Extended Thromboprophylaxis with Betrixaban in Acutely Ill Medical

Extended Thromboprophylaxis with Betrixaban in Acutely Ill Medical Patients August 23 rd, 2016 Presenter: Brandon Cave, Pharm. D PGY-2 Cardiology Pharmacy Resident West Palm Beach VA Medical Center Mentor: Paul P. Dobesh, Pharm. D, FCCP, BCPS – AQ Cardiology Professor of Pharmacy Practice College of Pharmacy University of Nebraska Medical Center

Disclosure • Dr. Cave does not have (nor does any immediate family member have) : – a vested interest in or affiliation with any corporate organization offering financial support or grant monies for this continuing education activity – any affiliation with an organization whose philosophy could potentially bias my presentation • The views expressed in this presentation reflect those of the author, and not necessarily those of the Department of Veterans Affairs • Dr. Dobesh has the following financial relationships to disclose: – Consultant for: • • • Portola Boehringer Ingelheim Pfizer/Bristol-Myers Squibb Daiichi Sankyo Janssen

VTE in the Acute Medically Ill • Acute medical illness increases venous thromboembolism (VTE) risk 5 -fold • Risk of DVT (symptomatic + asymptomatic) in medical patients is estimated to be between 5 – 30% IMPROVE Study (n=15, 156) Days after Hospital Admission In-Hospital VTE (n=79) Post-discharge VTE (n=64) All VTE (N=143) 1 -7 42 (53%) 0 42 (29%) 8 -30 32 (41%) 24 (38%) 56 (39%) 31 -60 5 (6%) 23 (36%) 28 (20%) 61 -91 0 17 (27%) 17 (12%) Spyropoulos AC, et al. Chest 2011; 140(3): 706 -14 Dobesh PP. Pharmacotherapy. 2009; 29(8): 943 -53 Heit JA, et al. J Thrombolysis. 2016; 41(1): 3 -14
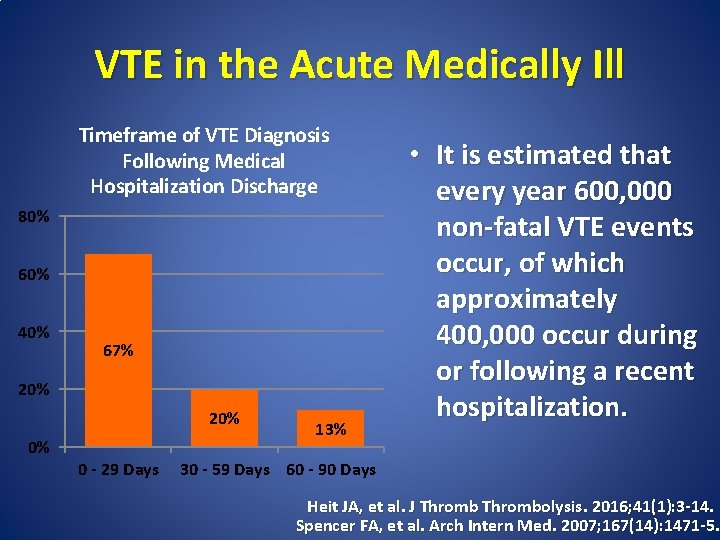
VTE in the Acute Medically Ill Timeframe of VTE Diagnosis Following Medical Hospitalization Discharge 80% 60% 40% 67% 20% 0% 0 - 29 Days 13% • It is estimated that every year 600, 000 non-fatal VTE events occur, of which approximately 400, 000 occur during or following a recent hospitalization. 30 - 59 Days 60 - 90 Days Heit JA, et al. J Thrombolysis. 2016; 41(1): 3 -14. Spencer FA, et al. Arch Intern Med. 2007; 167(14): 1471 -5.
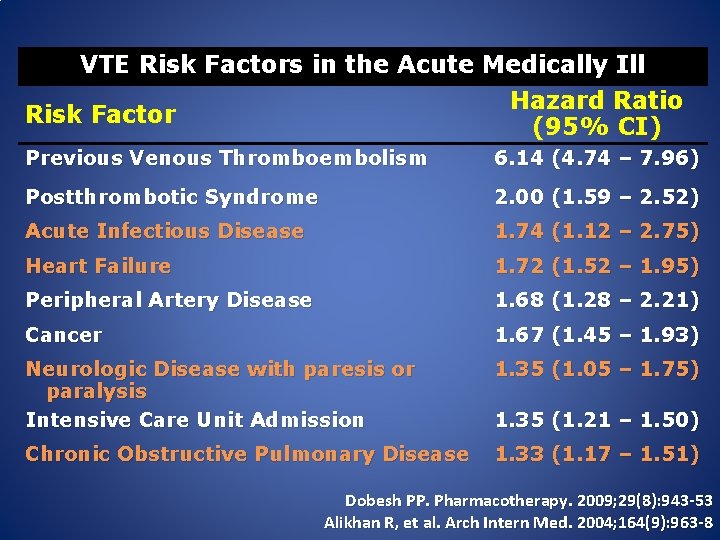
VTE Risk Factors in the Acute Medically Ill Hazard Ratio Risk Factor (95% CI) Previous Venous Thromboembolism 6. 14 (4. 74 – 7. 96) Postthrombotic Syndrome 2. 00 (1. 59 – 2. 52) Acute Infectious Disease 1. 74 (1. 12 – 2. 75) Heart Failure 1. 72 (1. 52 – 1. 95) Peripheral Artery Disease 1. 68 (1. 28 – 2. 21) Cancer 1. 67 (1. 45 – 1. 93) Neurologic Disease with paresis or paralysis Intensive Care Unit Admission 1. 35 (1. 05 – 1. 75) Chronic Obstructive Pulmonary Disease 1. 33 (1. 17 – 1. 51) 1. 35 (1. 21 – 1. 50) Dobesh PP. Pharmacotherapy. 2009; 29(8): 943 -53 Alikhan R, et al. Arch Intern Med. 2004; 164(9): 963 -8

9 th Ed. ACCP Guidelines 2012 • 2. 8 In acutely ill hospitalized patients who receive an initial course of thromboprophylaxis, we suggest against extending the duration of thromboprophylaxis beyond the period of patient immobilization or acute hospital stay (Grade 2 B) Kahn SR, et al. Chest. 2012; 141(2 Suppl): e 195 S-226 S

