Extended Treatment of VTE Who is the Right









- Slides: 22

Extended Treatment of VTE: Who is the Right Candidate? Moderator Jeffrey I. Weitz, MD, FRCP(C) Professor of Medicine and Biochemistry Mc. Master University Hamilton, Ontario, Canada

Panelists Jan Beyer-Westendorf, MD Assistant Professor University Hospital Carl Gustav Carus der Technische Universität Dresden, Germany Peter Verhamme, MD, Ph. D Professor Department of Cardiovascular Sciences University of Leuven, Belgium

Program Goals Increased knowledge regarding • VTE as a chronic disease Demonstrate greater confidence in their ability to • Decide when and how to extend anticoagulant treatment, based on current data

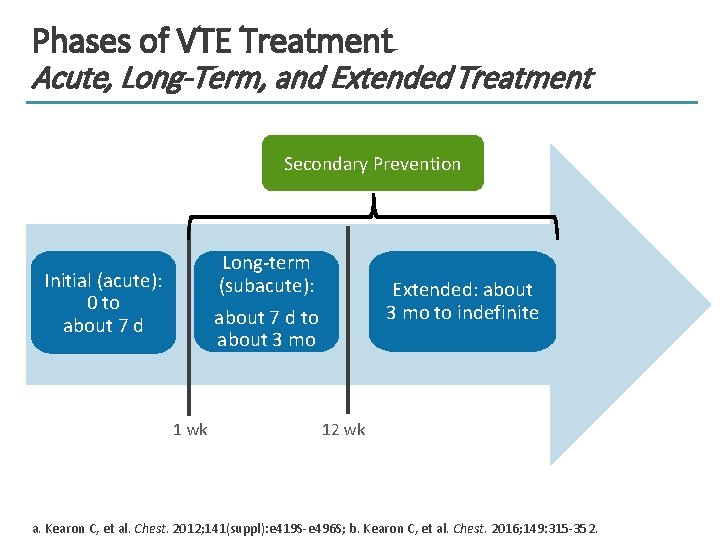
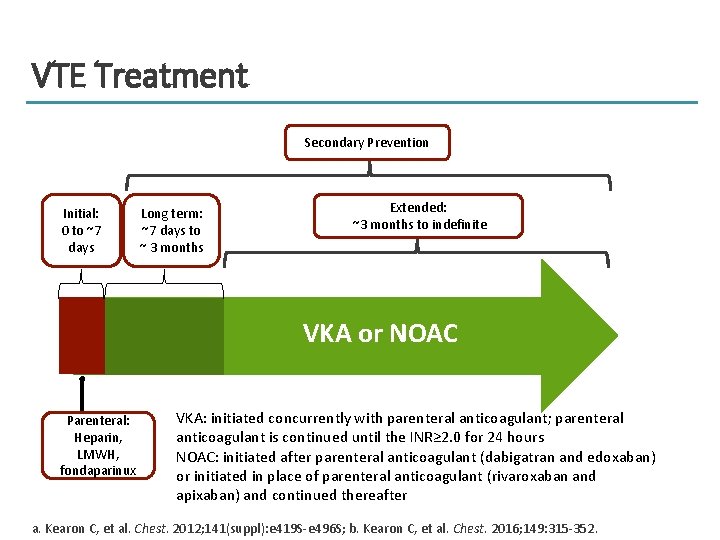
Phases of VTE Treatment Acute, Long-Term, and Extended Treatment Secondary Prevention Long-term (subacute): about 7 d to about 3 mo Initial (acute): 0 to about 7 d 1 wk Extended: about 3 mo to indefinite 12 wk a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

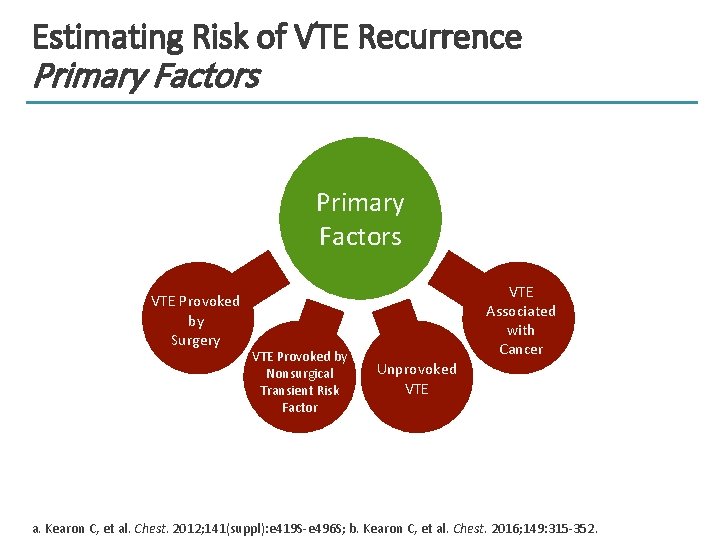
Estimating Risk of VTE Recurrence Primary Factors VTE Provoked by Surgery VTE Provoked by Nonsurgical Transient Risk Factor VTE Associated with Cancer Unprovoked VTE a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

