Extending the Use of Metformin Richard Day Clinical

















- Slides: 27

Extending the Use of Metformin Richard Day Clinical Pharmacology & Toxicology

Type 2 diabetes mellitus (T 2 DM) • 382 million people worldwide in 2013 • 592 million by 2035 (UN, 2013) • Hyperglycaemia - insulin resistance and ↓insulin secretion • Complications: vasculopathy, retinopathy, and nephropathy • 7 classes of oral anti-diabetic drugs

Metformin – Dimethylbiguanide Gallega Officinalis (French Lilac) • 1929 - Several biguanidines synthesised 1. • Introduction of insulin declining interest in biguanidines until 1950’s. • 1959 - Phenformin associated with fatal lactic acidosis; not observed with metformin. • 1977 - Phenformin & Buformin withdrawn in USA then elsewhere. • Metformin suffered collateral damage. 1) Hesse G, Taubmann G. Die Wirkung des Biguanids und seiner Derivate auf den Zuckerstoffwechsel. Naunyn- Schmiedebergs. Arch Exp Path Pharmacol 1929; 142: 290– 308.

Metformin – First line therapy for T 2 DM • Benefits: – Little or no hypoglycaemia – Little or no weight gain – Evidence for ↓ risk microvascular and macrovascular complications → Cardioprotective – Possible ↓ Risk of cancer

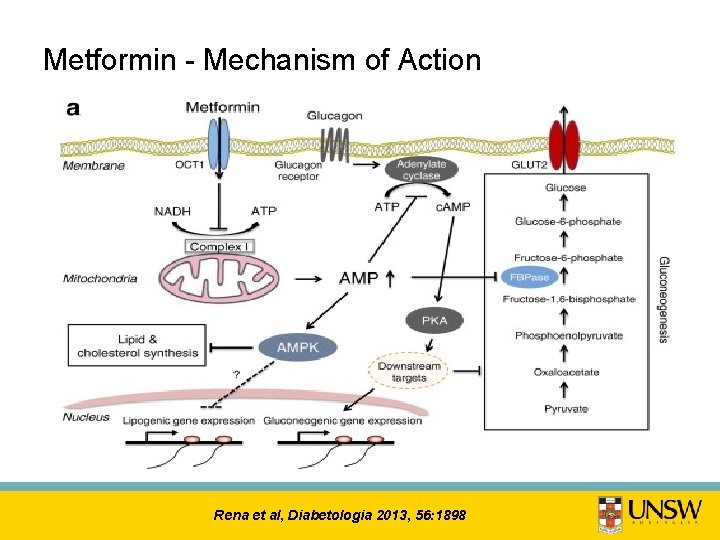
Metformin - Mechanism of Action Rena et al, Diabetologia 2013, 56: 1898

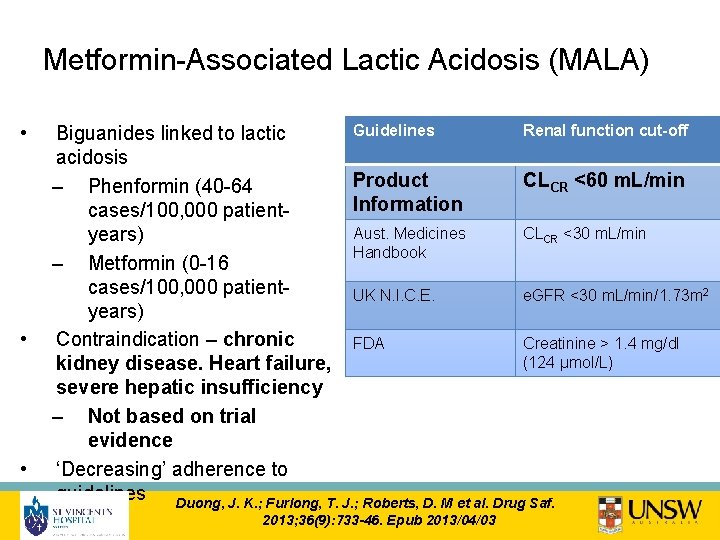
Metformin-Associated Lactic Acidosis (MALA) • • • Guidelines Renal function cut-off Biguanides linked to lactic acidosis Product CLCR <60 m. L/min – Phenformin (40 -64 Information cases/100, 000 patient. Aust. Medicines CLCR <30 m. L/min years) Handbook – Metformin (0 -16 cases/100, 000 patient. UK N. I. C. E. e. GFR <30 m. L/min/1. 73 m 2 years) Contraindication – chronic FDA Creatinine > 1. 4 mg/dl (124 μmol/L) kidney disease. Heart failure, severe hepatic insufficiency – Not based on trial evidence ‘Decreasing’ adherence to guidelines Duong, J. K. ; Furlong, T. J. ; Roberts, D. M et al. Drug Saf. 2013; 36(9): 733 -46. Epub 2013/04/03