Extended Duration Thromboprophylaxis • EXCLAIM (enoxaparin) – Extended-duration enoxaparin 40 mg SQ daily vs. placebo • All patients received enoxaparin 40 mg daily for 10 + 4 days prior to randomization • 28 -day treatment period
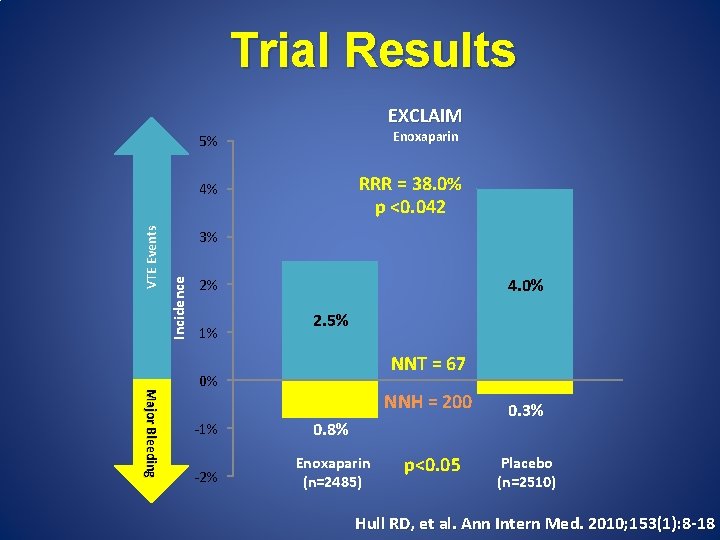
Trial Results 5% Enoxaparin 4% RRR = 38. 0% p <0. 042 3% Incidence VTE Events EXCLAIM 4. 0% 2% 1% 2. 5% NNT = 67 Major Bleeding 0% NNH = 200 -1% 0. 8% -2% Enoxaparin (n=2485) p<0. 05 0. 3% Placebo (n=2510) Hull RD, et al. Ann Intern Med. 2010; 153(1): 8 -18
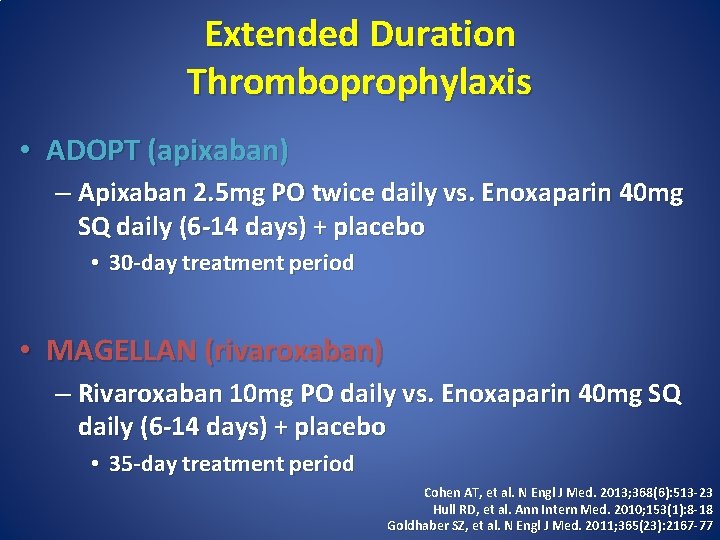
Extended Duration Thromboprophylaxis • ADOPT (apixaban) – Apixaban 2. 5 mg PO twice daily vs. Enoxaparin 40 mg SQ daily (6 -14 days) + placebo • 30 -day treatment period • MAGELLAN (rivaroxaban) – Rivaroxaban 10 mg PO daily vs. Enoxaparin 40 mg SQ daily (6 -14 days) + placebo • 35 -day treatment period Cohen AT, et al. N Engl J Med. 2013; 368(6): 513 -23 Hull RD, et al. Ann Intern Med. 2010; 153(1): 8 -18 Goldhaber SZ, et al. N Engl J Med. 2011; 365(23): 2167 -77
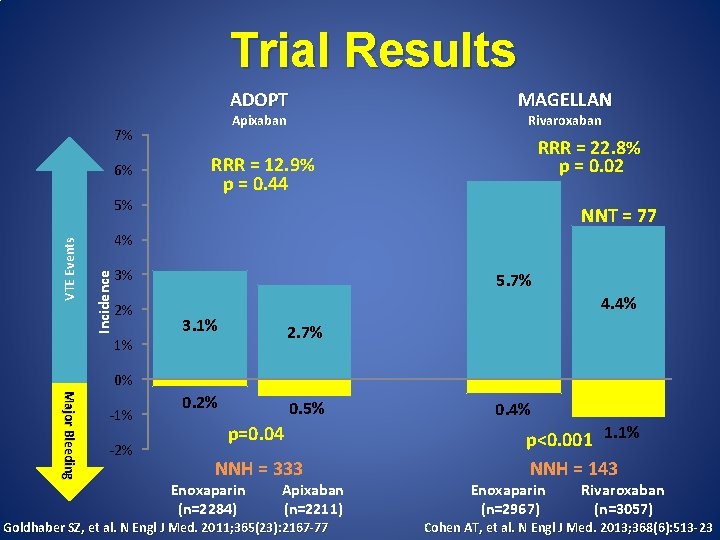
Trial Results ADOPT Apixaban 7% 6% Rivaroxaban RRR = 22. 8% p = 0. 02 RRR = 12. 9% p = 0. 44 NNT = 77 4% Incidence VTE Events 5% MAGELLAN 3% 2% 1% 5. 7% 4. 4% 3. 1% 2. 7% 0. 2% 0. 5% 0% Major Bleeding -1% -2% p=0. 04 NNH = 333 Enoxaparin (n=2284) Apixaban (n=2211) Goldhaber SZ, et al. N Engl J Med. 2011; 365(23): 2167 -77 0. 4% p<0. 001 1. 1% NNH = 143 Enoxaparin (n=2967) Rivaroxaban (n=3057) Cohen AT, et al. N Engl J Med. 2013; 368(6): 513 -23

Bottom Line • Despite positive results in VTE reduction in EXCLAIM and MAGELLAN, all previous trials come at the expense of increased major bleeding • Trials have varied in treatment duration and risk population • Studies indicate that there might be a need for extended prophylaxis, but the right mix of drug, dose, duration and population has not yet been found

