EXSCEL Primary Results Presentation EASD Lisbon Portugal Thursday

EXSCEL Primary Results Presentation EASD Lisbon, Portugal Thursday 14 th September, 2017

Study Rationale, Design and Conduct Robert J. Mentz, MD Duke Clinical Research Institute

Presenter Disclosure Information Research support: • Astra. Zeneca, Amgen, Glaxo. Smith. Kline Advisory boards: • Amgen, Boehringer-Ingelheim
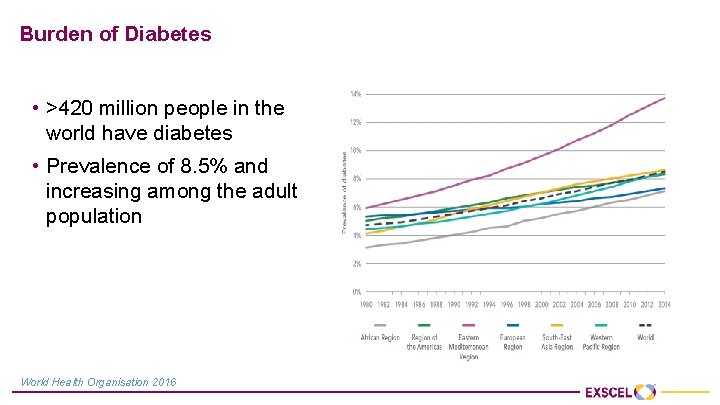
Burden of Diabetes • >420 million people in the world have diabetes • Prevalence of 8. 5% and increasing among the adult population World Health Organisation 2016
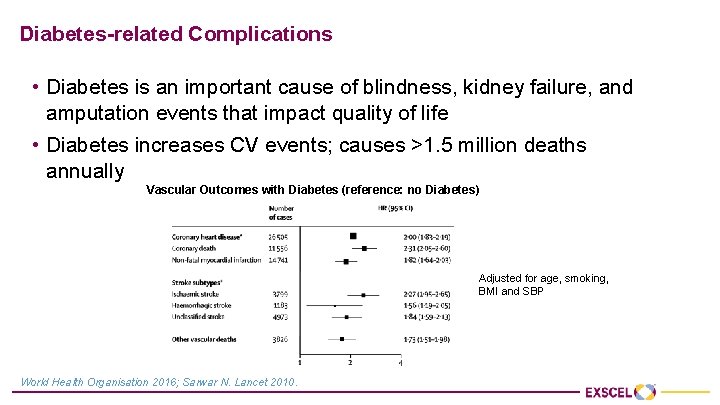
Diabetes-related Complications • Diabetes is an important cause of blindness, kidney failure, and amputation events that impact quality of life • Diabetes increases CV events; causes >1. 5 million deaths annually Vascular Outcomes with Diabetes (reference: no Diabetes) Adjusted for age, smoking, BMI and SBP World Health Organisation 2016; Sarwar N. Lancet 2010.

Evaluating Cardiovascular Risk in New Antidiabetic Therapies • Regulatory requirement that new therapies to treat T 2 DM both effectively lower glucose levels and demonstrate evidence of cardiovascular safety • Large cardiovascular outcomes trials required for novel T 2 DM therapies with robust assessment of cardiovascular end-points and important safety events

EXSCEL • Large, pragmatic, international trial designed to characterise the effects of once weekly GLP-1 receptor agonist, exenatide, on CVrelated outcomes in patients with T 2 DM, when added to usual diabetes care • Double-blind, placebo-controlled trial randomising participants to exenatide 2 mg once weekly injection or matching placebo • Academically led by the Diabetes Trials Unit, University of Oxford and the Duke Clinical Research Institute in collaboration with industry sponsorship
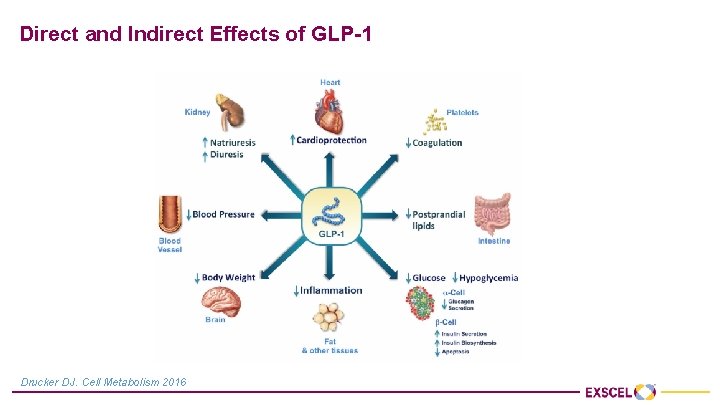
Direct and Indirect Effects of GLP-1 Drucker DJ. Cell Metabolism 2016

Pragmatic Study Design • Integration with usual care ‒ Management of diabetes, CV risk factors and pre-existing CV disease remained responsibility of usual care provider based on local practice patterns and guidelines ‒ Concomitant DM therapy at discretion of usual providers ‒ Concomitant open-label GLP-1 RA prohibited • Streamlined trial conduction ‒ Broad inclusion/exclusion criteria representative of patient population ‒ Wide range of CV risk; no enrichment for elderly individuals ‒ DPP-4 inhibitors permitted ‒ No run-in period ‒ Visits every 6 months to minimise interference with usual care ‒ All efficacy and safety (except for annual calcitonin) laboratory results collected as available from the usual care setting
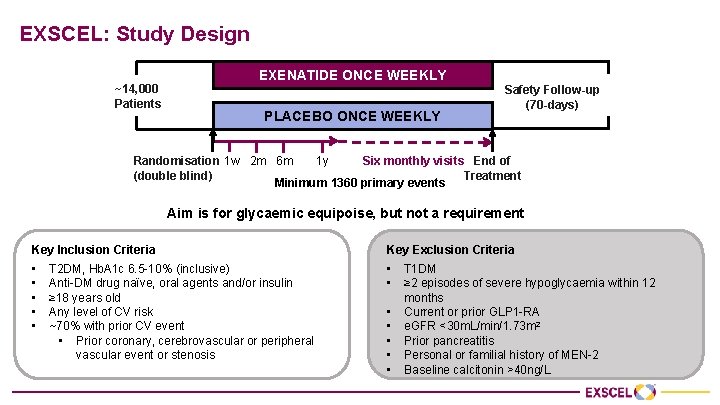
EXSCEL: Study Design ~14, 000 Patients EXENATIDE ONCE WEEKLY PLACEBO ONCE WEEKLY Safety Follow-up (70 -days) Six monthly visits End of Randomisation 1 w 2 m 6 m 1 y (double blind) Treatment Minimum 1360 primary events Aim is for glycaemic equipoise, but not a requirement Key Inclusion Criteria Key Exclusion Criteria • • T 2 DM, Hb. A 1 c 6. 5 -10% (inclusive) Anti-DM drug naïve, oral agents and/or insulin ≥ 18 years old Any level of CV risk ~70% with prior CV event • Prior coronary, cerebrovascular or peripheral vascular event or stenosis • • • T 1 DM ≥ 2 episodes of severe hypoglycaemia within 12 months Current or prior GLP 1 -RA e. GFR <30 m. L/min/1. 73 m 2 Prior pancreatitis Personal or familial history of MEN-2 Baseline calcitonin >40 ng/L

Primary and Secondary Endpoints Primary Endpoint Secondary Endpoints Time to first occurrence of 3 -point MACE composed of: • CV death • Non-fatal MI • Non-fatal stroke Safety hypothesis (non-inferiority) and efficacy hypothesis (superiority) Time to first occurrence of: 1) All-cause mortality 2) CV-related death 3) Fatal or non-fatal MI 4) Fatal or non-fatal stroke 5) Hospitalisation for acute coronary syndrome (ACS) 6) Hospitalisation for heart failure (HF)

