Expressions of the equilibrium constant K The equilibrium

Expressions of the equilibrium constant K • The equilibrium constant, K, (a dimensionless quantity) can be expressed in terms of fugacities for gas phase reactions or activities for aqueous phase reactions. • Fugacity ( a dimensionless quantity) is equal to the numerical value of partial pressure, i. e. pj/pθ where pθ = 1 bar). • The activity, a, is equal to the numerical value of the molality, i. e. bj/bθ where bθ = 1 mol kg-1.

For reactions occurring in electrolyte solutions • Effects of the interactions of ions on the reaction process should be considered. • Such a factor can be expressed with the activity coefficient, γ, which denotes distance from the ideal system where there is no ion-interaction. • The activity shall now be calculated as αj = γj*bj/bθ • For a reaction A + B ↔ C + D K =

The activities of solids are equal to 1 • α(solid) = 1 (!!!) • Illustration: Express the equilibrium constant for the heterogeneous reaction NH 4 Cl(s) ↔ NH 3(g) + HCl(g) • Solution: In term of fugacity (i. e. partial pressure): Kp = In term of molar fraction: Kx =

Estimate reaction compositions at equilibrium • Example 1: Given the standard Gibbs energy of reaction H 2 O(g) → H 2(g) + 1/2 O 2(g) at 2000 K is + 135. 2 k. J mol-1, suppose that steam at 200 k pa is passed through a furnace tube at that temperature. Calculate the mole fraction of O 2 present in the output gas stream. • Solution: (details will be discussed in class) ln. K = - (135. 2 x 103 J mol-1)/(8. 3145 JK-1 mol-1 x 2000 K) = - 8. 13037 K = 2. 9446 x 10 -4 K= Ptotal = 200 Kpa assuming the mole fraction of O 2 equals x PO 2 = x* Ptotal, PH 2 = 2(x*Ptotal) PH 2 O = Ptotal – PO 2 – PH 2 = (1 -3 x)Ptotal

Equilibria in biological systems • Biological standard state: p. H = 7. • For a reaction: A + v. H+(aq) ↔ P Δr. G = Δr. Gθ + RT the first two terms of the above eq. form Δr. G‡ = Δr. Gθ + 7 v. RTln 10

Example: For a particular reaction of the form A → B + 2 H+ in aqueous solution, it was found that Δr. Gθ = 20 k. J mol-1 at 28 o. C. Estimate the value of Δr. G‡. • Solution: Δr. G‡ = Δr. Gθ + 7 v. RTln 10 here v = - 2 !!! Δr. G‡ = 20 k. J mol-1 + 7(-2)(8. 3145 x 10 -3 k. J K-1 mol-1) x(273+ 28 K)ln 10 = 20 k. J mol-1 – 80. 676 k. J mol-1 = -61 k. J mol-1 (Note that when measured with the biological standard, the standard reaction Gibbs energy becomes negative!)
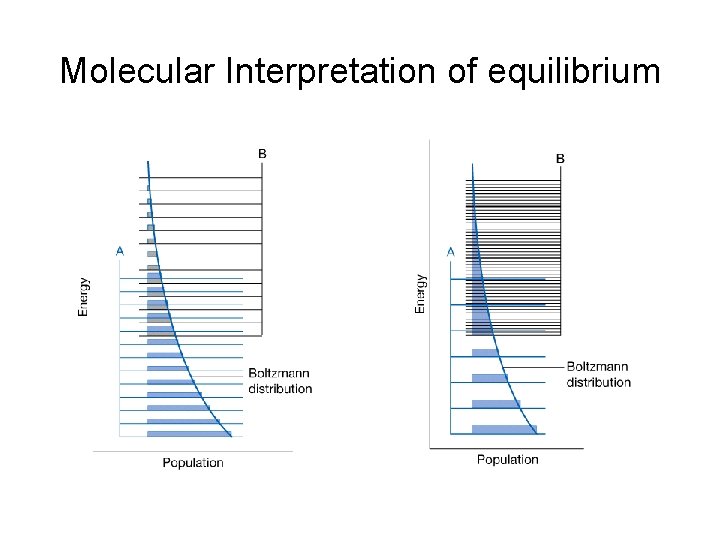
Molecular Interpretation of equilibrium

The response of equilibria to reaction conditions • Equilibria respond to changes in pressure, temperature, and concentrations of reactants and products. • The equilibrium constant is not affected by the presence of a catalyst.