Acute Medically Ill VTE Prevention with EXtended Duration Betrixaban A Multicenter, Randomized, Active-Controlled Efficacy and Safety Study Comparing Extended Duration Betrixaban with Standard of Care Enoxaparin for the Prevention of Venous Thromboembolism in Acute Medically Ill Patients Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
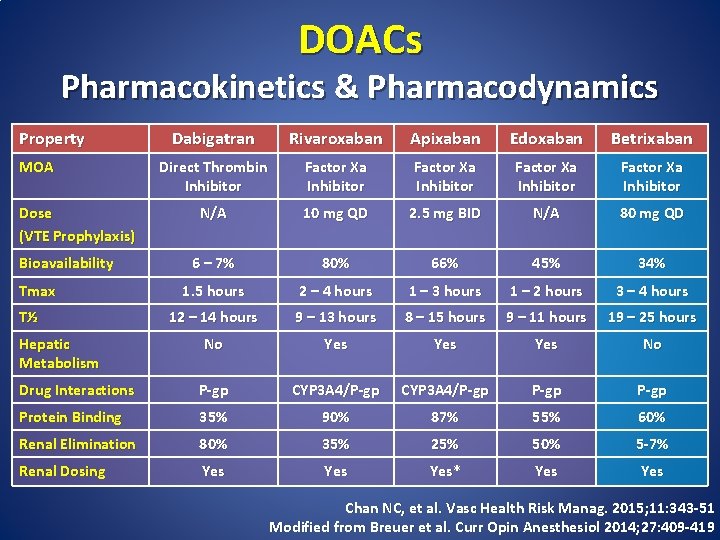
DOACs Pharmacokinetics & Pharmacodynamics Property Dabigatran Rivaroxaban Apixaban Edoxaban Betrixaban Direct Thrombin Inhibitor Factor Xa Inhibitor N/A 10 mg QD 2. 5 mg BID N/A 80 mg QD 6 – 7% 80% 66% 45% 34% 1. 5 hours 2 – 4 hours 1 – 3 hours 1 – 2 hours 3 – 4 hours 12 – 14 hours 9 – 13 hours 8 – 15 hours 9 – 11 hours 19 – 25 hours No Yes Yes No Drug Interactions P-gp CYP 3 A 4/P-gp Protein Binding 35% 90% 87% 55% 60% Renal Elimination 80% 35% 25% 50% 5 -7% Renal Dosing Yes Yes* Yes MOA Dose (VTE Prophylaxis) Bioavailability Tmax T½ Hepatic Metabolism Chan NC, et al. Vasc Health Risk Manag. 2015; 11: 343 -51 Modified from Breuer et al. Curr Opin Anesthesiol 2014; 27: 409 -419

APEX - Objective • Demonstrate superiority of extended duration anticoagulation (35 -42 days) with betrixaban as compared to standard of care anticoagulation (6 -14 days) with enoxaparin for VTE prevention in the acute medically ill Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
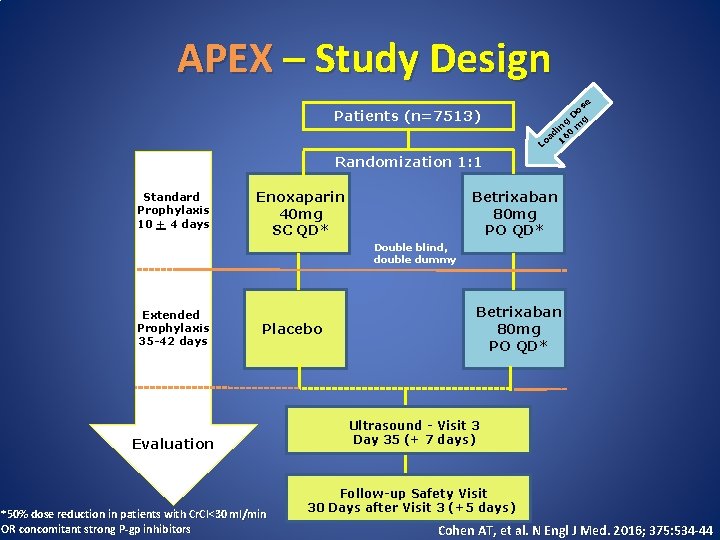
APEX – Study Design Patients (n=7513) e os D g g in 0 m d oa 16 L Randomization 1: 1 Standard Prophylaxis 10 + 4 days Enoxaparin 40 mg SC QD* Betrixaban 80 mg PO QD* Double blind, double dummy Extended Prophylaxis 35 -42 days Placebo Evaluation *50% dose reduction in patients with Cr. Cl<30 ml/min OR concomitant strong P-gp inhibitors Betrixaban 80 mg PO QD* Ultrasound - Visit 3 Day 35 (+ 7 days) Follow-up Safety Visit 30 Days after Visit 3 (+5 days) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX – Patient Population • Inclusion Criteria: – Age/Risk Factors: • > 75 years old OR • 60 – 74 years with D-dimer > 2 x ULN OR • 40 – 59 years with D-dimer > 2 x ULN and a history of either VTE or cancer – Hospitalized for one of the following acute presentation: • • • Acute on chronic heart failure decompensation Acute on chronic respiratory failure Acute infection without septic shock Acute rheumatic disorders Acute ischemic stroke (w/ immobilization) – Anticipated to be severely immobilized for at least 24 hours after randomization and expected to be severely or moderately immobile for > 3 days – Anticipated length of hospitalization > 3 days Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX – Patient Population • Selected Exclusion Criteria: – End stage renal disease with Cr. Cl <15 ml/min, or requiring dialysis – Body weight <45 kg – Anticipated need for prolonged anticoagulation or concomitant dual antiplatelet therapy – Major surgery or procedure within 3 months prior to enrollment or anticipated during study period – History of clinically significant bleeding within 6 months prior to enrollment • History of significant GI, pulmonary or GU bleeding, ongoing chronic peptic ulcer disease or acute gastritis within 2 years prior to enrollment • History of intracranial bleeding (<3 years), head trauma (<3 months), or known intracranial lesions – Hemoglobin < 9. 5 g/d. L on two consecutive measurements or unstable/declining hemoglobin – Liver disease including cirrhosis; LFTs >3 x ULN – Receipt of parenteral anticoagulant >96 hours or receipt of oral anticoagulant <96 hours prior to enrollment Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX – Methodology • In 2012, the FDA published a guidance document that recommended “identifying patients with the greatest likelihood of having a disease related endpoint and that are more likely to respond to drug treatment” in order to “enrich” the study population and increase the likelihood of finding a benefit • In response to this, the APEX protocol was amended and designed to enroll pre-specified high risk subpopulations with elevated D-dimer and age >75 – All based on the findings from the MAGELLAN study Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
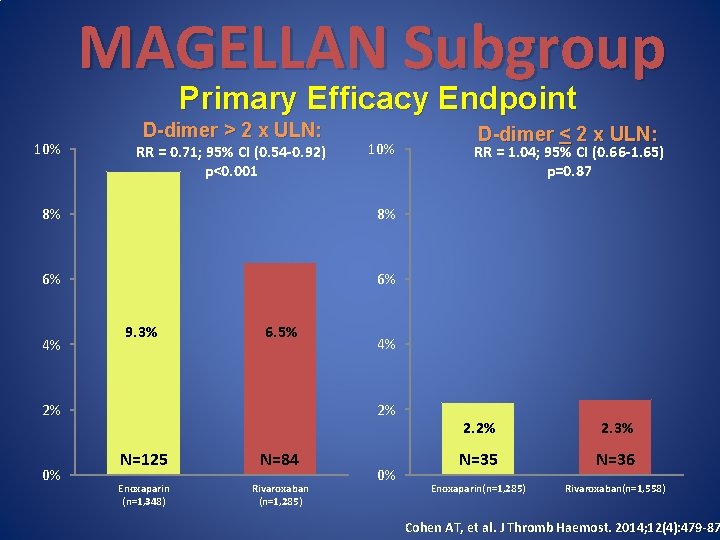
MAGELLAN Subgroup Primary Efficacy Endpoint 10% D-dimer > 2 x ULN: RR = 0. 71; 95% CI (0. 54 -0. 92) p<0. 001 10% 8% 8% 6% 6% 4% 9. 3% 6. 5% 0% N=125 N=84 Enoxaparin (n=1, 348) Rivaroxaban (n=1, 285) RR = 1. 04; 95% CI (0. 66 -1. 65) p=0. 87 4% 2% 2% D-dimer < 2 x ULN: 0% 2. 2% 2. 3% N=35 N=36 Enoxaparin(n=1, 285) Rivaroxaban(n=1, 558) Cohen AT, et al. J Thromb Haemost. 2014; 12(4): 479 -87
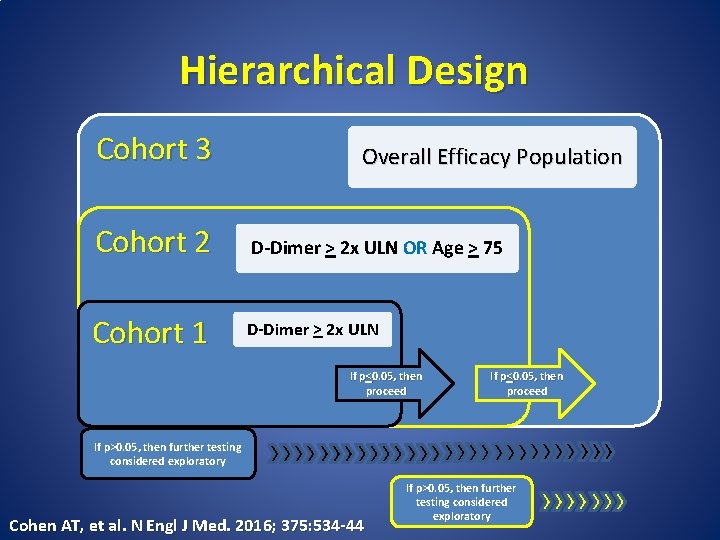
Hierarchical Design Cohort 3 Overall Efficacy Population Cohort 2 D-Dimer > 2 x ULN OR Age > 75 Cohort 1 D-Dimer > 2 x ULN If p<0. 05, then proceed If p>0. 05, then further testing considered exploratory Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44 If p>0. 05, then further testing considered exploratory