• UK Prospective Diabetes Study (UKPDS) o 20 year study (1978 -1998) o 23 centres; 4, 075 patients (1704 patients > 120% ideal BMI) • Conventional therapy vs intensive pharmacological therapy o Insulin, sulphonylurea or metformin • Cardiovascular disease accounted for 62% of the total mortality in the overweight patients in the conventional treatment group. • The metformin group had: o 36% lower risk (p=0· 011) of all-cause mortality, o 39% lower risk (p=0· 010) of myocardial infarction, o 30% lower risk (p=0· 020) of macrovascular disease (myocardial infarction, sudden death, angina, stroke, and peripheral disease), than the conventional group.


• • Design: 304 patients, mean age = 63 yr, randomised to glipizide (30 mg daily) or metformin (1. 5 g daily) for 3 years. [5 yr follow-up]. End points: Times to composite of recurrent cardiovascular events [death from CV or any cause; MI; stroke; arterial re-vascularisation]. Findings: Metformin reduced major cardiovascular events at median of 5. 0 years compared with glipizide. Results indicated a potential benefit of metformin therapy on cardiovascular outcomes in high-risk patients. Diabetes Care 2013; 36: 1304 -1311

Conclusions— “The totality of evidence indicates that metformin is at least as safe as other glucose-lowering treatments in patients with diabetes mellitus and HF and even in those with reduced left ventricular ejection fraction or concomitant chronic kidney disease. Until trial data become available, metformin should be considered the treatment of choice for patients with diabetes mellitus and HF”.

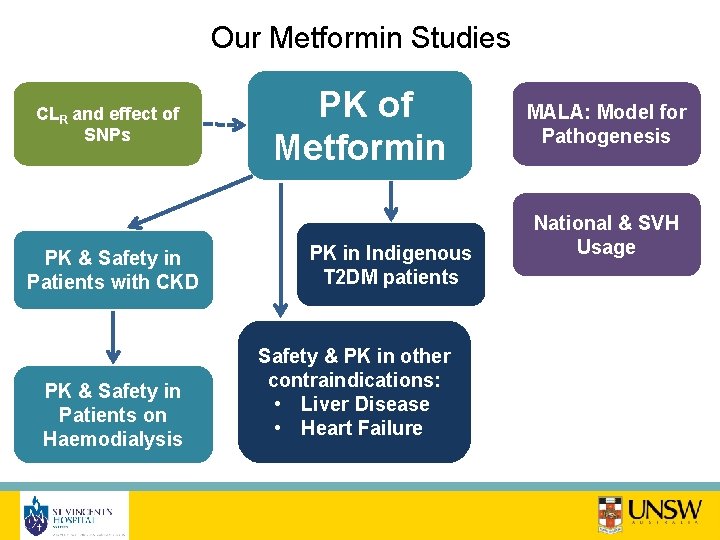
Our Metformin Studies CLR and effect of SNPs PK & Safety in Patients with CKD PK & Safety in Patients on Haemodialysis PK of Metformin PK in Indigenous T 2 DM patients Safety & PK in other contraindications: • Liver Disease • Heart Failure MALA: Model for Pathogenesis National & SVH Usage

