Expressing Measurements Scientific notation A number is written

Expressing Measurements �Scientific notation �A number is written as the product of two numbers � A coefficient � 10 raised to a power Example: Put IN Scientific Notation A. 602, 000 B. 0. 00774560 6. 02 x 108 7. 74560 x 10 -3 C. 2. 753 x 100

Expressing Measurements Example: Put OUT of Scientific Notation A. 3. 51 x 104 B. 5. 001 x 10 -5 35, 100 . 00005001 Example: Put in CORRECT Scientific Notation A. 237. 1 x 103 B. 43578. 1 x 10 -5 2. 317 x 105 4. 35781 x 10 -1

Scientific Measurement �Measurement depends on 1. Accuracy (“Correctness”) A measure of how close a measurement comes to the actual or true value of whatever is measured 2. Precision (“Reproducibility”) A measure of how close a series of measurements are to one another Examples:

Scientific Measurement �Measurement is never certain because measurement instruments are never free of flaws. �So we only count numbers that are SIGNIFICANT ***All certain numbers plus one uncertain number***

Rules for Counting SIG FIGS NON ZERO INTERGERS: are ALWAYS significant Examples: 352. 1 m. L = _____ sig figs 62 in. = _____ sig figs 43, 978. 6821 g = _____ sig figs

Rules for Counting SIG FIGS 2. ZEROS (There are three classes) LEADING ZEROS are NEVER significant Examples: . 034 g = _____ sig figs 0. 0006178 m = _____ sig figs

Rules for Counting SIG FIGS B. CAPTIVE ZEROS are ALWAYS significant Examples: 205 mi = _____ sig figs 10, 005 g = _____ sig figs

Rules for Counting SIG FIGS C. TRAILING ZEROS are SOMETIMES significant. • Only when there is a decimal point* Examples: 16, 000 mi = _____ sig figs 16, 000. 0 mi = _____ sig figs 2. 3500 m = _____ sig figs

Rules for Counting SIG FIGS 3. EXACT NUMBERS are INFINATELY significant Examples: Counting students Conversions

OPERATIONS & SIG FIGS “The result of calculations involving measurements can only be as precise as the least precise measurement”

OPERATIONS & SIG FIGS 1. MULTIPLICATION AND DIVISION - The product (x) or quotient (/) contain the same number of sig figs as the measurement with the least number of sig figs. Example: 24 cm x 31. 8 cm = 763. 2 cm 2 760 cm 2

OPERATIONS & SIG FIGS 2. ADDITION AND SUBTRACTION - The sum or difference has the same number of decimal places as the number with the least decimal places. Examples: 7. 52 cm + 8. 7 cm = 16. 22 cm 16. 2 cm 39 m + 0. 7893 m = 39. 7893 m 40. m

Metric System �History – people did not have measuring devices readily available �English (customary units) � Foot = length of king’s foot at the time � Inch = length between 1 st & 2 nd knuckles � Pound (lb) = “Liberty” or “Justice” used to measure grains �Metric � Meter – originally 1/10, 000 of distance from North Pole to equator � Gram – weight of 1 cm 3 block and 1 cc of H 2 O in syringe � Liter = 1, 000 cm 3

SI Units �Treaty in 1875 to come up with standard system �International System of Measurements (SI) � French: Le Système international d'unités �Basic SI Units �Length = meter (m) �Mass = kilogram (kg); gram is too small to use as basic unit �Time = seconds (s) �Electric current = ampere (amp) �Temperature = Kelvin (K); never use °K; based on absolute zero �Amount of a substance = mole (mol) �Luminous intensity = candela �All other units are derived from these 7 basic units

�Area = l × w = m × m �Denisty = mass/volume = kg/l × w × h (m × m) �psi = lb/in 2 kg/m