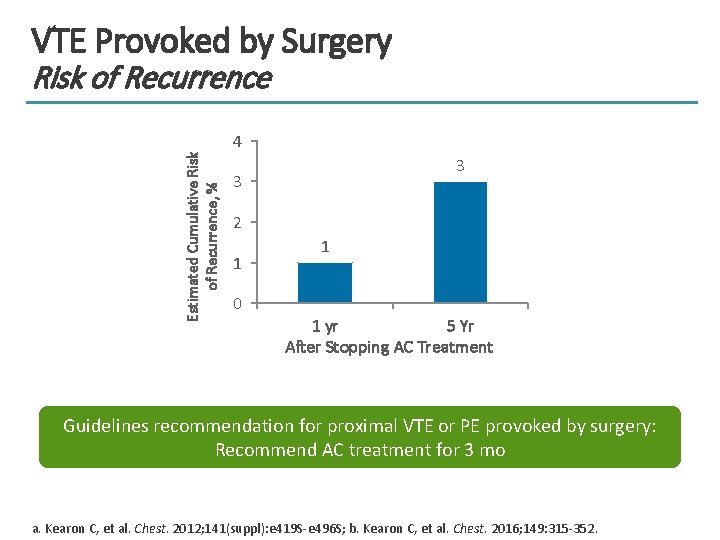
VTE Provoked by Surgery Risk of Recurrence Estimated Cumulative Risk of Recurrence, % 4 3 3 2 1 0 1 1 yr 5 Yr After Stopping AC Treatment Guidelines recommendation for proximal VTE or PE provoked by surgery: Recommend AC treatment for 3 mo a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

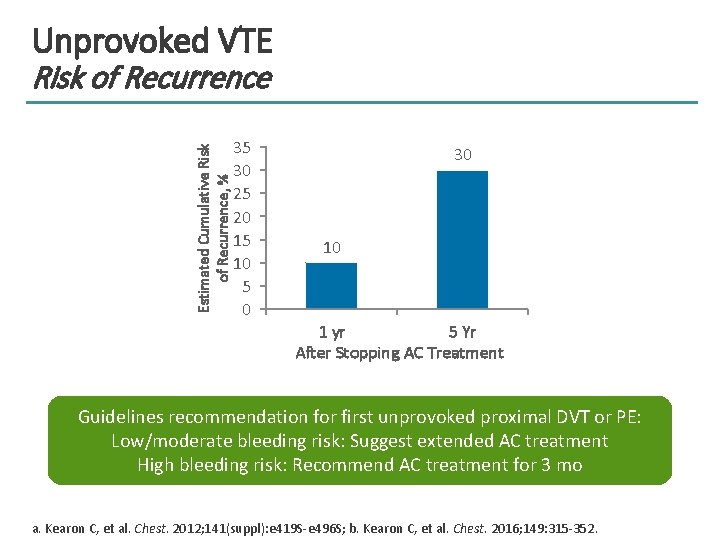
Unprovoked VTE Estimated Cumulative Risk of Recurrence, % Risk of Recurrence 35 30 25 20 15 10 5 0 30 10 1 yr 5 Yr After Stopping AC Treatment Guidelines recommendation for first unprovoked proximal DVT or PE: Low/moderate bleeding risk: Suggest extended AC treatment High bleeding risk: Recommend AC treatment for 3 mo a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