Pharmacokinetics (PK) of metformin • Hydrophillic base, cationic (>99. 9%) @ p. H 7. 4 • Not metabolised, eliminated unchanged, • Renal clearance (CLR) ≈ 4 x creatinine clearance (CLCR) • Bioavailability (F): 25 – 75 % with average of 55% • Half-life (t 1/2): 5 - 6 hours (longer terminal half-life) • Volume of distribution (VD/F): 600 -800 L Graham et al, Clinical Pharmacokinetics 2011; 50(2): 1 -18


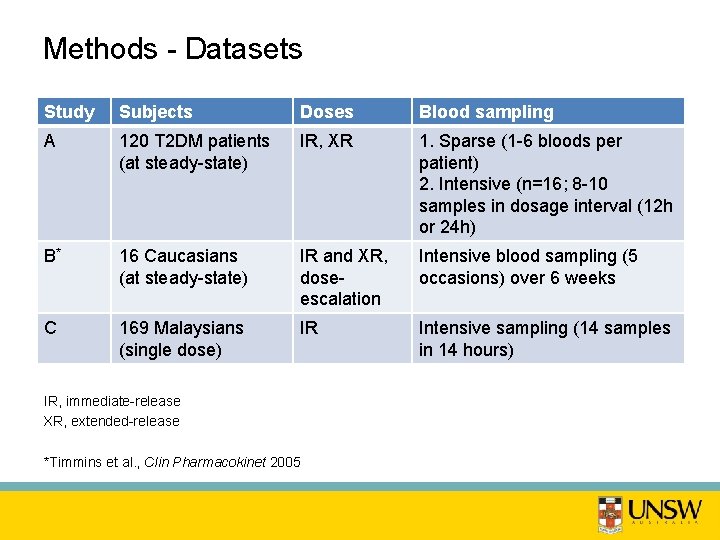
Methods - Datasets Study Subjects Doses Blood sampling A 120 T 2 DM patients (at steady-state) IR, XR 1. Sparse (1 -6 bloods per patient) 2. Intensive (n=16; 8 -10 samples in dosage interval (12 h or 24 h) B* 16 Caucasians (at steady-state) IR and XR, doseescalation Intensive blood sampling (5 occasions) over 6 weeks C 169 Malaysians (single dose) IR Intensive sampling (14 samples in 14 hours) IR, immediate-release XR, extended-release *Timmins et al. , Clin Pharmacokinet 2005

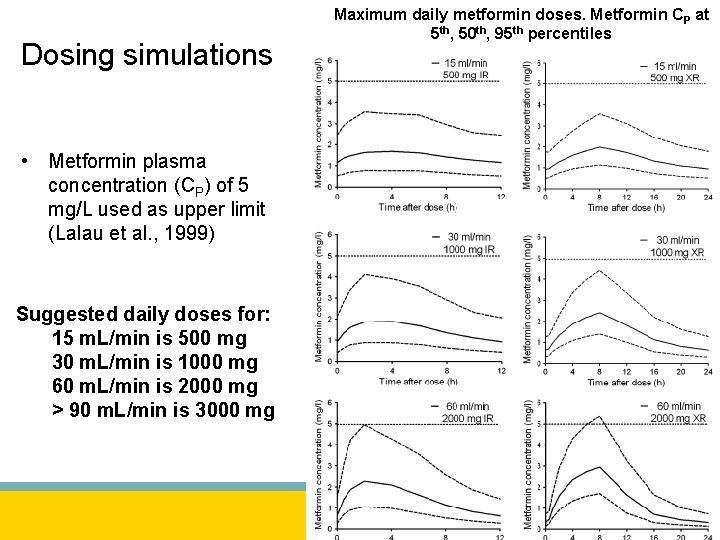
Dosing simulations • Metformin plasma concentration (CP) of 5 mg/L used as upper limit (Lalau et al. , 1999) Suggested daily doses for: 15 m. L/min is 500 mg 30 m. L/min is 1000 mg 60 m. L/min is 2000 mg > 90 m. L/min is 3000 mg Maximum daily metformin doses. Metformin CP at 5 th, 50 th, 95 th percentiles