How equilibria respond to pressure • Equilibrium constant K is a function of the standard reaction Gibbs energy, Δr. Gθ. • Standard reaction Gibbs energy Δr. Gθ is defined at a single standard pressure and thus is independent of pressure. • The equilibrium constant is therefore independent of pressure:

• K is independent of pressure does NOT mean that the equilibrium composition is independent of the pressure!!! consider the reaction 2 A(g) ↔ B(g) assuming that the mole fraction of A equals x. A at quilibrium, then x. B = 1. 0 – x. A, K= because K does not change, x. A must change in response to any variation in Ptotal!!!

Le Chatelier’s Principle • A system at equilibrium, when subject to a disturbance, responds in a way that tends to minimize the effect of the disturbance.

Example: Predict the effect of an increase in pressure on the Haber reaction, 3 H 2(g) + N 2(g) ↔ 2 NH 3(g). • Solution: According to Le Chatelier’s Principle, an increase in pressure will favor the product. prove: K= Therefore, to keep K unchanged, the equilibrium mole fractions Kx will change by a factor of 4 if doubling the pressure ptotal.

The response of equilibria to temperature • According to Le Chatelier’s Principle: Exothermic reactions: increased temperature favors the reactants. Endothermic reactions: increased temperature favors the products. • The van’t Hoff equation: (a) (7. 23 a) (b) (7. 23 b)

Derivation of the van’t Hoff equation: • Differentiate ln. K with respect to temperature • Using Gibbs-Helmholtz equation (eqn 3. 53 8 th edition) thus • Because d(1/T)/d. T = -1/T 2:
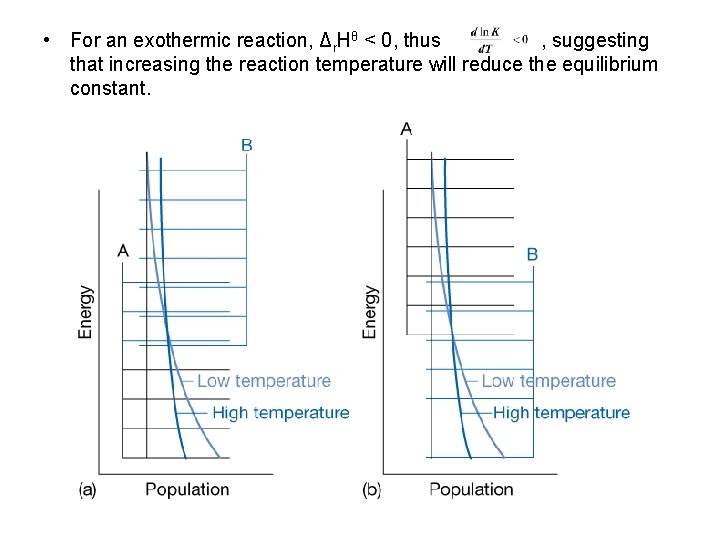
• For an exothermic reaction, Δr. Hθ < 0, thus , suggesting that increasing the reaction temperature will reduce the equilibrium constant.

Applications of the van’t Hoff equation • Provided the reaction enthalpy, Δr. Hθ, can be assumed to be independent of temperature, eqn. 7. 23 b (or 9. 26 b in 7 th edition) illustrates that a plot of –ln. K against 1/T should yield a straight line of slope Δr. Hθ/R. • Example: The date below show the equilibrium constant measured at different temperatures. Calculate the standard reaction enthalpy for the system. T/K 350 400 450 500 K 3. 94 x 10 -4 1. 41 x 10 -2 1. 86 x 10 -1 1. 48 Solution: 1/T -ln. K 2. 86 x 10 -3 7. 83 2. 50 x 10 -3 4. 26 2. 22 x 10 -3 1. 68 2. 00 x 10 -3 -0. 39
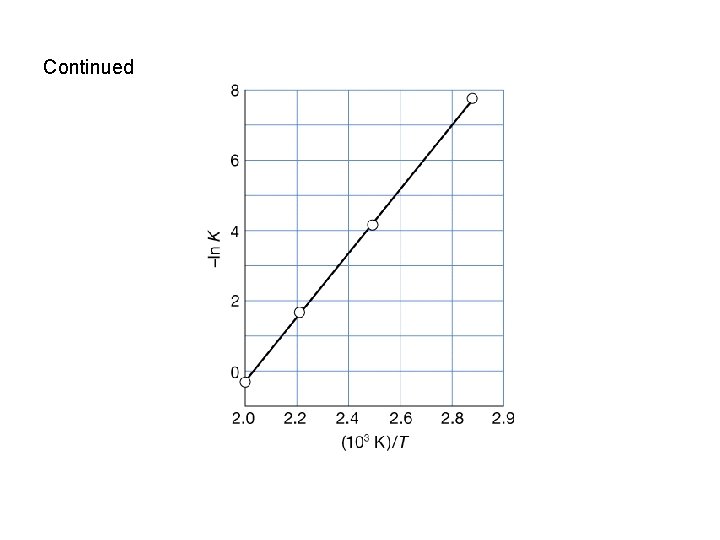
Continued