Additional Key Objectives • Time to initiation of next antihyperglycaemic agent • Number of episodes of severe hypoglycaemia (hypoglycaemia requiring assistance) • Changes in markers of CV risk • Hb. A 1 c, weight, blood pressure

Power and Sample Size Calculations • Non-inferiority (safety) • Assuming true HR of 1. 00, 611 confirmed primary events provides 90% power to exclude the upper bound of the 95% CI of 1. 3 • Superiority (efficacy) • Assuming true HR of 0. 85, 1360 confirmed primary events provides 85% power with a 2 -sided α=0. 05 • ~14, 000 randomised participants with primary CV composite event rate of 2. 2% per year required to achieve target of 1360 confirmed primary CV composite endpoints

Statistical Analysis Plan • Primary non-inferiority safety analysis for MACE-3 • Non-inferiority if upper limit of two-sided CI for the estimated HR <1. 3 • If the objective for safety is met, the efficacy objective will be considered • Prespecified conditional hierarchical analyses, each at one sided α=0. 025 • • All-cause death Each of the components of the primary composite endpoint Hospitalisation for ACS Hospitalisation for HF • Analysis sets • Intention-to-treat population (primary analysis) • Safety population (all participants in the intention-to-treat population who took at least one dose of study medication)

Protocol-Specified Study Drug Discontinuation Criteria • Severe hypoglycaemia: ≥ 2 episodes since the prior trial visit despite down-titration or discontinuation of non-trial antihyperglycaemic agents • Renal dysfunction: Severe, irreversible (confirmed by two consecutive e. GFR <30 ml/min/1. 73 m 2) or renal replacement therapy • Calcitonin: Annual measurement ≥ 50 ng/L

Safety Documentation and Event Adjudication Safety and Event Documentation Event Adjudication* • Detailed collection of all primary and secondary endpoints • Non-serious AEs generally not recorded (only pancreatitis, neoplasms) • Serious adverse events (SAEs) documented using traditional collection and reporting procedures, with the exception of Clinical Events of Interest: • CV events (e. g. procedures, hospitalisations) • Diabetic complications (e. g. retinopathy, blindness, amputation, neuropathy, albuminuria, end stage renal failure) • Expected events (e. g. infection, conditions associated with diabetes such as hyperlipidemia/dyslipidemia, hypertension, gout) • Components of the primary and secondary endpoints • All neoplasms • Pancreatitis *Adjudication performed by independent Clinical Events Classification (CEC) Committee using prespecified criteria and definitions

Participant Characteristics, Follow-Up and Changes in Key Risk Factors M. Angelyn Bethel, MD Diabetes Trials Unit, University of Oxford