APEX – Statistical Analysis • Sample size calculation determined during study (based on expected results similar to MAGELLAN) resulting in final sample size to maintain a power of 85% at 1 -sided alpha 0. 005 in Cohort 1 • Primary and secondary outcomes using chi-square tests with type I error of 0. 05 • Analysis of all efficacy outcomes conducted via Cochran-Mantel-Haenszel tests to calculate relative risk ratio and incidence rates, which was further stratified by the two randomization factors of elevated D-dimer and dose assignment (80 mg/40 mg) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX - Endpoints • Primary Efficacy Endpoint – Composite of asymptomatic proximal DVT, symptomatic proximal or distal DVT, non-fatal PE or VTE-related death • Secondary Efficacy Endpoints – Symptomatic VTE – Composite of adjudicated asymptomatic proximal DVT, symptomatic proximal or distal DVT, non-fatal PE or allcause death • Primary Safety Endpoint – Major bleeding (ISTH definition) • Net Clinical Benefit – Composite of primary efficacy and safety endpoints Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
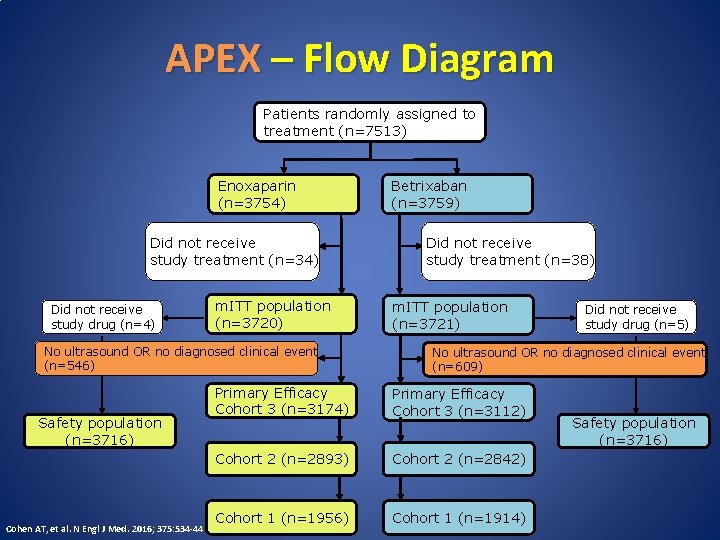
APEX – Flow Diagram Patients randomly assigned to treatment (n=7513) Enoxaparin (n=3754) Did not receive study treatment (n=34) Did not receive study drug (n=4) m. ITT population (n=3720) No ultrasound OR no diagnosed clinical event (n=546) Safety population Did not receive study drug (n=5) (n=3716) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44 Betrixaban (n=3759) Did not receive study treatment (n=38) m. ITT population (n=3721) Did not receive study drug (n=5) No ultrasound OR no diagnosed clinical event (n=609) Primary Efficacy Cohort 3 (n=3174) Primary Efficacy Cohort 3 (n=3112) Cohort 2 (n=2893) Cohort 2 (n=2842) Cohort 1 (n=1956) Cohort 1 (n=1914) Safety population (n=3716)
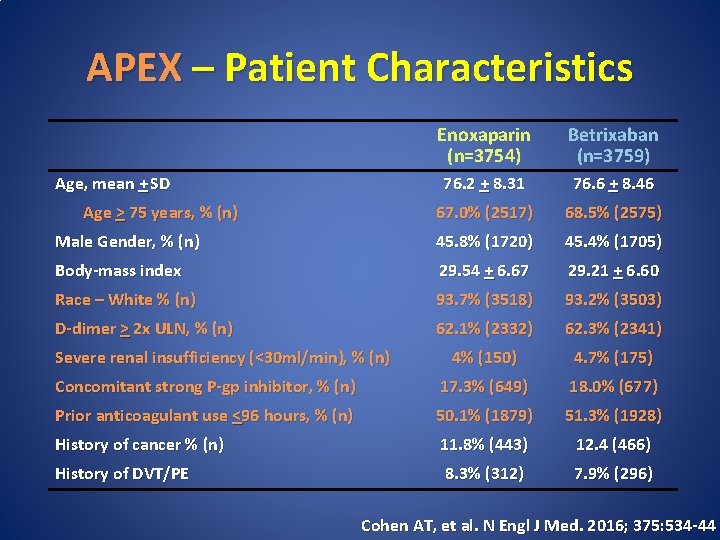
APEX – Patient Characteristics Enoxaparin (n=3754) Betrixaban (n=3759) 76. 2 + 8. 31 76. 6 + 8. 46 67. 0% (2517) 68. 5% (2575) Male Gender, % (n) 45. 8% (1720) 45. 4% (1705) Body-mass index 29. 54 + 6. 67 29. 21 + 6. 60 Race – White % (n) 93. 7% (3518) 93. 2% (3503) D-dimer > 2 x ULN, % (n) 62. 1% (2332) 62. 3% (2341) 4% (150) 4. 7% (175) Concomitant strong P-gp inhibitor, % (n) 17. 3% (649) 18. 0% (677) Prior anticoagulant use <96 hours, % (n) 50. 1% (1879) 51. 3% (1928) History of cancer % (n) 11. 8% (443) 12. 4 (466) History of DVT/PE 8. 3% (312) 7. 9% (296) Age, mean + SD Age > 75 years, % (n) Severe renal insufficiency (<30 ml/min), % (n) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX – Patient Characteristics (Cont. ) Primary Reason for Hospital Admission Enoxaparin (n=3754) Betrixaban (n=3759) Acute CHF NYHA III-IV, % (n) Acute infection, % (n) Acute respiratory failure, % (n) Acute ischemic stroke % (n) Acute rheumatic disorder, % (n) 44. 5% (1672) 28. 2% (1058) 12. 6% (474) 11. 5% (432) 3. 1% (117) 44. 6% (1677) 29. 6% (1112) 11. 9% (448) 10. 9% (411) 2. 9% (1. 9) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