J. K. Duong, D. M. Roberts, T. J. Furlong, S. S. , Kumar, J. R. Greenfield, C. M. Kirkpatrick, G. G. Graham, K. M. Williams & R. O. Day

Metformin Safety in CKD Methods • Patients with CKD currently taking metformin (n=22; creatinine clearances 15– 40 ml/min) were: • Ceased metformin • Then prescribed low doses of metformin (500 mg daily) for 6 weeks • Blood samples for metformin, lactate & creatinine Duong et al, Metformin. Diabetes, Obesity & Metabolism 2012; 14: 963

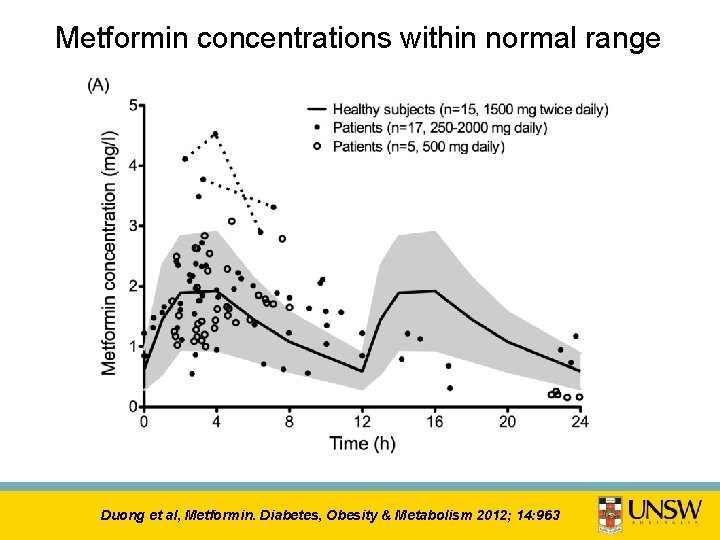
Metformin concentrations within normal range Duong et al, Metformin. Diabetes, Obesity & Metabolism 2012; 14: 963

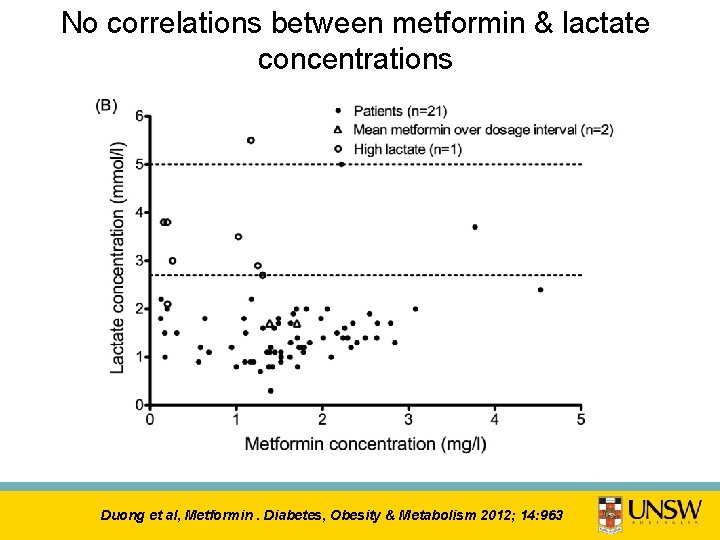
No correlations between metformin & lactate concentrations Duong et al, Metformin. Diabetes, Obesity & Metabolism 2012; 14: 963

Metformin Safety in CKD • Metformin can be safely used in patients with CKD • Daily doses should be lower • Large variability in concentrations! – likely due to variable absorption • Metformin & lactate concentrations should be monitored regularly Duong et al, Metformin. Diabetes, Obesity & Metabolism 2012; 14: 963