Presenter Disclosure Information Research Support: • Astra Zeneca, Bayer, Merck Serono Advisory Boards: • Boehringer-Ingelheim, Novo Nordisk Other Support: • Astra Zeneca, Merck, Novo Nordisk, Sanofi
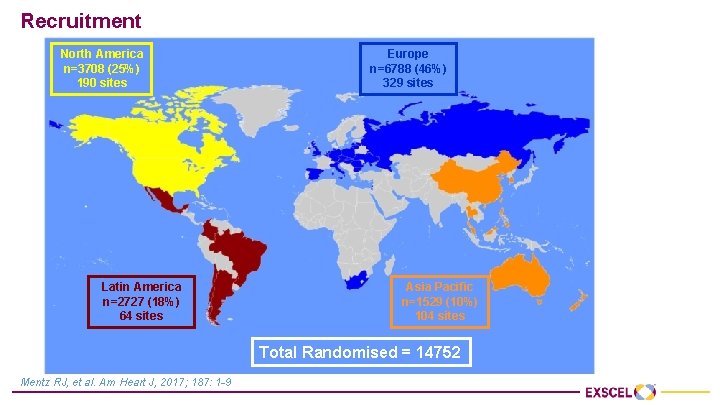
Recruitment North America n=3708 (25%) 190 sites Latin America n=2727 (18%) 64 sites Europe n=6788 (46%) 329 sites Asia Pacific n=1529 (10%) 104 sites Total Randomised = 14752 Mentz RJ, et al. Am Heart J, 2017; 187: 1 -9
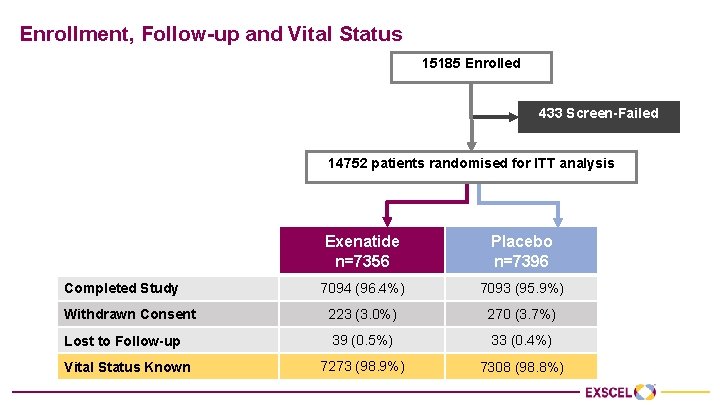
Enrollment, Follow-up and Vital Status 15185 Enrolled 433 Screen-Failed 14752 patients randomised for ITT analysis Exenatide n=7356 Placebo n=7396 7094 (96. 4%) 7093 (95. 9%) Withdrawn Consent 223 (3. 0%) 270 (3. 7%) Lost to Follow-up 39 (0. 5%) 33 (0. 4%) 7273 (98. 9%) 7308 (98. 8%) Completed Study Vital Status Known
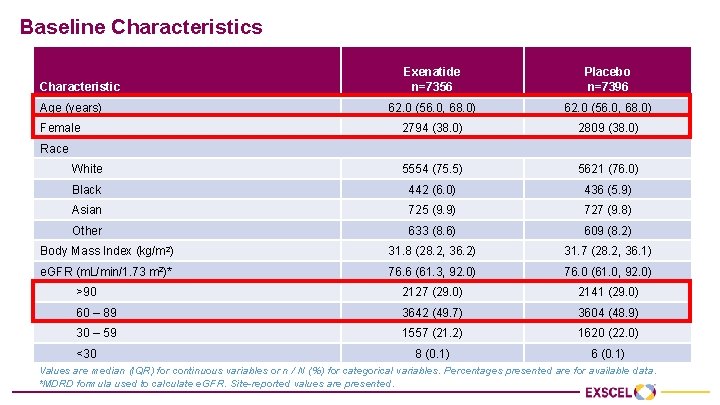
Baseline Characteristics Exenatide n=7356 Placebo n=7396 62. 0 (56. 0, 68. 0) 2794 (38. 0) 2809 (38. 0) White 5554 (75. 5) 5621 (76. 0) Black 442 (6. 0) 436 (5. 9) Asian 725 (9. 9) 727 (9. 8) Other 633 (8. 6) 609 (8. 2) Body Mass Index (kg/m 2) 31. 8 (28. 2, 36. 2) 31. 7 (28. 2, 36. 1) e. GFR (m. L/min/1. 73 m 2)* 76. 6 (61. 3, 92. 0) 76. 0 (61. 0, 92. 0) >90 2127 (29. 0) 2141 (29. 0) 60 – 89 3642 (49. 7) 3604 (48. 9) 30 – 59 1557 (21. 2) 1620 (22. 0) 8 (0. 1) 6 (0. 1) Characteristic Age (years) Female Race <30 Values are median (IQR) for continuous variables or n / N (%) for categorical variables. Percentages presented are for available data. *MDRD formula used to calculate e. GFR. Site-reported values are presented.
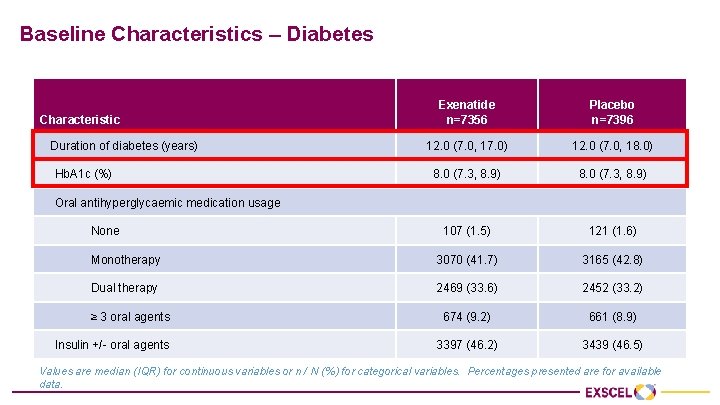
Baseline Characteristics – Diabetes Exenatide n=7356 Placebo n=7396 12. 0 (7. 0, 17. 0) 12. 0 (7. 0, 18. 0) 8. 0 (7. 3, 8. 9) 107 (1. 5) 121 (1. 6) Monotherapy 3070 (41. 7) 3165 (42. 8) Dual therapy 2469 (33. 6) 2452 (33. 2) 674 (9. 2) 661 (8. 9) 3397 (46. 2) 3439 (46. 5) Characteristic Duration of diabetes (years) Hb. A 1 c (%) Oral antihyperglycaemic medication usage None ≥ 3 oral agents Insulin +/- oral agents Values are median (IQR) for continuous variables or n / N (%) for categorical variables. Percentages presented are for available data.