APEX - Results Primary Efficacy Endpoint Composite of Asymptomatic Proximal DVT, Symptomatic Proximal or Distal DVT, Non-fatal PE, or VTE-Related Death 10% 9% Event Rate (%) 8% 7% p=0. 054 RRR 19. 4% 5% 3% NNT = 58 p=0. 029 p=0. 006 RRR 20% 6% 4% NNT = 70 8. 49% 6. 90% 5. 63% RRR 24% 7. 05% 5. 30% 2% 1% 0% N=132 N=166 N=160 7. 03% N=204 N=165 N=223 Betrixaban Enoxaparin Cohort 1 Cohort 2 Cohort 3 Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
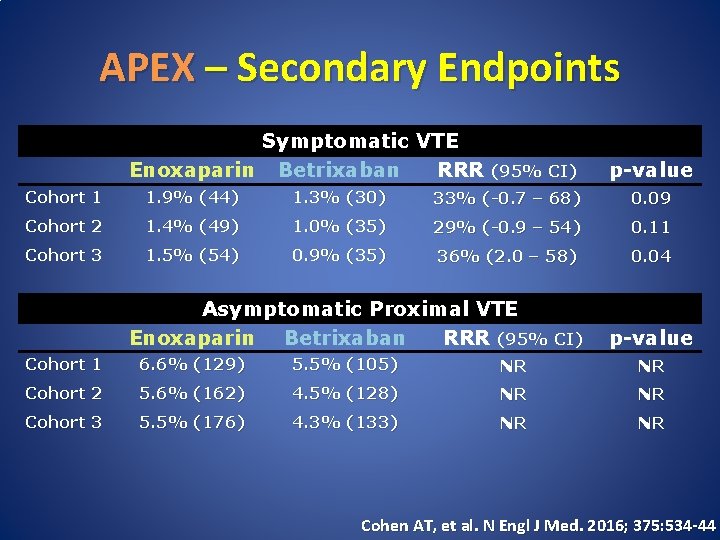
APEX – Secondary Endpoints Symptomatic VTE Enoxaparin Betrixaban RRR (95% CI) p-value Cohort 1 1. 9% (44) 1. 3% (30) 33% (-0. 7 – 68) 0. 09 Cohort 2 1. 4% (49) 1. 0% (35) 29% (-0. 9 – 54) 0. 11 Cohort 3 1. 5% (54) 0. 9% (35) 36% (2. 0 – 58) 0. 04 Asymptomatic Proximal VTE Enoxaparin Betrixaban RRR (95% CI) p-value Cohort 1 6. 6% (129) 5. 5% (105) NR NR Cohort 2 5. 6% (162) 4. 5% (128) NR NR Cohort 3 5. 5% (176) 4. 3% (133) NR NR Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
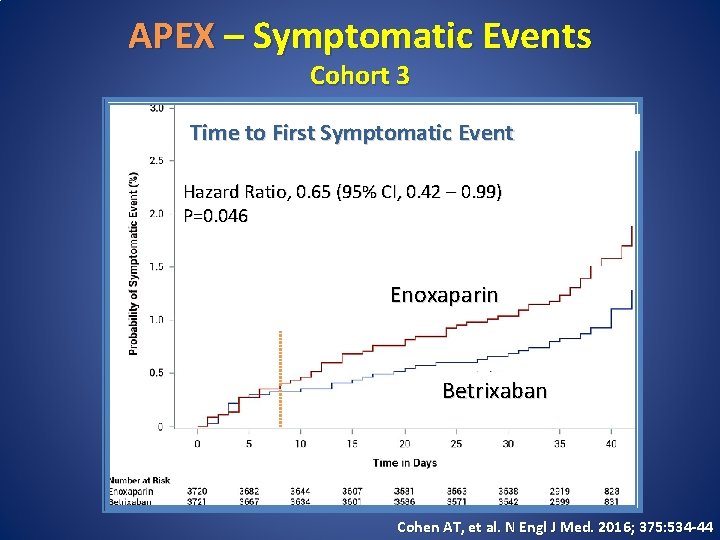
APEX – Symptomatic Events Cohort 3 Time to First Symptomatic Event Hazard Ratio, 0. 65 (95% CI, 0. 42 – 0. 99) P=0. 046 Enoxaparin Betrixaban Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
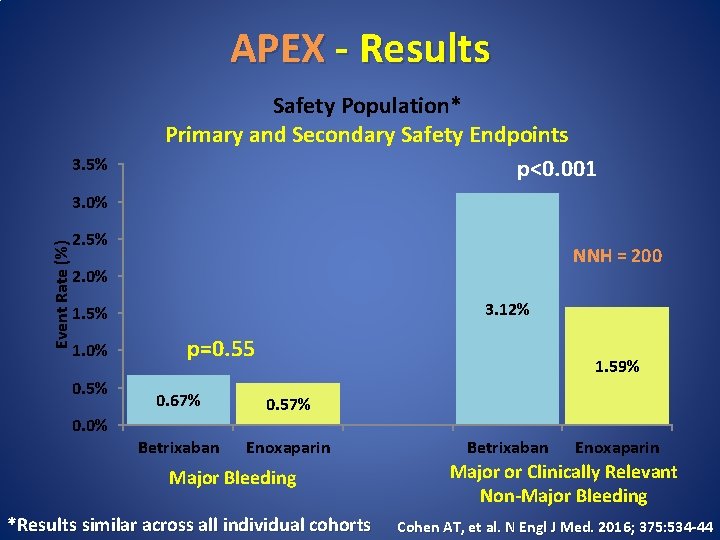
APEX - Results 3. 5% Safety Population* Primary and Secondary Safety Endpoints p<0. 001 Event Rate (%) 3. 0% 2. 5% NNH = 200 2. 0% 3. 12% 1. 5% 1. 0% 0. 5% 0. 0% p=0. 55 1. 59% 0. 67% 0. 57% Betrixaban Enoxaparin Major Bleeding *Results similar across all individual cohorts Betrixaban Enoxaparin Major or Clinically Relevant Non-Major Bleeding Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
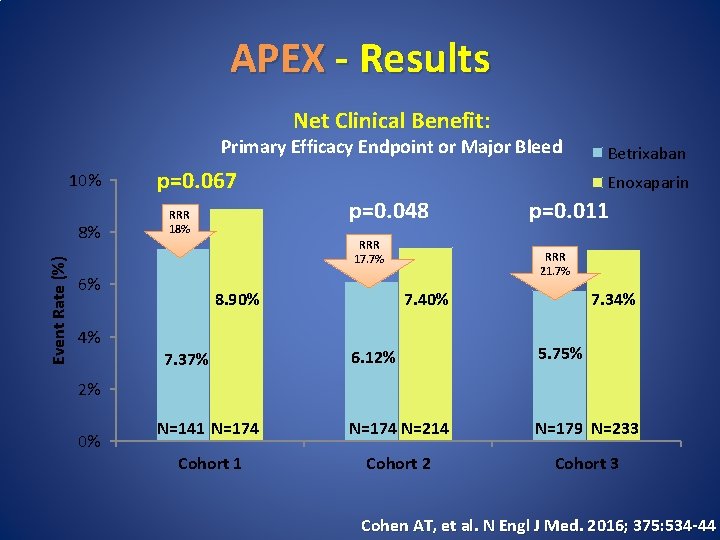
APEX - Results Net Clinical Benefit: Primary Efficacy Endpoint or Major Bleed 10% Event Rate (%) 8% p=0. 067 Enoxaparin p=0. 048 RRR 18% RRR 17. 7% 6% 8. 90% 4% Betrixaban p=0. 011 RRR 21. 7% 7. 34% 7. 40% 6. 12% 5. 75% N=141 N=174 N=214 N=179 N=233 Cohort 1 Cohort 2 Cohort 3 7. 37% 2% 0% Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