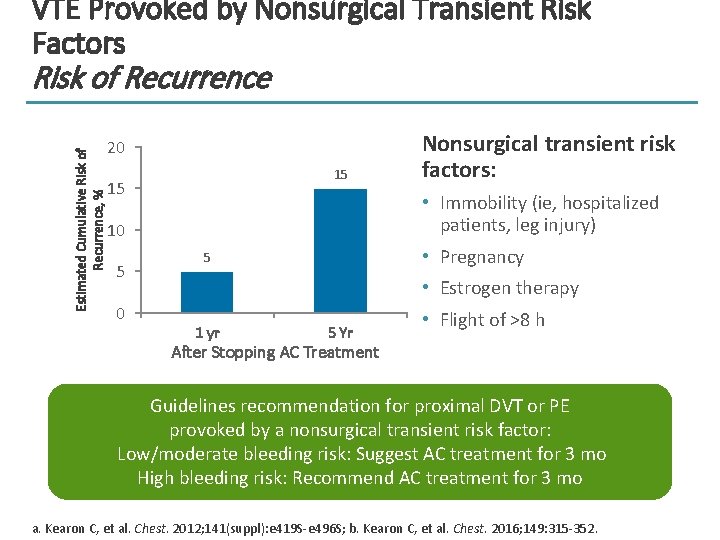
VTE Provoked by Nonsurgical Transient Risk Factors Estimated Cumulative Risk of Recurrence, % Risk of Recurrence 20 15 15 • Immobility (ie, hospitalized patients, leg injury) 10 5 0 Nonsurgical transient risk factors: • Pregnancy 5 • Estrogen therapy 1 yr 5 Yr • Flight of >8 h After Stopping AC Treatment Guidelines recommendation for proximal DVT or PE provoked by a nonsurgical transient risk factor: Low/moderate bleeding risk: Suggest AC treatment for 3 mo High bleeding risk: Recommend AC treatment for 3 mo a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

VTE Associated With Cancer Risk of Recurrence • Annualized recurrence: 15% • Risk varies depending on whether cancer: – Is metastatic – Being treated with chemotherapy – Rapidly progressing Guidelines recommended for DVT or PE and active cancer: Low/moderate bleeding risk: Recommend extended AC treatment High bleeding risk: Suggest extended AC treatment a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

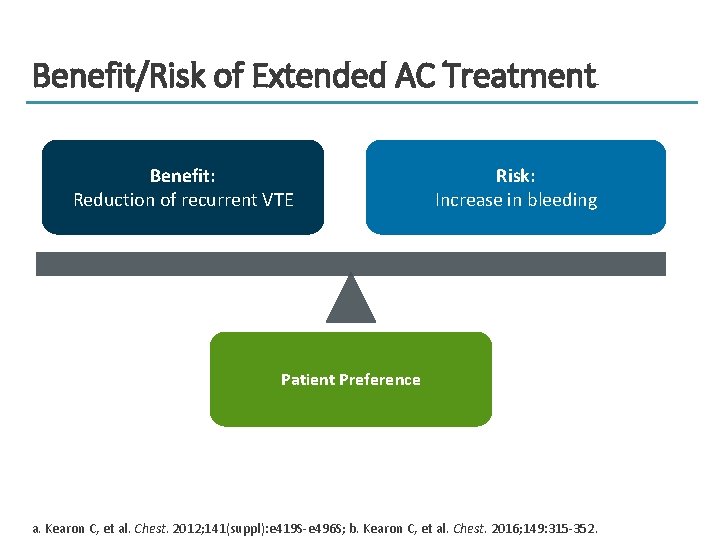
Benefit/Risk of Extended AC Treatment Benefit: Reduction of recurrent VTE Risk: Increase in bleeding Patient Preference a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

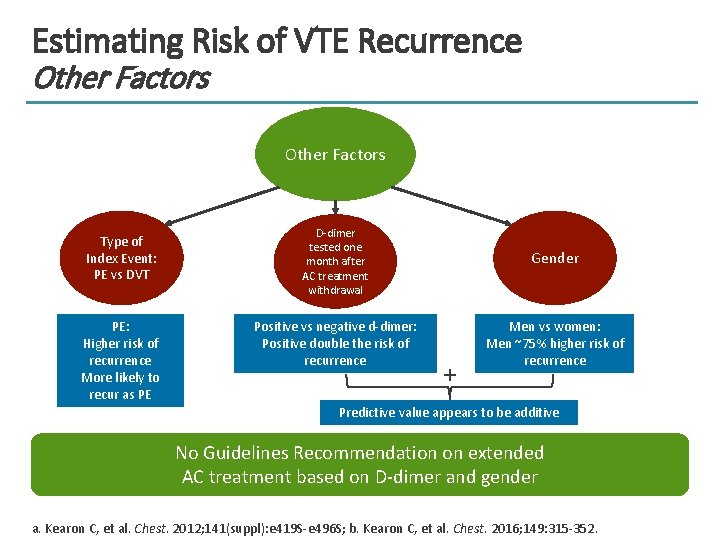
Estimating Risk of VTE Recurrence Other Factors Type of Index Event: PE vs DVT PE: Higher risk of recurrence More likely to recur as PE D-dimer tested one month after AC treatment withdrawal Gender Positive vs negative d-dimer: Positive double the risk of recurrence Men vs women: Men ~75% higher risk of recurrence + Predictive value appears to be additive No Guidelines Recommendation on extended AC treatment based on D-dimer and gender a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