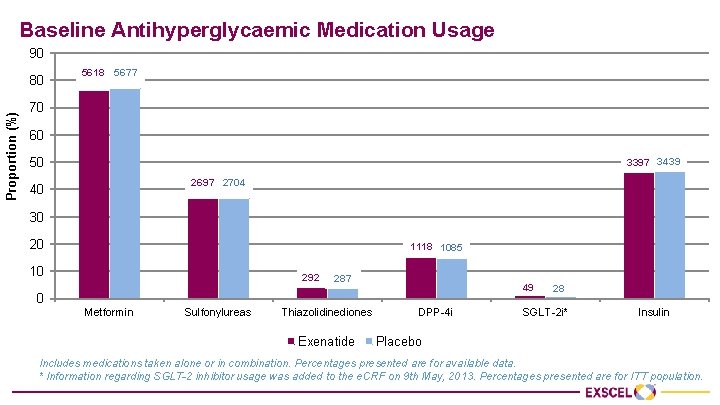
Baseline Antihyperglycaemic Medication Usage 90 Proportion (%) 80 5618 5677 70 60 50 3397 3439 2697 2704 40 30 20 1118 1085 10 0 292 Metformin Sulfonylureas 287 Thiazolidinediones Exenatide 49 DPP-4 i 28 SGLT-2 i* Insulin Placebo Includes medications taken alone or in combination. Percentages presented are for available data. * Information regarding SGLT-2 inhibitor usage was added to the e. CRF on 9 th May, 2013. Percentages presented are for ITT population.
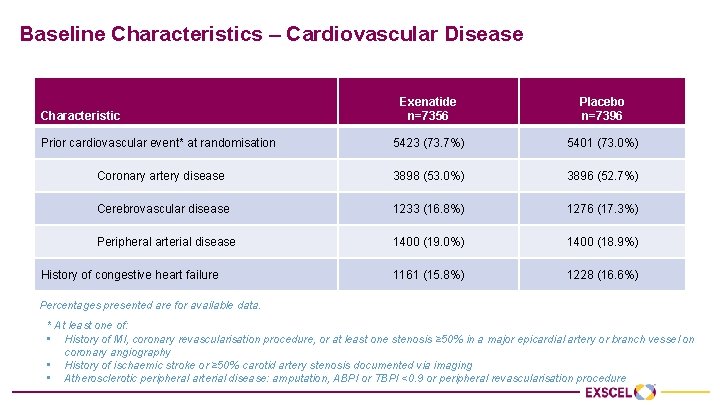
Baseline Characteristics – Cardiovascular Disease Exenatide n=7356 Placebo n=7396 5423 (73. 7%) 5401 (73. 0%) Coronary artery disease 3898 (53. 0%) 3896 (52. 7%) Cerebrovascular disease 1233 (16. 8%) 1276 (17. 3%) Peripheral arterial disease 1400 (19. 0%) 1400 (18. 9%) 1161 (15. 8%) 1228 (16. 6%) Characteristic Prior cardiovascular event* at randomisation History of congestive heart failure Percentages presented are for available data. * At least one of: • History of MI, coronary revascularisation procedure, or at least one stenosis ≥ 50% in a major epicardial artery or branch vessel on coronary angiography • History of ischaemic stroke or ≥ 50% carotid artery stenosis documented via imaging • Atherosclerotic peripheral arterial disease: amputation, ABPI or TBPI <0. 9 or peripheral revascularisation procedure
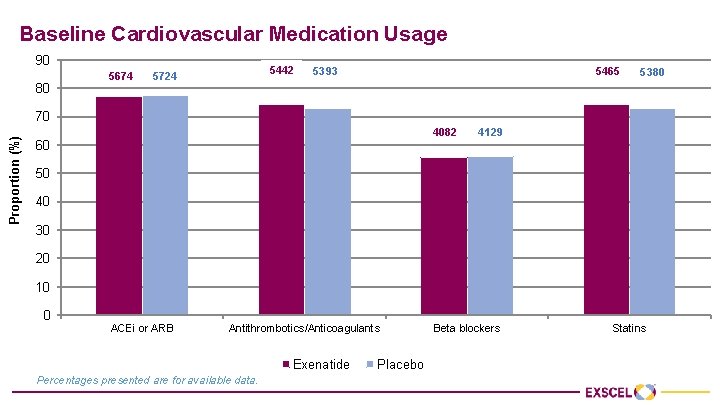
Baseline Cardiovascular Medication Usage 90 80 5674 5442 5724 5393 5465 5380 Proportion (%) 70 4082 60 4129 50 40 30 20 10 0 ACEi or ARB Antithrombotics/Anticoagulants Exenatide Percentages presented are for available data. Placebo Beta blockers Statins
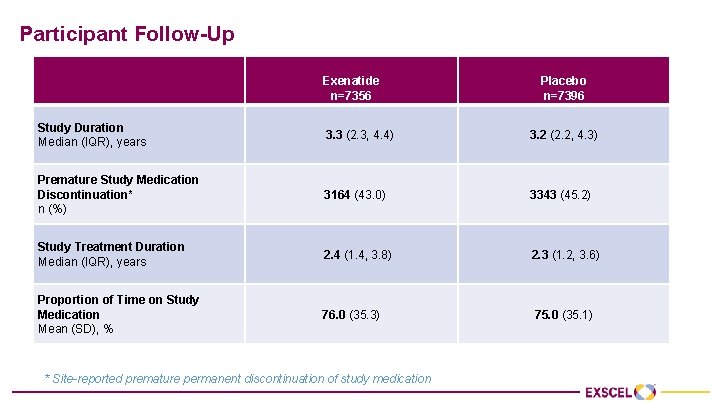
Participant Follow-Up Study Duration Median (IQR), years Premature Study Medication Discontinuation* n (%) Study Treatment Duration Median (IQR), years Proportion of Time on Study Medication Mean (SD), % Exenatide n=7356 Placebo n=7396 3. 3 (2. 3, 4. 4) 3. 2 (2. 2, 4. 3) 3164 (43. 0) 3343 (45. 2) 2. 4 (1. 4, 3. 8) 2. 3 (1. 2, 3. 6) 76. 0 (35. 3) 75. 0 (35. 1) * Site-reported premature permanent discontinuation of study medication