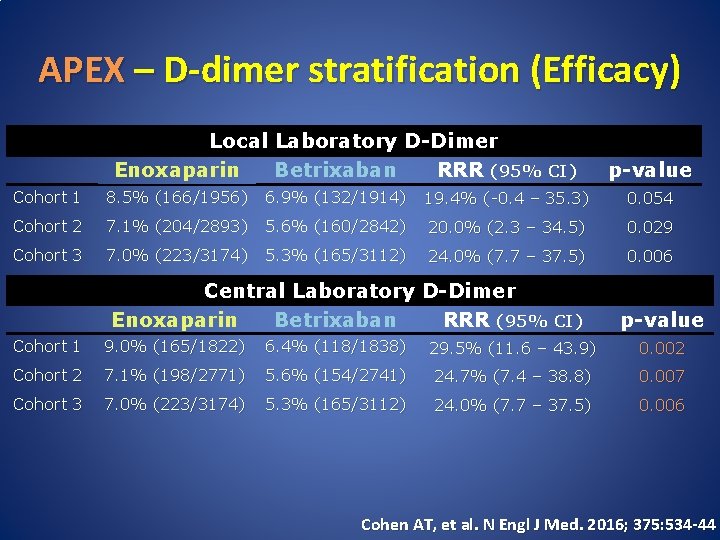
APEX – D-dimer stratification (Efficacy) Local Laboratory D-Dimer Enoxaparin Betrixaban RRR (95% CI) p-value Cohort 1 8. 5% (166/1956) 6. 9% (132/1914) 19. 4% (-0. 4 – 35. 3) 0. 054 Cohort 2 7. 1% (204/2893) 5. 6% (160/2842) 20. 0% (2. 3 – 34. 5) 0. 029 Cohort 3 7. 0% (223/3174) 5. 3% (165/3112) 24. 0% (7. 7 – 37. 5) 0. 006 Central Laboratory D-Dimer Enoxaparin Betrixaban RRR (95% CI) p-value Cohort 1 9. 0% (165/1822) 6. 4% (118/1838) 29. 5% (11. 6 – 43. 9) 0. 002 Cohort 2 7. 1% (198/2771) 5. 6% (154/2741) 24. 7% (7. 4 – 38. 8) 0. 007 Cohort 3 7. 0% (223/3174) 5. 3% (165/3112) 24. 0% (7. 7 – 37. 5) 0. 006 Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
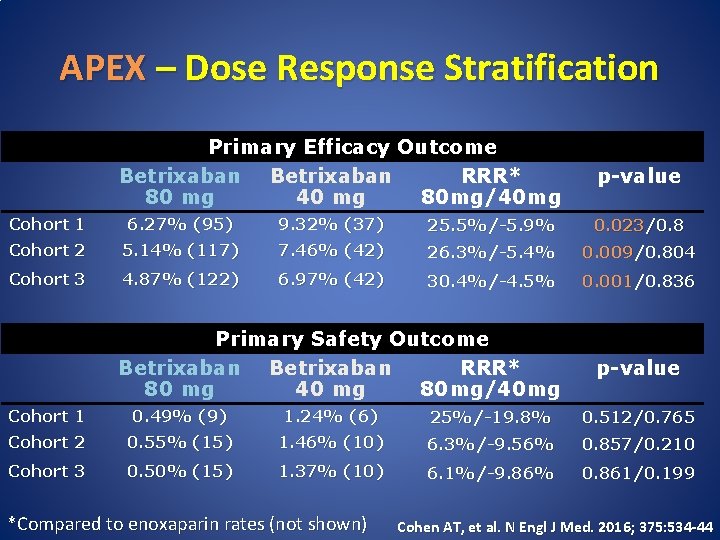
APEX – Dose Response Stratification Primary Efficacy Outcome Betrixaban RRR* 80 mg 40 mg 80 mg/40 mg p-value Cohort 1 6. 27% (95) 9. 32% (37) 25. 5%/-5. 9% 0. 023/0. 8 Cohort 2 5. 14% (117) 7. 46% (42) 26. 3%/-5. 4% 0. 009/0. 804 Cohort 3 4. 87% (122) 6. 97% (42) 30. 4%/-4. 5% 0. 001/0. 836 Primary Safety Outcome Betrixaban RRR* 80 mg 40 mg 80 mg/40 mg p-value Cohort 1 0. 49% (9) 1. 24% (6) 25%/-19. 8% 0. 512/0. 765 Cohort 2 0. 55% (15) 1. 46% (10) 6. 3%/-9. 56% 0. 857/0. 210 Cohort 3 0. 50% (15) 1. 37% (10) 6. 1%/-9. 86% 0. 861/0. 199 *Compared to enoxaparin rates (not shown) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
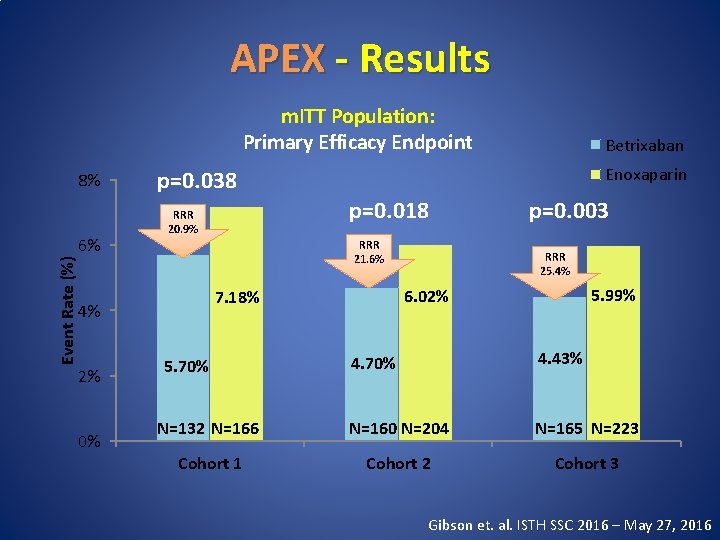
APEX - Results m. ITT Population: Primary Efficacy Endpoint 8% Event Rate (%) 6% 0% Enoxaparin p=0. 038 p=0. 018 RRR 20. 9% RRR 21. 6% p=0. 003 RRR 25. 4% 5. 99% 6. 02% 7. 18% 4% 2% Betrixaban 4. 70% 4. 43% N=132 N=166 N=160 N=204 N=165 N=223 Cohort 1 Cohort 2 Cohort 3 5. 70% Gibson et. al. ISTH SSC 2016 – May 27, 2016