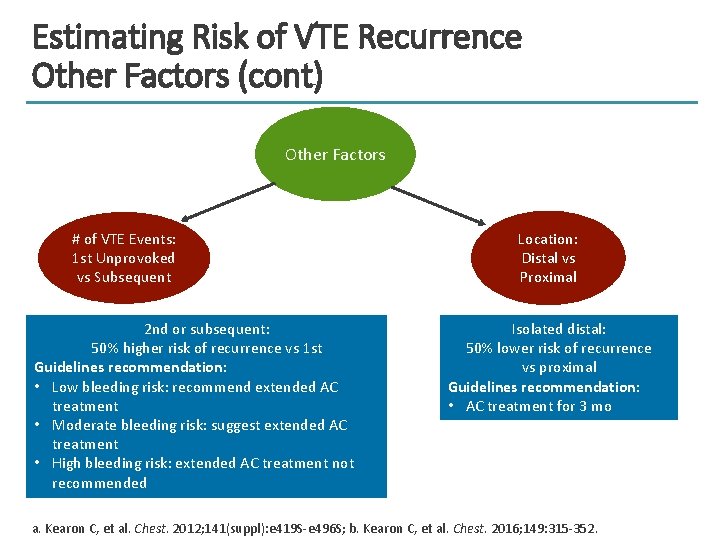
Estimating Risk of VTE Recurrence Other Factors (cont) Other Factors # of VTE Events: 1 st Unprovoked vs Subsequent 2 nd or subsequent: 50% higher risk of recurrence vs 1 st Guidelines recommendation: • Low bleeding risk: recommend extended AC treatment • Moderate bleeding risk: suggest extended AC treatment • High bleeding risk: extended AC treatment not recommended Location: Distal vs Proximal Isolated distal: 50% lower risk of recurrence vs proximal Guidelines recommendation: • AC treatment for 3 mo a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

VTE Treatment Secondary Prevention Initial: 0 to ~7 days Long term: ~7 days to ~ 3 months Extended: ~3 months to indefinite VKA or NOAC Parenteral: Heparin, LMWH, fondaparinux VKA: initiated concurrently with parenteral anticoagulant; parenteral anticoagulant is continued until the INR≥ 2. 0 for 24 hours NOAC: initiated after parenteral anticoagulant (dabigatran and edoxaban) or initiated in place of parenteral anticoagulant (rivaroxaban and apixaban) and continued thereafter a. Kearon C, et al. Chest. 2012; 141(suppl): e 419 S-e 496 S; b. Kearon C, et al. Chest. 2016; 149: 315 -352.