Changes in key risk factors
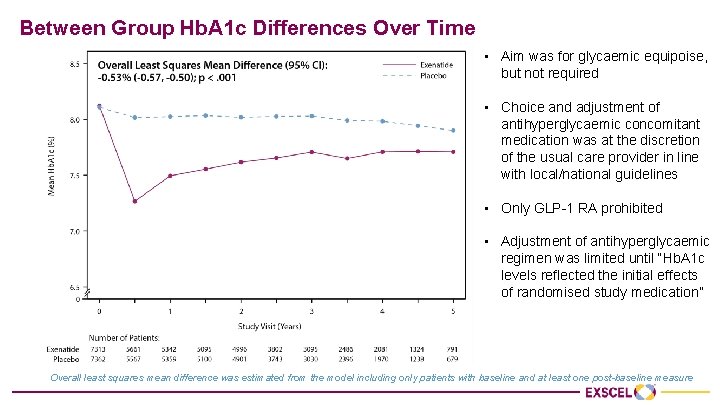
Between Group Hb. A 1 c Differences Over Time • Aim was for glycaemic equipoise, but not required • Choice and adjustment of antihyperglycaemic concomitant medication was at the discretion of the usual care provider in line with local/national guidelines • Only GLP-1 RA prohibited • Adjustment of antihyperglycaemic regimen was limited until “Hb. A 1 c levels reflected the initial effects of randomised study medication” Overall least squares mean difference was estimated from the model including only patients with baseline and at least one post-baseline measure
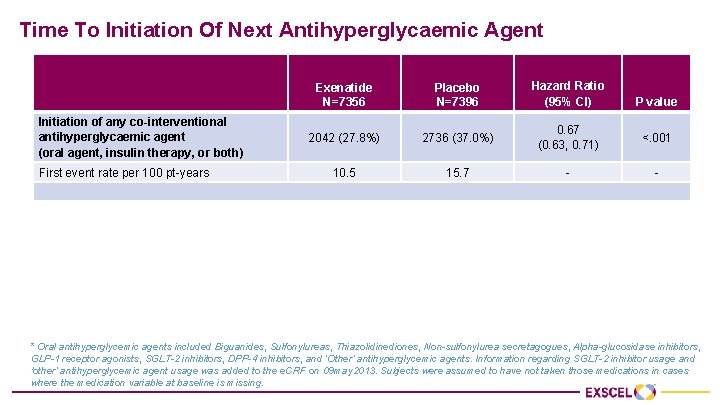
Time To Initiation Of Next Antihyperglycaemic Agent Initiation of any co-interventional antihyperglycaemic agent (oral agent, insulin therapy, or both) First event rate per 100 pt-years Initiation of additional oral antihyperglycaemic agent * First event rate per 100 pt-years Initiation of first chronic insulin therapy First event rate per 100 pt-years Exenatide N=7356 Placebo N=7396 Hazard Ratio (95% CI) 2042 (27. 8%) 2736 (37. 0%) 0. 67 (0. 63, 0. 71) <. 001 10. 5 15. 7 - - 1743 (23. 7%) 2289 (30. 9%) 0. 70 (0. 66, 0. 75) <. 001 8. 6 12. 2 - - 515 (7. 0%) 811 (11. 0%) 0. 61 (0. 54, 0. 68) <. 001 2. 3 3. 8 - - P value * Oral antihyperglycemic agents included Biguanides, Sulfonylureas, Thiazolidinediones, Non-sulfonylurea secretagogues, Alpha-glucosidase inhibitors, GLP-1 receptor agonists, SGLT-2 inhibitors, DPP-4 inhibitors, and ‘Other’ antihyperglycemic agents. Information regarding SGLT-2 inhibitor usage and 'other’ antihyperglycemic agent usage was added to the e. CRF on 09 may 2013. Subjects were assumed to have not taken those medications in cases where the medication variable at baseline is missing.
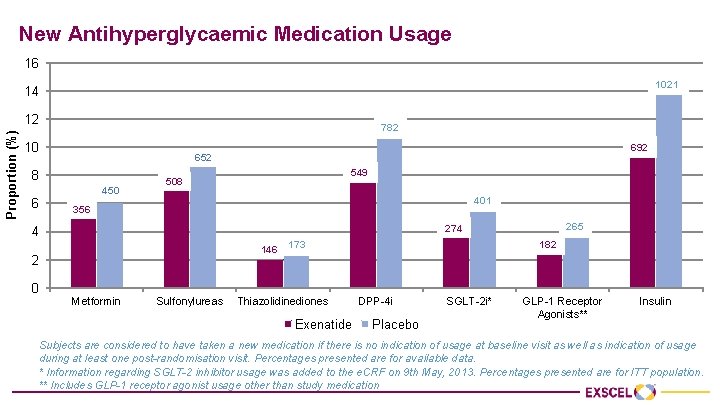
New Antihyperglycaemic Medication Usage 16 1021 14 Proportion (%) 12 782 10 8 6 450 549 508 401 356 265 274 4 182 146 173 2 0 692 652 Metformin Sulfonylureas Thiazolidinediones Exenatide DPP-4 i Placebo SGLT-2 i* GLP-1 Receptor Agonists** Insulin Subjects are considered to have taken a new medication if there is no indication of usage at baseline visit as well as indication of usage during at least one post-randomisation visit. Percentages presented are for available data. * Information regarding SGLT-2 inhibitor usage was added to the e. CRF on 9 th May, 2013. Percentages presented are for ITT population. ** Includes GLP-1 receptor agonist usage other than study medication
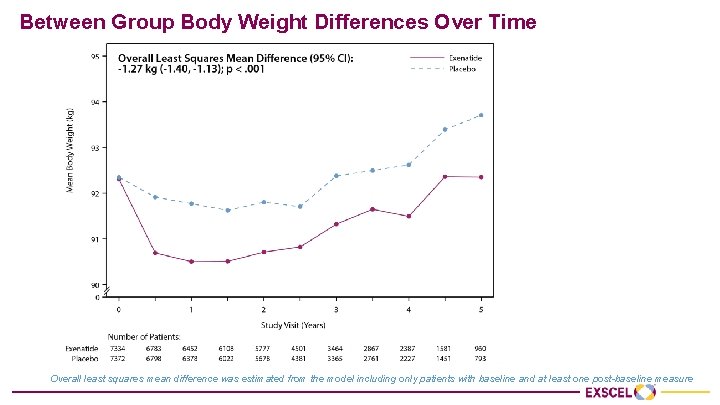
Between Group Body Weight Differences Over Time Overall least squares mean difference was estimated from the model including only patients with baseline and at least one post-baseline measure
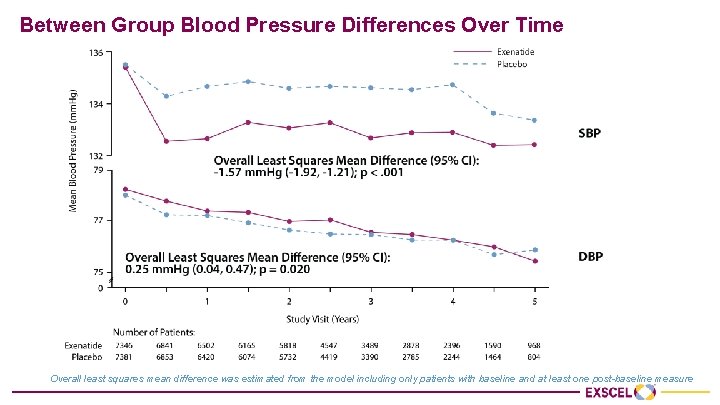
Between Group Blood Pressure Differences Over Time Overall least squares mean difference was estimated from the model including only patients with baseline and at least one post-baseline measure
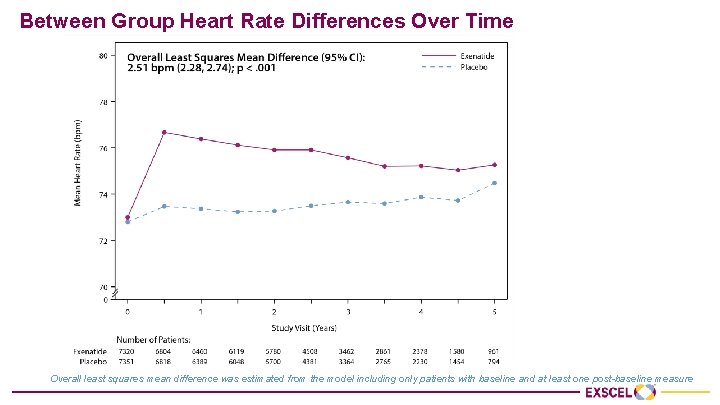
Between Group Heart Rate Differences Over Time Overall least squares mean difference was estimated from the model including only patients with baseline and at least one post-baseline measure