Metric Prefixes �Basic Units �Mass = gram (g) �Length = meter (m) �Volume = liter (L) There are three major parts to the metric system: 1. the seven base units (example: meters) 2. the prefixes (example: kilometer) 3. units built up from the base units. (example: density kg/m 3)
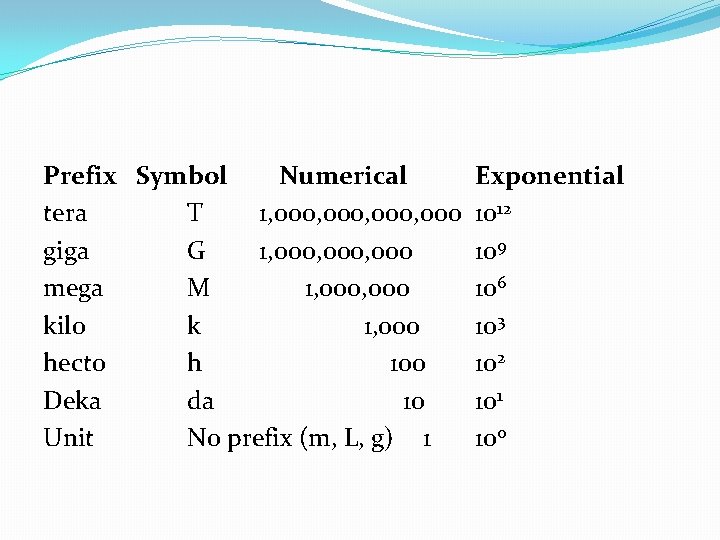
Prefix Symbol Numerical tera T 1, 000, 000 giga G 1, 000, 000 mega M 1, 000 kilo k 1, 000 hecto h 100 Deka da 10 Unit No prefix (m, L, g) 1 Exponential 1012 109 106 103 102 101 100
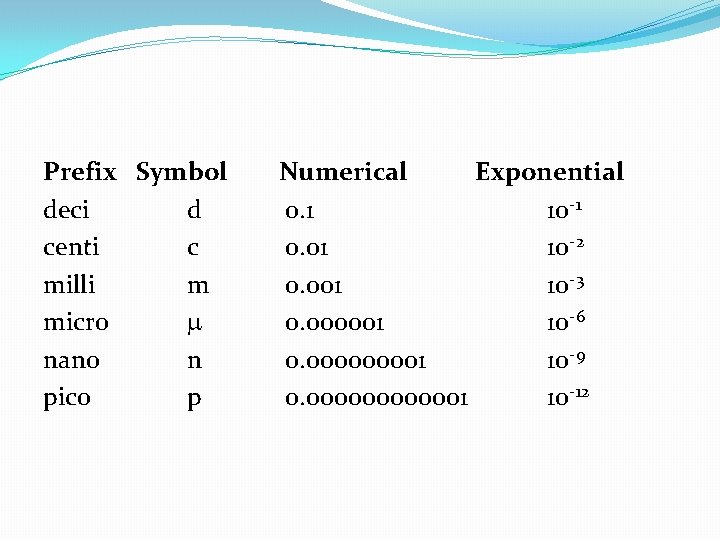
Prefix Symbol deci d centi c milli m micro nano n pico p Numerical Exponential 0. 1 10 -1 0. 01 10 -2 0. 001 10 -3 0. 000001 10 -6 0. 00001 10 -9 0. 0000001 10 -12

Putting it All Together � 1 kilometer = 1 x 103 m = 1000 m � 1 picometer = 1 x 10 -12 m = 0. 0000001 m � 1 milligram = 1 x 10 -3 g = 0. 001 g

�Note: There are units between these numbers (i. e. , 104, 10 -5, etc. ) but they don’t have a prefix, so we don’t discuss them �Need to know all the units, especially �k h d u d c m �“King Henry drinks up delicious chocolate milk. ”

Metric Conversions • memorize the metric prefixes names and symbols. • determine which of two prefixes represents a larger amount. • determine the exponential "distance" between two prefixes.

Practice! � 22. 6 mm = _______ m �Answer: 0. 0226 m (already in correct # of sig figs) �. 61 gh = _____ cg �Answer: 6100 cg � 78. 5 m. L = _____ L �Answer: 0. 0785 L

Memorize These!!! � 1 L = 1 dm 3 � dm 3 used for solid volume; L or m. L used for liquid volume � 1 m. L = 1 cm 3 � 1 m. L of H 2 O = 1 g = 1 cm 3 (a. k. a. , “cc” in hospitals)

These are Tricky Ones �Whenever you see squares or cubes, slow down and handle the problem differently � 12. 0 cm 2 = _____ mm 2 �Answer: 1200 mm 2 � 21 m. L = _____ cm 3 �Answer: 21 cm 3

Just for Fun � 5. 82 mm = ______ Tm � 35. 2 dm 2 = _____ hm 2 � 2. 79 L = _____dm 3 � 45 km/min = _____ m/s �Hardest one I can give you (no setup = no credit): � 3 weeks = _____ s
- Slides: 25