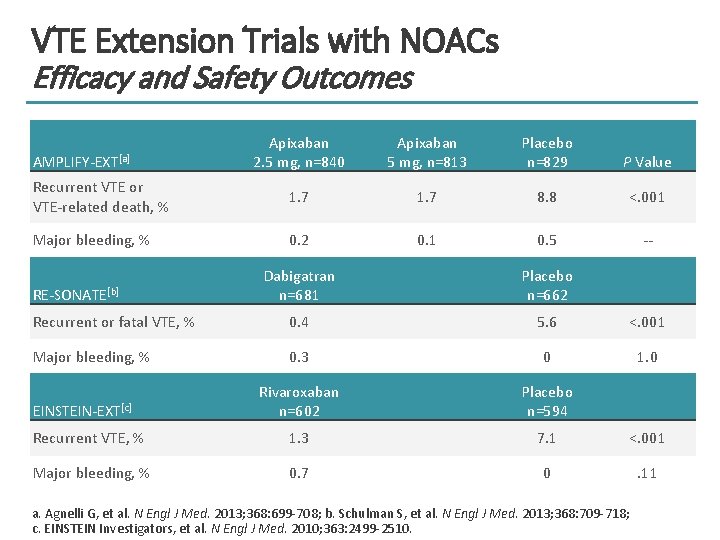
VTE Extension Trials with NOACs Efficacy and Safety Outcomes Apixaban 2. 5 mg, n=840 Apixaban 5 mg, n=813 Placebo n=829 P Value Recurrent VTE or VTE-related death, % 1. 7 8. 8 <. 001 Major bleeding, % 0. 2 0. 1 0. 5 -- AMPLIFY-EXT[a] Dabigatran n=681 Placebo n=662 Recurrent or fatal VTE, % 0. 4 5. 6 <. 001 Major bleeding, % 0. 3 0 1. 0 Rivaroxaban n=602 Placebo n=594 Recurrent VTE, % 1. 3 7. 1 <. 001 Major bleeding, % 0. 7 0 . 11 RE-SONATE[b] EINSTEIN-EXT[c] a. Agnelli G, et al. N Engl J Med. 2013; 368: 699 -708; b. Schulman S, et al. N Engl J Med. 2013; 368: 709 -718; c. EINSTEIN Investigators, et al. N Engl J Med. 2010; 363: 2499 -2510.

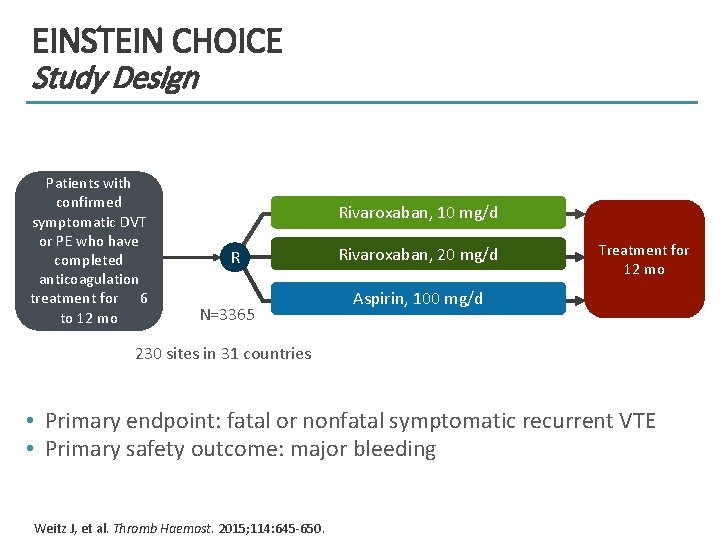
EINSTEIN CHOICE Study Design Patients with confirmed symptomatic DVT or PE who have completed anticoagulation treatment for 6 to 12 mo Rivaroxaban, 10 mg/d R N=3365 Rivaroxaban, 20 mg/d Treatment for 12 mo Aspirin, 100 mg/d 230 sites in 31 countries • Primary endpoint: fatal or nonfatal symptomatic recurrent VTE • Primary safety outcome: major bleeding Weitz J, et al. Thromb Haemost. 2015; 114: 645 -650.

EINSTEIN CHOICE Patient Population Patients were excluded if they had: • Symptomatic VTE recurrence during a 6 to 12 mo treatment period • Indication for extended AC therapy (recurrent VTE, severe thrombophilia, or antiphospholipid syndrome) Target Population: Patients for whom treating physician was uncertain about the need for continued AC therapy Weitz J, et al. Thromb Haemost. 2015; 114: 645 -650.

INSPIRE Aspirin for Prevention of Recurrent VTE CONTENT NO LONGER AVAILABLE • Major vascular events: composite of recurrent VTE, MI, stroke, or CV death • Net clinical benefit: recurrent VTE, MI, stroke, all-cause mortality, and major bleeding Simes J, et al. Circulation. 2014; 130: 1062 -1071.