Safety Data Bernard Zinman, MD Lunenfeld Tanenbaum Research Institute, University of Toronto

Presenter Disclosure Information Grant support: • Boehringer-Ingelheim, Astra. Zeneca, Novo Nordisk Consulting fees: • Astra. Zeneca, Boehringer-Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk, Sanofi Aventis

General Principles • All data presented in this section relate to the Safety Population (participants in the Intention-to-Treat population who took at least one dose of study medication) • SAEs were recorded in detail • Non-serious AEs were not generally recorded (only pancreatitis and neoplasms) • Clinical events of interest (exempt from traditional AE/SAE collection and reporting): • CV events (e. g. procedures, hospitalisations) • Diabetic complications (e. g. retinopathy, blindness, amputation, neuropathy, albuminuria, end stage renal failure) • Expected events (e. g. infection, metabolic conditions associated with diabetes such as hyperlipidemia/dyslipidemia, hypertension, and gout)
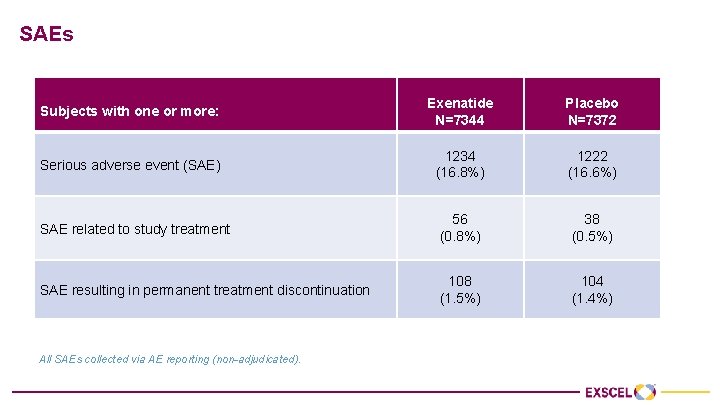
SAEs Subjects with one or more: Exenatide N=7344 Placebo N=7372 Serious adverse event (SAE) 1234 (16. 8%) 1222 (16. 6%) SAE related to study treatment 56 (0. 8%) 38 (0. 5%) SAE resulting in permanent treatment discontinuation 108 (1. 5%) 104 (1. 4%) All SAEs collected via AE reporting (non-adjudicated).
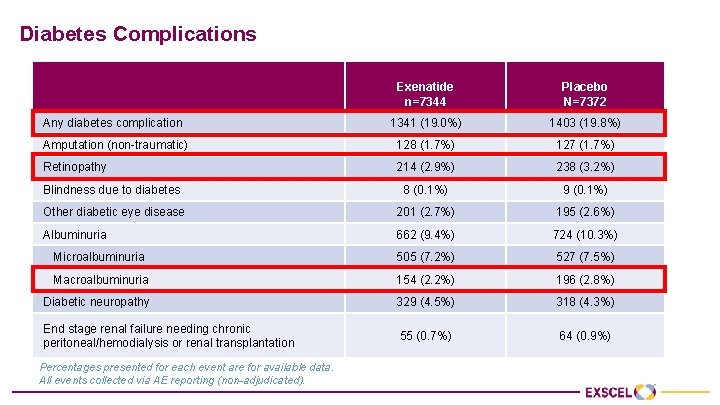
Diabetes Complications Exenatide n=7344 Placebo N=7372 Any diabetes complication 1341 (19. 0%) 1403 (19. 8%) Amputation (non-traumatic) 128 (1. 7%) 127 (1. 7%) 214 (2. 9%) 238 (3. 2%) Blindness due to diabetes 8 (0. 1%) 9 (0. 1%) Other diabetic eye disease 201 (2. 7%) 195 (2. 6%) Albuminuria 662 (9. 4%) 724 (10. 3%) Microalbuminuria 505 (7. 2%) 527 (7. 5%) Macroalbuminuria 154 (2. 2%) 196 (2. 8%) Diabetic neuropathy 329 (4. 5%) 318 (4. 3%) End stage renal failure needing chronic peritoneal/hemodialysis or renal transplantation 55 (0. 7%) 64 (0. 9%) Retinopathy Percentages presented for each event are for available data. All events collected via AE reporting (non-adjudicated).
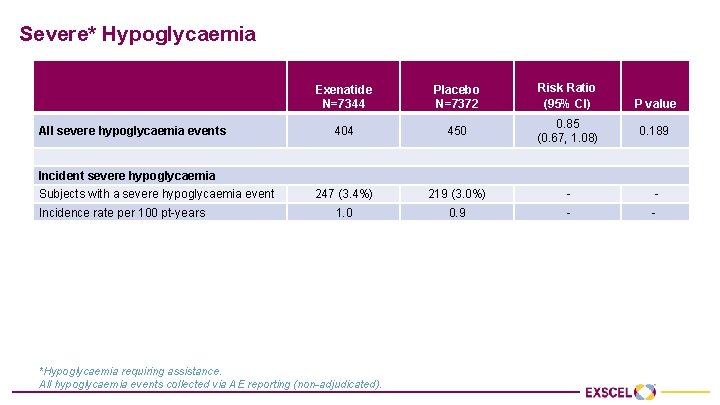
Severe* Hypoglycaemia Exenatide N=7344 Placebo N=7372 Risk Ratio (95% CI) 404 450 0. 85 (0. 67, 1. 08) 247 (3. 4%) 219 (3. 0%) - - 1. 0 0. 9 - - 1 185 148 - - 2 39 40 - - ≥ 3 23 31 - - 1. 6 1. 8 - - All severe hypoglycaemia events P value 0. 189 Incident severe hypoglycaemia Subjects with a severe hypoglycaemia event Incidence rate per 100 pt-years Recurrent severe hypoglycaemia Number of events per subject: Rate of recurrence per 100 pt-years *Hypoglycaemia requiring assistance. All hypoglycaemia events collected via AE reporting (non-adjudicated).
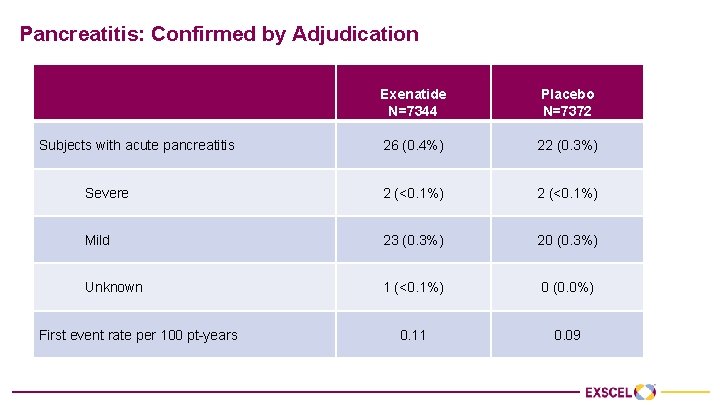
Pancreatitis: Confirmed by Adjudication Exenatide N=7344 Placebo N=7372 26 (0. 4%) 22 (0. 3%) Severe 2 (<0. 1%) Mild 23 (0. 3%) 20 (0. 3%) Unknown 1 (<0. 1%) 0 (0. 0%) 0. 11 0. 09 Subjects with acute pancreatitis First event rate per 100 pt-years
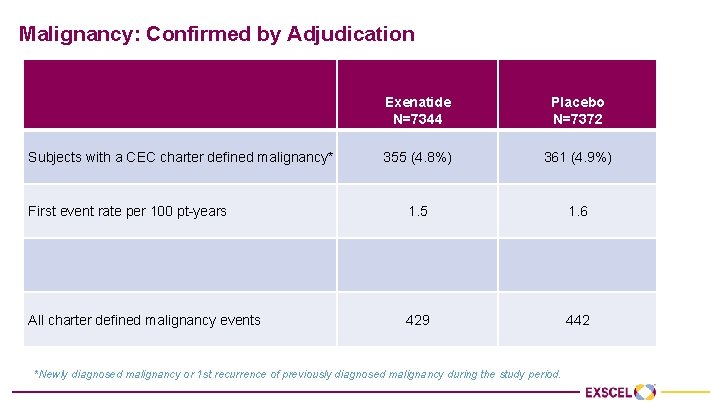
Malignancy: Confirmed by Adjudication Exenatide N=7344 Placebo N=7372 355 (4. 8%) 361 (4. 9%) First event rate per 100 pt-years 1. 5 1. 6 All charter defined malignancy events 429 442 Subjects with a CEC charter defined malignancy* *Newly diagnosed malignancy or 1 st recurrence of previously diagnosed malignancy during the study period.
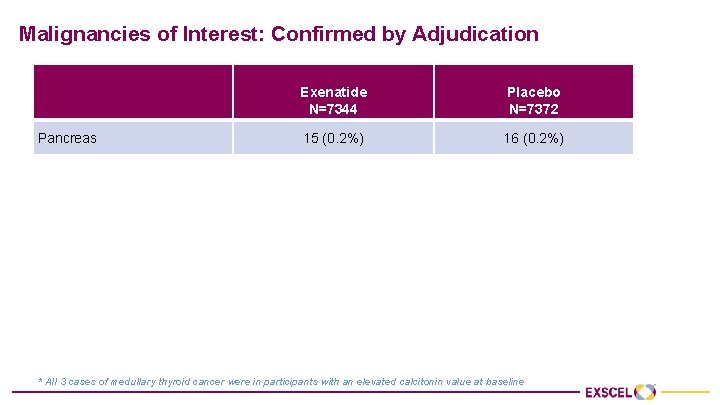
Malignancies of Interest: Confirmed by Adjudication Exenatide N=7344 Placebo N=7372 Pancreas 15 (0. 2%) 16 (0. 2%) Thyroid 14 (0. 2%) 6 (0. 1%) Medullary * 2 1 Papillary 10 4 Follicular 2 0 Other/Unknown 0 1 * All 3 cases of medullary thyroid cancer were in participants with an elevated calcitonin value at baseline
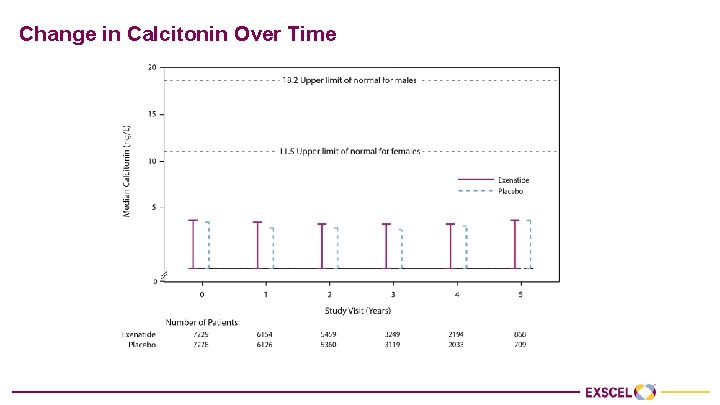
Change in Calcitonin Over Time