Metformin in Diabetic Haemodialysis Patients Methods • Patients (n = 4) with T 2 DM + ESKD who were: o Metformin naïve o Undergoing regular haemodiafiltration (HDF) o 3 sessions/week o Prescribed metformin (IR) for 12 weeks o 500 mg post-HDF (i. e. 1500 mg/week) Smith et al Am J Kidney Dis. 2016; 68(6): 990 -992 4

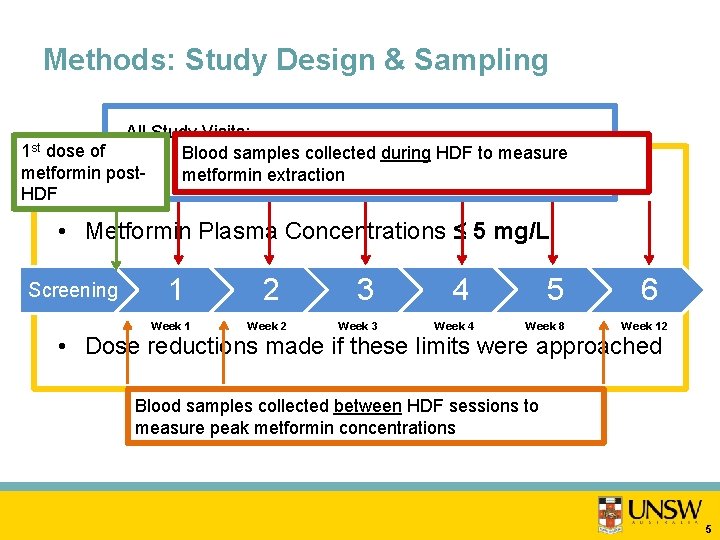
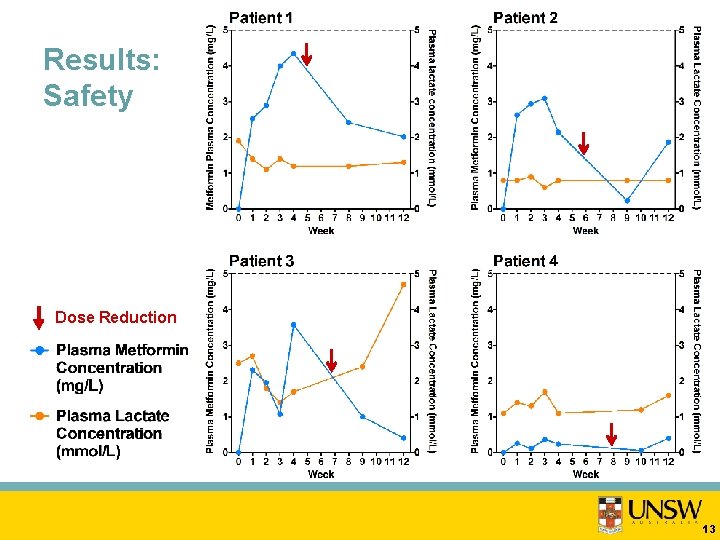
Methods: Study Design & Sampling All Study Visits: dose of Blood samples collected during HDFHDF to measure • Occurred during the patient’s regular sessions metformin postmetformin extraction • Pre-dialysis blood sample biochemical monitoring Safety Monitoring: HDF 1 st • Metformin Plasma Concentrations ≤ 5 mg/L Screening • Lactate 1 Concentrations 2 Week 1 Week 2 ≤ 3 5 mmol/L 4 Week 3 Week 4 5 Week 8 6 Week 12 • Dose reductions made if these limits were approached Blood samples collected between HDF sessions to measure peak metformin concentrations 5