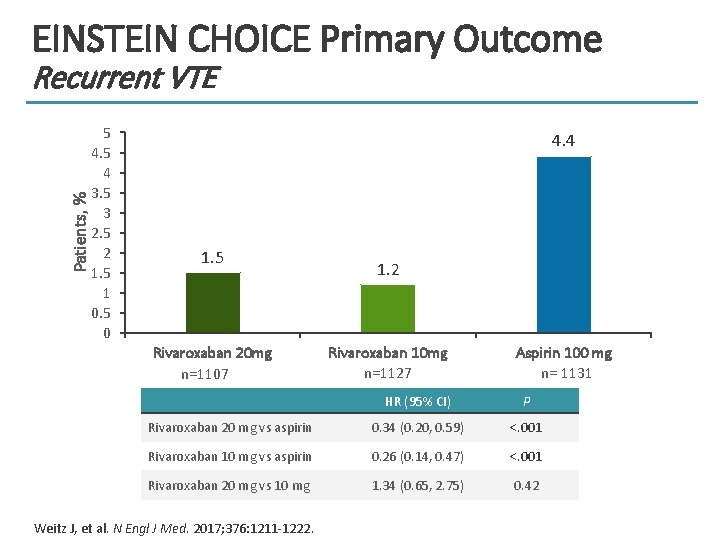
EINSTEIN CHOICE Primary Outcome Patients, % Recurrent VTE 5 4 3. 5 3 2. 5 2 1. 5 1 0. 5 0 4. 4 1. 5 Rivaroxaban 20 mg n=1107 1. 2 Rivaroxaban 10 mg n=1127 Aspirin 100 mg n= 1131 HR (95% CI) P Rivaroxaban 20 mg vs aspirin 0. 34 (0. 20, 0. 59) <. 001 Rivaroxaban 10 mg vs aspirin 0. 26 (0. 14, 0. 47) <. 001 Rivaroxaban 20 mg vs 10 mg 1. 34 (0. 65, 2. 75) 0. 42 Weitz J, et al. N Engl J Med. 2017; 376: 1211 -1222.

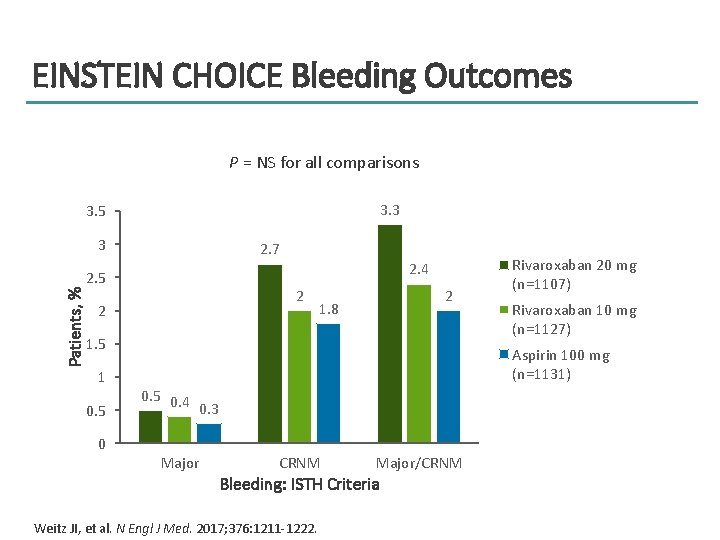
EINSTEIN CHOICE Bleeding Outcomes P = NS for all comparisons 3. 3 3. 5 3 2. 7 2. 4 Patients, % 2. 5 2 2 2 1. 8 1. 5 1 0. 5 Rivaroxaban 20 mg (n=1107) Rivaroxaban 10 mg (n=1127) Aspirin 100 mg (n=1131) 0. 5 0. 4 0. 3 0 Major CRNM Major/CRNM Bleeding: ISTH Criteria Weitz JI, et al. N Engl J Med. 2017; 376: 1211 -1222.

EINSTEIN CHOICE Clinical Implications 10 -mg dose: • For most patients • For patients with bleeding concerns 20 -mg dose: • For high-risk patients – – Recurrent VTE Severe thrombophilia Antiphospholipid syndrome Cancer

Summary • VTE is a chronic disease and a major health concern • Many patients remain at high risk for recurrence • NOACs provide a simplified treatment option for these patients – Acute phase: high-dose treatment up front with apixaban for 1 week or rivaroxaban for 3 weeks – Long-term phase: Continue with NOAC dose – Extended phase (for eligible patients): option of lower-dose rivaroxaban • Data from trials such as EINSTEIN CHOICE demonstrate that extended treatment with NOACs is safe and effective

Thank you for participating in this activity. You may now revisit those questions presented at the beginning of the activity to see what you've learned by clicking on the Earn Credit link. The CME posttest will follow. Please also take a moment to complete the program evaluation at the end.