Cardiovascular Outcomes Adrian F. Hernandez, MD Duke Clinical Research Institute

Presenter Disclosure Information Research funding: • Astra. Zeneca, Glaxo. Smith. Kline, Merck Consulting fees: • Amgen, Astra. Zeneca, Bayer, Boehringer-Ingelheim, Merck, Sanofi Aventis

Primary Endpoint

Primary Endpoint Time to first occurrence of 3 -point MACE composed of: • CV death • Non-fatal MI • Non-fatal stroke Safety hypothesis (non-inferiority) and efficacy hypothesis (superiority)
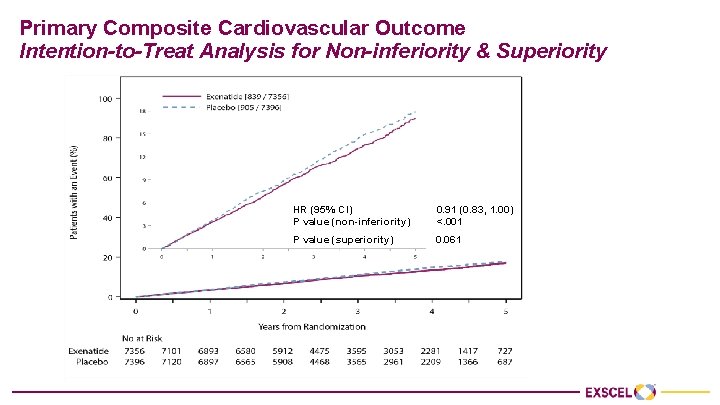
Primary Composite Cardiovascular Outcome Intention-to-Treat Analysis for Non-inferiority & Superiority HR (95% CI) 0. 91 (0. 83, 1. 00) P value (non-inferiority) <. 001 P value (superiority) 0. 061
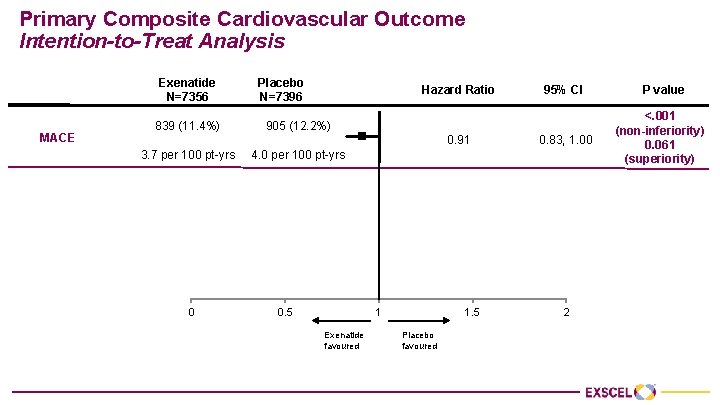
Primary Composite Cardiovascular Outcome Intention-to-Treat Analysis Exenatide N=7356 MACE Placebo N=7396 Hazard Ratio 839 (11. 4%) 905 (12. 2%) 3. 7 per 100 pt-yrs 4. 0 per 100 pt-yrs 95% CI 0. 91 0. 83, 1. 00 0. 88 0. 73, 1. 05 CV-death 229 (3. 1%) 258 (3. 5%) Non-fatal MI 455 (6. 2%) 470 (6. 4%) 0. 95 0. 84, 1. 09 155 (2. 1%) 177 (2. 4%) 0. 86 0. 70, 1. 07 Non-fatal stroke 0 0. 5 1 Exenatide favoured 1. 5 Placebo favoured 2 P value <. 001 (non-inferiority) 0. 061 (superiority) 0. 628 (homogeneity among components)
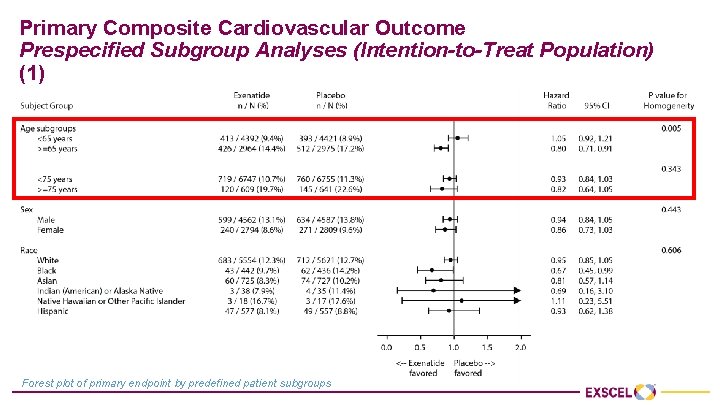
Primary Composite Cardiovascular Outcome Prespecified Subgroup Analyses (Intention-to-Treat Population) (1) Forest plot of primary endpoint by predefined patient subgroups
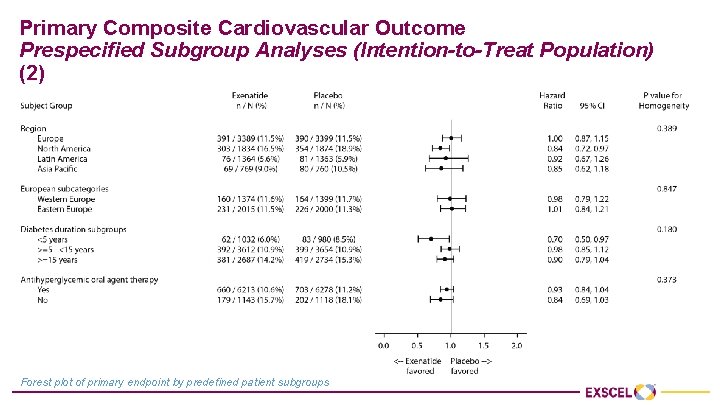
Primary Composite Cardiovascular Outcome Prespecified Subgroup Analyses (Intention-to-Treat Population) (2) Forest plot of primary endpoint by predefined patient subgroups
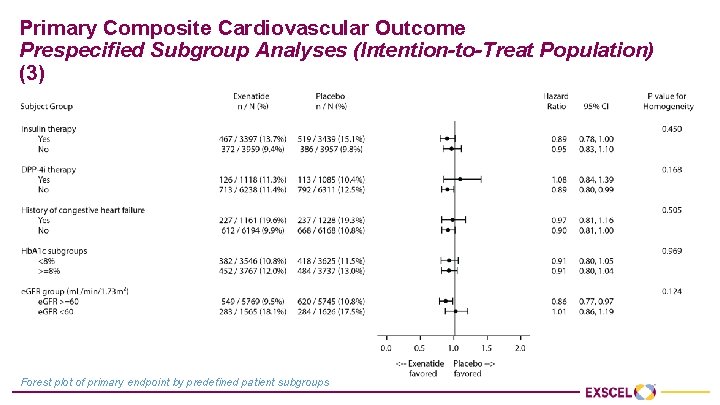
Primary Composite Cardiovascular Outcome Prespecified Subgroup Analyses (Intention-to-Treat Population) (3) Forest plot of primary endpoint by predefined patient subgroups
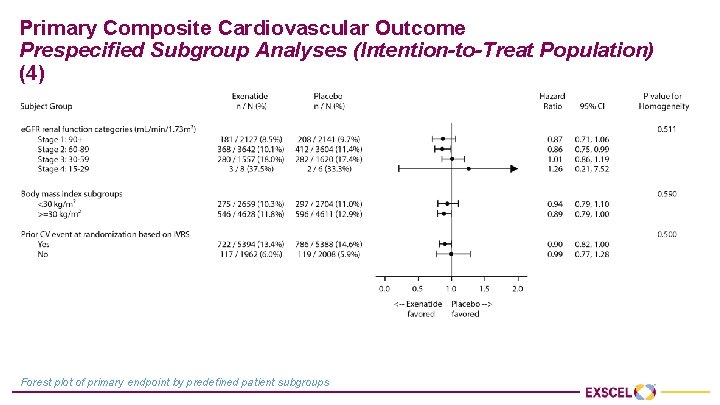
Primary Composite Cardiovascular Outcome Prespecified Subgroup Analyses (Intention-to-Treat Population) (4) Forest plot of primary endpoint by predefined patient subgroups

