Weigh your products and measure their melting points. Put your samples in labeled vials and give them to your TA.

Partition Coefficients 10 g t-butyl alcohol 100 m. L ether 100 m. L water
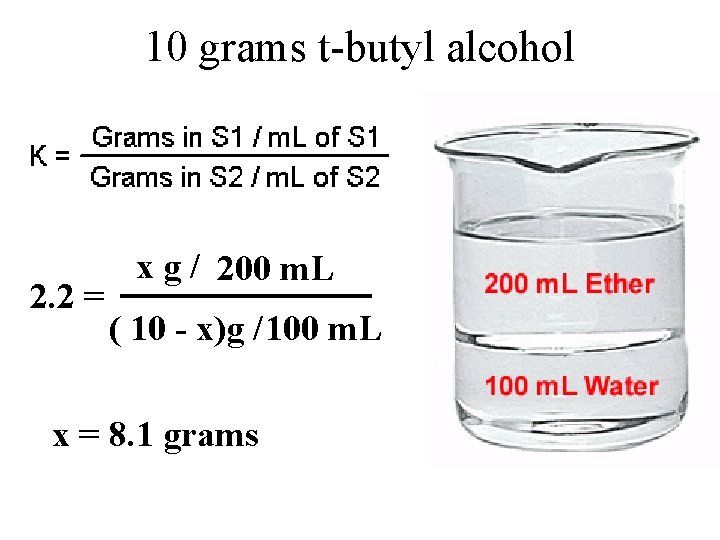
10 grams t-butyl alcohol 2. 2 = x g / 200 m. L ( 10 - x)g /100 m. L x = 8. 1 grams

Which is better? Extract once with 200 m. L or twice with 100 m. L?

If K = 1. 0, a = 10 grams, S 2 = 100 m. L Extract once with 200 m. L yields 6. 7 grams Extract once with 100 m. L yields 5. 0 grams Second extraction with 100 m. L yields 2. 5 grams Total 2 extractions with 100 m. L = 7. 5 grams
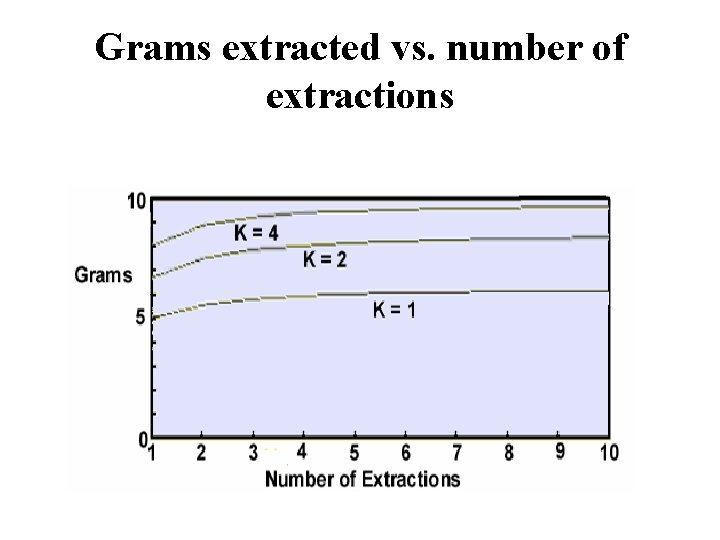
Grams extracted vs. number of extractions
- Slides: 29