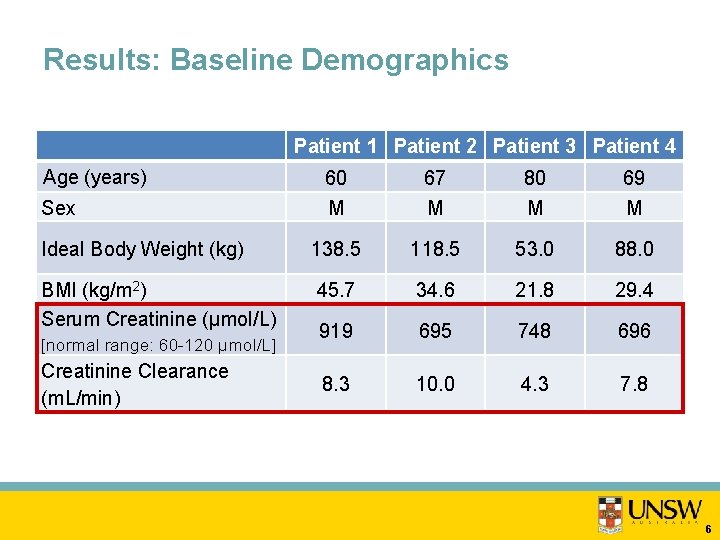
Results: Baseline Demographics Patient 1 Patient 2 Patient 3 Patient 4 Age (years) 60 67 80 69 Sex M M Ideal Body Weight (kg) 138. 5 118. 5 53. 0 88. 0 BMI (kg/m 2) Serum Creatinine (μmol/L) 45. 7 34. 6 21. 8 29. 4 919 695 748 696 8. 3 10. 0 4. 3 7. 8 [normal range: 60 -120 μmol/L] Creatinine Clearance (m. L/min) 6

Results: Safety Dose Reduction 13

Metformin Dialysis Study II Protocol Progress at Cessation • Longitudinal study (CTN) of • 8 patients entered diabetic patients in dialysis • Patient 1, aged 63, died ~ 45 minutes taking metformin 250 mg post dialysis post each dialysis session – Been on metformin since first study • Metformin & lactate – Pre-dialysis metformin 2. 2 mg/L; concentration monitored pre lactate 0. 8 mmol/L (4 days prior) -dialysis – Cardiac arrest*, unspecified • Proposed 50 subjects at • Patient 2, aged 64, died at home post SVH and Liverpool dialysis# centres – 2 weeks on metformin – Pre-dialysis metformin 1. 1 mg/L; lactate 2. 7 mmol/L (7 days prior) – Presumed MI (coroner’s verdict) * MI with ventricular fibrillation time of death - awaiting autopsy. Died at home over weekend. Last dialysis session Friday; did not turn up for dialysis Monday morning; TOE & cardioversion for arrhythmia 1 #Unknown

Conclusions • Safe use of metformin in HDF patients still worth pursuing – Diabetes control and cardiovascular risk reduction • Monitoring for metformin and lactate concentrations needed • Is there an ‘interaction’ between metformin & dialysis patients ‘increasing’ cardiovascular risk? • Approach to answering this question under consideration • Ethical aspects also under consideration

Acknowledgements ILP Students Manit Arora, 2009 Ji Moon, 2013 Hannah Braithwaite, 2014 Suraj Gangaram, 2015 BSc Hons Students Suraj Gangaram, 2014 Janna Duong, 2009 Shaun Kumar, 2010 Ph. D Students Janna Duong, 2010 - 13 Shaun Kumar, 2012 -2015 Felicity Smith, 2014 International Students Jeroen Punt, Holland, 2008 Anna Lindstrom, Sweden, 2009 Marjolein Kroonen, Holland, 2011 Clinical Pharmacology Professor Garry Graham, Dr Sophie Stocker, Dr Jane Carland, Professor Ken Williams Registrars Dr Darren Roberts Dr Jonathan Brett Collaborators Dr Tim Furlong & Dialysis Unit Nurses A/Prof Jerry Greenfield A/Prof Mark Danta External Collaborators Professor Carl Kirkpatrick, Monash U Professor Susan Davis, Monash U Professor Kathy Giacomini, UCSF Dr Peter Timmins, UK Dr Tung Lee, Malaysia Dr Hiddo Heerspink, Netherlands Funding St Vincent’s Clinic Foundation, 2009 ARC Linkage Grant , LP 0990670 Diabetes Australia Research Trust, 2014 NH&MRC Programme Grants 1054146