Secondary Endpoints

Secondary Endpoints Time to first occurrence of: 1. All-cause mortality 2. CV-related death 3. Fatal or non-fatal MI 4. Fatal or non-fatal stroke 5. Hospitalisation for acute coronary syndrome (ACS) 6. Hospitalisation for heart failure (HF)
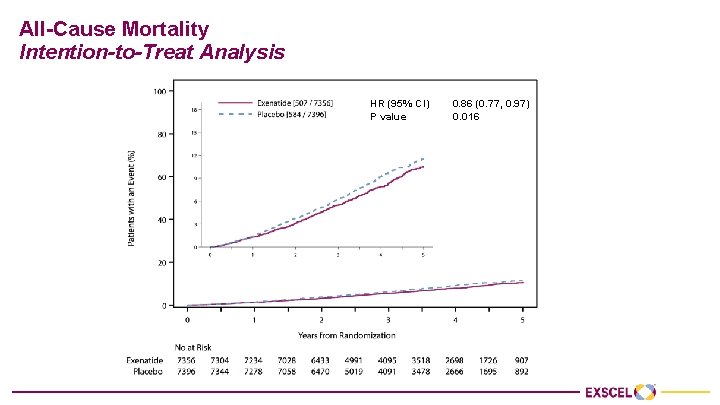
All-Cause Mortality Intention-to-Treat Analysis HR (95% CI) 0. 86 (0. 77, 0. 97) P value 0. 016
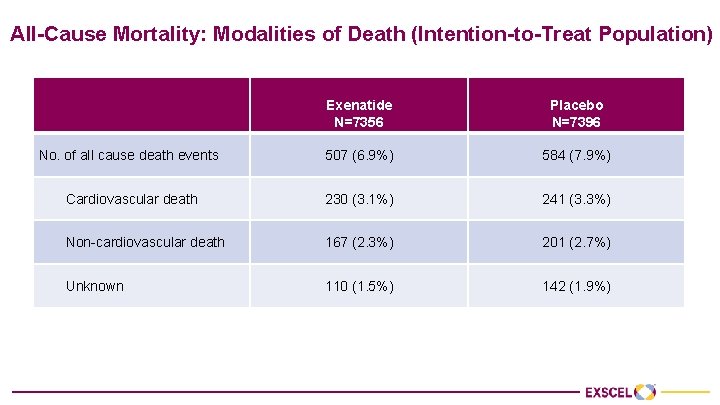
All-Cause Mortality: Modalities of Death (Intention-to-Treat Population) Exenatide N=7356 Placebo N=7396 No. of all cause death events 507 (6. 9%) 584 (7. 9%) Cardiovascular death 230 (3. 1%) 241 (3. 3%) Non-cardiovascular death 167 (2. 3%) 201 (2. 7%) Unknown 110 (1. 5%) 142 (1. 9%)
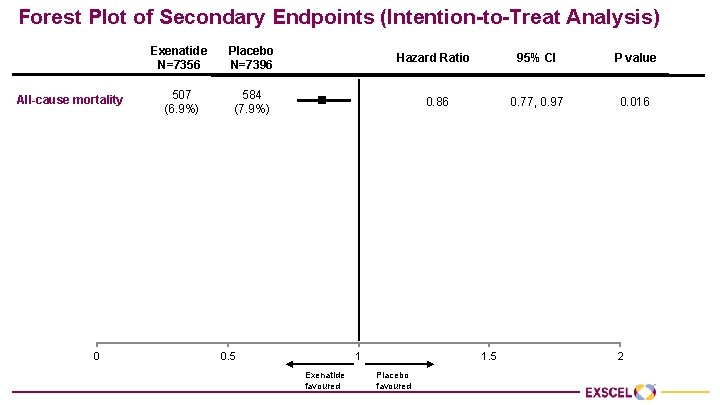
Forest Plot of Secondary Endpoints (Intention-to-Treat Analysis) Exenatide N=7356 Placebo N=7396 All-cause mortality 507 (6. 9%) 584 (7. 9%) CV-death 340 (4. 6%) Fatal or non-fatal MI Fatal or non-fatal stroke 95% CI P value 0. 86 0. 77, 0. 97 0. 016 383 (5. 2%) 0. 88 0. 76, 1. 02 0. 096 483 (6. 6%) 493 (6. 7%) 0. 97 0. 85, 1. 10 0. 622 187 (2. 5%) 218 (2. 9%) 0. 85 0. 70, 1. 03 0. 095 Hospitalisation for ACS 602 (8. 2%) 570 (7. 7%) 1. 05 0. 94, 1. 18 0. 402 Hospitalisation for heart failure 219 (3. 0%) 231 (3. 1%) 0. 94 0. 78, 1. 13 0. 485 0 Hazard Ratio 0. 5 1 Exenatide favoured 1. 5 Placebo favoured 2
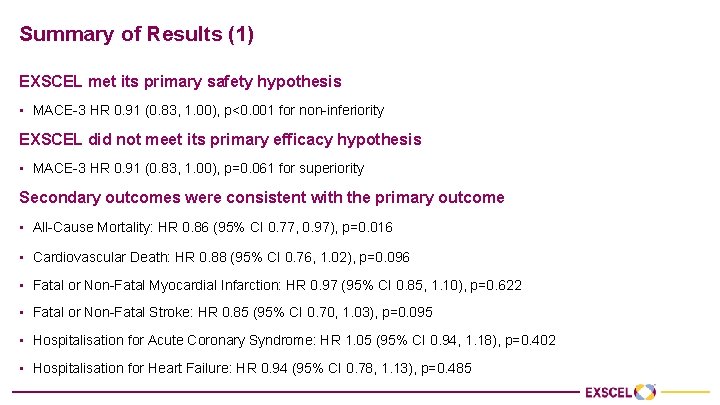
Summary of Results (1) EXSCEL met its primary safety hypothesis • MACE-3 HR 0. 91 (0. 83, 1. 00), p<0. 001 for non-inferiority EXSCEL did not meet its primary efficacy hypothesis • MACE-3 HR 0. 91 (0. 83, 1. 00), p=0. 061 for superiority Secondary outcomes were consistent with the primary outcome • All-Cause Mortality: HR 0. 86 (95% CI 0. 77, 0. 97), p=0. 016 • Cardiovascular Death: HR 0. 88 (95% CI 0. 76, 1. 02), p=0. 096 • Fatal or Non-Fatal Myocardial Infarction: HR 0. 97 (95% CI 0. 85, 1. 10), p=0. 622 • Fatal or Non-Fatal Stroke: HR 0. 85 (95% CI 0. 70, 1. 03), p=0. 095 • Hospitalisation for Acute Coronary Syndrome: HR 1. 05 (95% CI 0. 94, 1. 18), p=0. 402 • Hospitalisation for Heart Failure: HR 0. 94 (95% CI 0. 78, 1. 13), p=0. 485

Summary of Results (2) Safety Events Similar rates of: • Pancreatitis • Charter defined malignancy • Specifically, no differences in pancreatic or medullary thyroid cancer • Severe hypoglycaemia

Acknowledgements and Thanks

EXSCEL: Governance Executive Committee Responsible for the overall scientific, professional and operational conduct of the trial • • • Rury Holman (Co-chair, UK) Adrian Hernandez (Co-chair, US) John Buse (US) Juliana Chan (Hong Kong) Aldo Maggioni (Italy) Steven Marso (US) Neil Poulter (UK) Ambady Ramachandran (India) Bernard Zinman (Canada) Nayyar Iqbal (US) Peter Ohman (US) Academic Members Sponsor Members *Former co-chair: Robert M. Califf Data Safety Monitoring Board Responsible for safeguarding the interests of trial participants, monitoring the main outcome measures and overall trial conduct Clinical Events Classification (CEC) Committee Responsible for the conduct of CEC operations, including independent adjudication of pre-specified outcomes Jean Rouleau (Chair, Canada) Primary Investigator: Jennifer Green (DCRI) Stuart Pocock (UK) Fred Gorelick (US) John Mc. Murray (UK) Matt Riddle (US) Robert Gagel (US) Tim Collier (Independent Statistician, UK) Strategic Advisory Committee (SAC) Operations Committee Responsible for providing strategic oversight of the operational conduct of the study, across all participating institutions Responsible for facilitating trial progress at the regional level, serving as the interface between the Executive Committee and study sites Karen Hannan (Co-chair, DCRI) Angelyn Bethel (Co-chair, DTU)

EXSCEL: Operations Committee Argentina: Rafael Diaz Lithuania: Neli Jakuboniene Australia: Tania Markovic Malaysia: Sim Kui Hian Former members: Austria: Guntram Schernthaner Mexico: Jose Luis Leiva-Pons Australia: Ian Caterson Netherlands: Adriaan Kooy China: Jianping Weng, Dayi Hu, Ge Junbo New Zealand: Russell Scott France: Faiez Zannad Belgium: Chantal Mathieu Brazil: Renato Lopes Bulgaria: Tsvetalina Tankova Canada: Shaun Goodman, Jean Francois Yale Chile: Fernando Lanas China: Ji Linong, Huo Yong, Chanyu Pan Colombia: Miguel Urina Philippines: Araceli Panelo Poland: Piotr Dziemidok India: Misra Anoop, Mithal Ambrish Peru: John Adaly Gallegos Romania: Ioan Andrei Veresiu Russia: Alexander V. Dreval Slovakia: Jan Murin South Africa: Mohamed Omar Czech Republic: Petr Widimsky South Korea: Kun-Ho Yoon Germany: Markolf Hanefeld Spain: Albert Le. Cube Torello Hong Kong: Alice Kong Taiwan: Wayne Sheu Hungary: Matyas Keltai Thailand: Piyamitr Sritara Israel: Julio Wainstein Ukraine: Alexander Parkhomenko Italy: Stefano del Prato United Kingdom: Naveed Sattar Latvia: Valdis Pirags United States: David Aguliera, Richard Bergenstal

Participants and Sites • We thank the participants, without whom this study and these analyses would not have been possible • We also thank the many investigators from 687 sites in 35 countries who worked diligently to help ensure EXSCEL was run to the highest possible standards

Partners We thank the following academic partners and contract research organisations for their assistance: • Canadian VIGOUR Centre • PAREXEL International

Results online at www. nejm. org DOI: 10. 1056/NEJMoa 1612917

Slides available at: www. dtu. ox. ac. uk/EXSCEL
- Slides: 67