APEX – Author’s Conclusion • Based on protocol-specified hierarchical analysis, there was no significant difference in primary outcome in patients with D-dimer >2 x ULN • However, when other larger and more inclusive cohorts are taken into account, there is evidence to suggest that the risk of VTE was lower with betrixaban than with enoxaparin+placebo Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

Limitations/Thoughts • Hierarchical analysis – trial design – What would we be saying if the cohorts were flipped? – Superiority trial – on treatment analysis? • Overestimation of dose reduction in patients with renal insufficiency/drug-interactions – First study to include patients with Cr. Cl <30 ml/min – Enoxaparin 20 mg daily • Generalizability – > 90% of patients were white

Limitations/Thoughts • D-Dimer – Specificity and sensitivity – Central (exploratory) vs. local (pre-specified) laboratory results • In-hospital period results – Benefit to oral versus subcutaneous injection prophylaxis? • Optimal duration of therapy – 35 – 42 days – Perhaps longer?

Limitations/Thoughts • Clinical vs. Statistical Relevance – Underpowered? • For the primary outcome in Cohort 1, betrixaban was associated with a 19% reduction in events (p=0. 054) • 15% of patients were not included in analysis due to incomplete diagnostics • The authors acknowledge that Cohort 1 may have been underpowered as the sample size calculation was based on minimum number of patients in the overall cohort – Overpowered? • Alpha of 0. 005 used in power calculation versus alpha of 0. 05 used in statistical analysis • Statistical significance found in Cohorts 2 and 3, but not Cohort ever managed to achieve a priori clinically significant 35% reduction
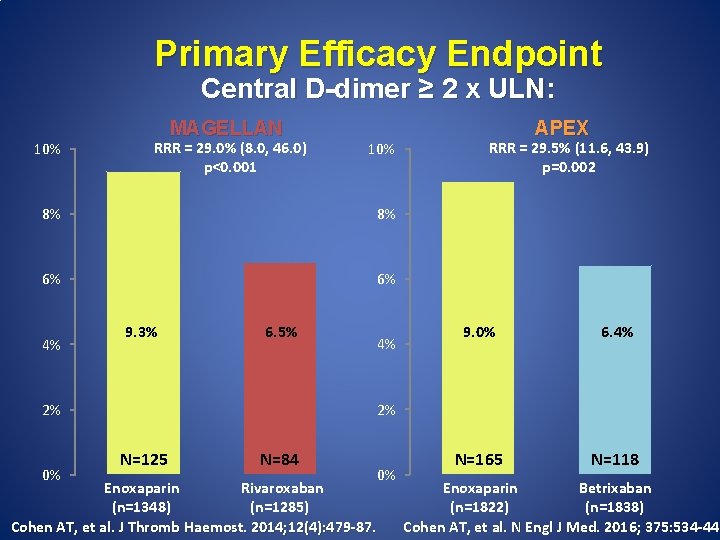
Primary Efficacy Endpoint Central D-dimer ≥ 2 x ULN: 10% MAGELLAN RRR = 29. 0% (8. 0, 46. 0) p<0. 001 10% 8% 8% 6% 6% 4% 9. 3% 6. 5% RRR = 29. 5% (11. 6, 43. 9) p=0. 002 9. 0% 6. 4% N=165 N=118 2% 2% 0% 4% APEX N=125 N=84 Enoxaparin Rivaroxaban (n=1348) (n=1285) Cohen AT, et al. J Thromb Haemost. 2014; 12(4): 479 -87. 0% Enoxaparin Betrixaban (n=1822) (n=1838) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44