• Example: The equilibrium constant of the reaction 2 SO 2(g) + O 2(g) ↔ 2 SO 3(g) is 4. 0 x 1024 at 300 K, 2. 5 x 1010 at 500 K, and 2. 0 x 104 at 700 K. Estimate the reaction enthalpy at 500 K. Solution: discussion: 1. Do we need a balanced reaction equation here? 2. What can be learned about the reaction based on the information provided? 3. Will the enthalpy become different at 300 K or 700 K?

Calculate the value of K at different temperatures • The equilibrium constant at a temperature T 2 can be obtained in terms of the know equilibrium constant K 1 at T 1. • Since the standard reaction enthalpy is also a function of temperature, when integrating the equation 9. 26 b from T 1 to T 2, we should assume that Δr. Hө is constant within that interval. • so ln(K 2) – ln(K 1) = (9. 28)

Example, The Haber reaction N 2(g) + 3 H 2(g) ↔ 2 NH 3(g) At 298 K, the equilibrium constant K = 6. 1 x 105. The standard enthalpy of formation for NH 3 equals -46. 1 k. J mol-1. What is the equilibrium constant at 500 K? Answer: First, calculate the standard reaction enthalpy, Δr. Hө = 2*Δf. Hө(NH 3) - 3* Δf. Hө(H 2) - Δf. Hө(N 2) = 2*(-46. 1) – 3*0 - 1*0 = - 92. 2 k. J mol-1 then ln(K 2) – ln(6. 1*105) = ln(K 2) = -1. 71 K 2 = 0. 18 *(-92. 2*1000 J mol-1) (1/500 – 1/298)
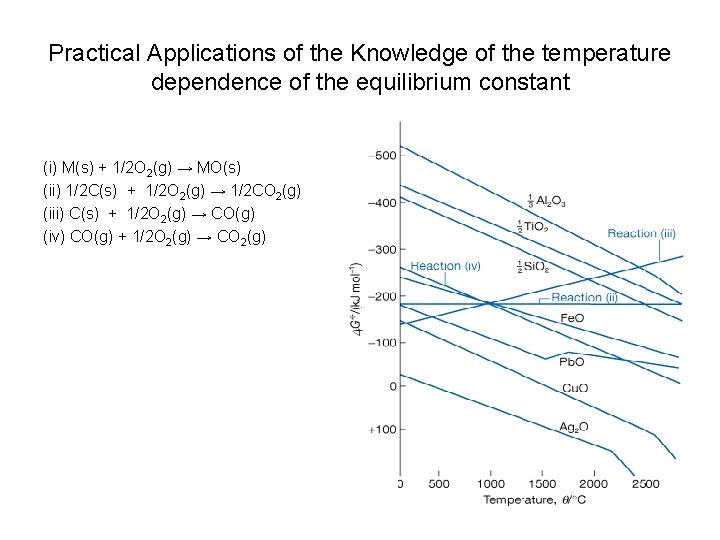
Practical Applications of the Knowledge of the temperature dependence of the equilibrium constant (i) M(s) + 1/2 O 2(g) → MO(s) (ii) 1/2 C(s) + 1/2 O 2(g) → 1/2 CO 2(g) (iii) C(s) + 1/2 O 2(g) → CO(g) (iv) CO(g) + 1/2 O 2(g) → CO 2(g)
- Slides: 21