Summary • Extended-duration betrixaban resulted in a non-significant reduction of the primary efficacy outcome in patients with a D-Dimer >2 x ULN prior to randomization (p=0. 054); however, there was significant benefit in the overall efficacy study population (p=0. 006) • Betrixaban was associated with a reduction in the rate of symptomatic events through 35 days in the overall study population (p=0. 003), but not in the first two pre-defined cohorts (p=0. 09 and p=0. 11). • Stratification through central laboratory D-dimer, was associated a statistically significant reduction in the primary efficacy endpoint with betrixaban in the first primary analysis cohort (p=0. 002) • Betrixaban was not associated with a significant increase in major (p=0. 55) or fatal bleeding in the overall safety population; however was associated with an increase in major plus non-major clinically relevant bleeding (p<0. 001) • Betrixaban had a favorable net-clinical benefit in the overall efficacy study population. (p=0. 011) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44
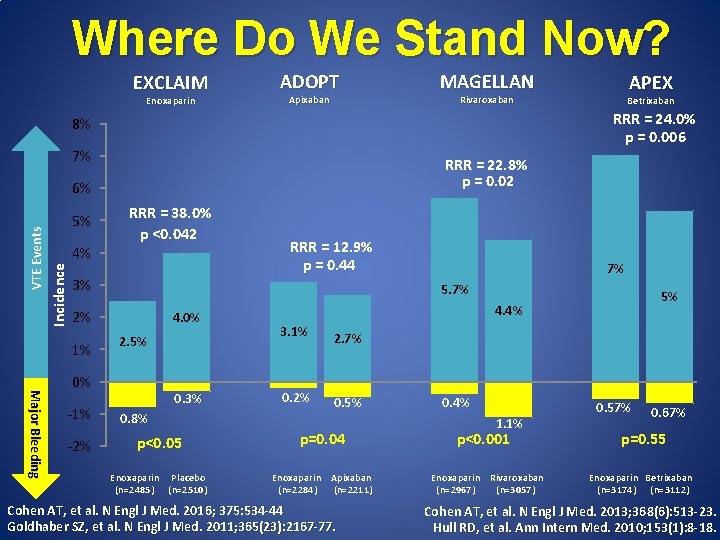
Where Do We Stand Now? EXCLAIM Enoxaparin ADOPT Apixaban MAGELLAN 7% RRR = 22. 8% p = 0. 02 Incidence VTE Events 6% 4% RRR = 38. 0% p <0. 042 RRR = 12. 9% p = 0. 44 3% 4. 0% 2. 5% Major Bleeding 0% -1% -2% 7% 5. 7% 2% 1% Betrixaban RRR = 24. 0% p = 0. 006 8% 5% APEX Rivaroxaban 0. 3% 0. 8% 5% 4. 4% 3. 1% 2. 7% 0. 2% 0. 5% p<0. 05 p=0. 04 Enoxaparin Placebo (n=2485) (n=2510) Enoxaparin Apixaban (n=2284) (n=2211) Cohen AT, et al. N Engl J Med. 2016; 375: 534 -44 Goldhaber SZ, et al. N Engl J Med. 2011; 365(23): 2167 -77. 0. 4% 1. 1% p<0. 001 Enoxaparin Rivaroxaban (n=2967) (n=3057) 0. 57% 0. 67% p=0. 55 Enoxaparin Betrixaban (n=3174) (n=3112) Cohen AT, et al. N Engl J Med. 2013; 368(6): 513 -23. Hull RD, et al. Ann Intern Med. 2010; 153(1): 8 -18.
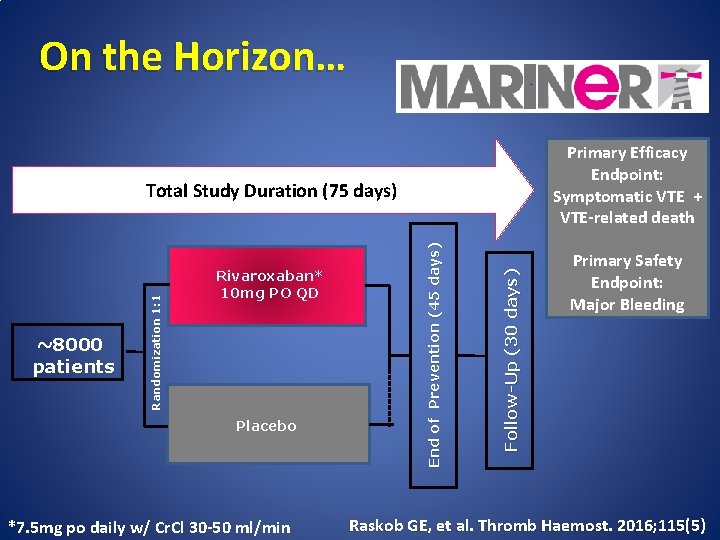
On the Horizon… Primary Efficacy Endpoint: Symptomatic VTE + VTE-related death Placebo *7. 5 mg po daily w/ Cr. Cl 30 -50 ml/min Follow-Up (30 days) Rivaroxaban* 10 mg PO QD End of Prevention (45 days) ~8000 patients Randomization 1: 1 Total Study Duration (75 days) Primary Safety Endpoint: Major Bleeding Raskob GE, et al. Thromb Haemost. 2016; 115(5)

Questions?

References • • • • Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9 th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012; 141(2 Suppl): e 195 S-226 S. Cohen AT, Harrington RA, Goldhaber SZ, et al. Extended Thromboprophylaxis with Betrixaban in Acutely Ill Medical Patients. N Engl J Med. 2016 ; 375: 534 -44 Cohen AT, Harrington R, Goldhaber SZ, et al. The design and rationale for the Acute Medically Ill Venous Thromboembolism Prevention with Extended Duration Betrixaban (APEX) study. Am Heart J. 2014; 167(3): 335 -41. Goldhaber SZ, Leizorovicz A, Kakkar AK, et al. Apixaban versus enoxaparin for thromboprophylaxis in medically ill patients. N Engl J Med. 2011; 365(23): 2167 -77. Raskob GE, Spyropoulos AC, Zrubek J, et al. The MARINER trial of rivaroxaban after hospital discharge for medical patients at high risk of VTE. Design, rationale, and clinical implications. Thromb Haemost. 2016; 115(5 ) Cohen AT, Spiro TE, Büller HR, et al. Rivaroxaban for thromboprophylaxis in acutely ill medical patients. N Engl J Med. 2013; 368(6): 513 -23. Hull RD, Schellong SM, Tapson VF, et al. Extended-duration venous thromboembolism prophylaxis in acutely ill medical patients with recently reduced mobility: a randomized trial. Ann Intern Med. 2010; 153(1): 8 -18. Heit JA, Spencer FA, White RH. The epidemiology of venous thromboembolism. J Thrombolysis. 2016; 41(1): 3 -14. Chan NC, Bhagirath V, Eikelboom JW. Profile of betrixaban and its potential in the prevention and treatment of venous thromboembolism. Vasc Health Risk Manag. 2015; 11: 343 -51. Cohen AT, Spiro TE, Spyropoulos AC, et al. D-dimer as a predictor of venous thromboembolism in acutely ill, hospitalized patients: a subanalysis of the randomized controlled MAGELLAN trial. J Thromb Haemost. 2014; 12(4): 479 -87. Dobesh PP. Economic burden of venous thromboembolism in hospitalized patients. Pharmacotherapy. 2009; 29(8): 943 -53. Alikhan R, Cohen AT, Combe S, et al. Risk factors for venous thromboembolism in hospitalized patients with acute medical illness: analysis of the MEDENOX Study. Arch Intern Med. 2004; 164(9): 963 -8. Spyropoulos AC, Anderson FA, Fitzgerald G, et al. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011; 140(3): 706 - 14. Spencer FA, Lessard D, Emery C, Reed G, Goldberg RJ. Venous thromboembolism in the outpatient setting. Arch Intern Med. 2007; 167: 1471– 5.
- Slides: 43