Exposure Control Strategy Return On Investment ROI Rob

Exposure Control Strategy & Return On Investment (ROI) Rob Mc Cafferty May 18, 2011

Watson At-A-Glance Stock Ticker: NYSE: WPI Founded : 1984 Revenues (2010): Approximately $3. 6 billion Employees : Approximately 6, 000 Mission: To improve the quality of life for patients around the world through the development and distribution of trusted generics and advanced, specialty branded pharmaceuticals. Business Divisions: Global Generics, Global Brands, Anda Distribution, CRO, API and Eden Biodesign Distinctions: 3 rd largest generic manufacturer in the US 5 th largest generic manufacturer globally
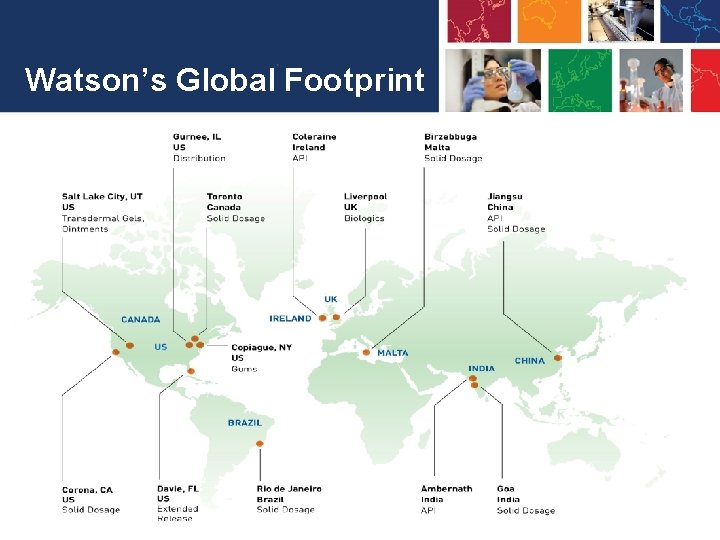
Watson’s Global Footprint

Acknowledgements • Watson Pharmaceuticals – Ana Prieto • Teva – Erella Dimentstein • Barr Laboratories – Mark Ostapczuk • Mercer ORC Networks – Tom Cecich • Sikorsky – Dave Ehrets

Topics • Exposure Control Strategy – – • Overview Challenges Procedure Requirements ROI – – – Why is Return On Investment (ROI) Important? Competing Business Objectives? How ROI Helps Exposure Control Benefit Highlights Examples • Multi Product Facility Containment ROI • Single Product Facility Containment ROI • Rigid or Flexible - determining the most cost effective solution • • Summary Resources

Exposure Control Strategy • Goal – Implement an exposure reduction strategy to reduce exposure risks at the source through primary controls • Drivers – Risk Reduction – Employee Safety – Quality (Airborne & Mechanical Transfer) – Consistency – New facilities, existing facilities and acquisitions – Financial Benefits - ROI • Elements • Procedure • Determine most cost effective solution • Demonstrate Value – ROI

Exposure Control Strategy Challenges • Operations may tend towards facility/infrastructure controls in isolation – Facility controls may not based on primary containment being in place – Equipment and therefore primary process containment may be out of scope – Considering facilities or products not processes • • Capital Request Process Inadequate allocation of resources Validation/Quality road blocks Knowing how to begin & what the best options are

Exposure Control Procedure • Purpose: – Drive exposure control (containment) below the OEL – Prescribe facility/infrastructure controls & PPE based on primary process controls – Ensure consistency throughout the organization for new and existing facilities and processes • Primary Components – Requirements – Risk Matrices – Tool box Process Containment
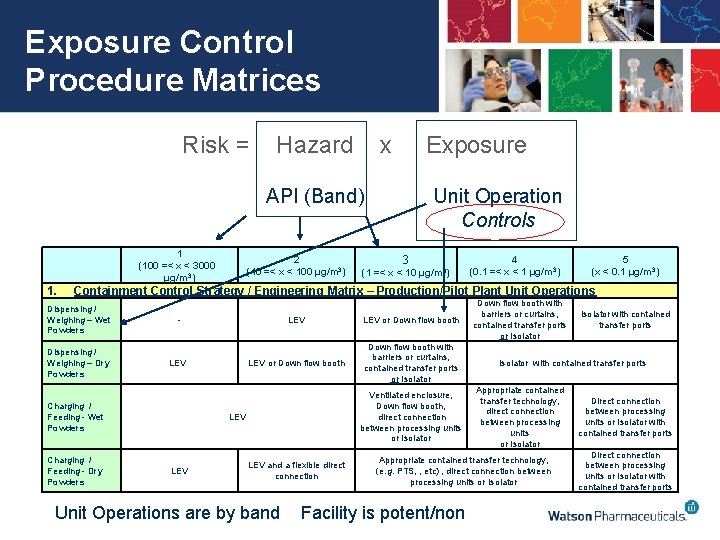
Exposure Control Procedure Matrices Risk = Hazard x Exposure API (Band) 1 (100 =< x < 3000 µg/m 3) 1. 2 (10 =< x < 100 µg/m 3) Unit Operation Controls 3 (1 =< x < 10 µg/m 3) 4 (0. 1 =< x < 1 µg/m 3) 5 (x < 0. 1 µg/m 3) Containment Control Strategy / Engineering Matrix – Production/Pilot Plant Unit Operations Dispensing / Weighing – Wet Powders Dispensing / Weighing – Dry Powders - LEV Charging / Feeding - Wet Powders Charging / Feeding - Dry Powders LEV or Down flow booth with barriers or curtains, contained transfer ports or Isolator Ventilated enclosure, Down flow booth, direct connection between processing units or Isolator LEV LEV and a flexible direct connection Unit Operations are by band Down flow booth with barriers or curtains, contained transfer ports or Isolator with contained transfer ports Appropriate contained transfer technology, direct connection between processing units or Isolator Appropriate contained transfer technology, (e. g. PTS, , etc) , direct connection between processing units or Isolator Facility is potent/non Isolator with contained transfer ports Direct connection between processing units or Isolator with contained transfer ports

Exposure Control Procedure Matrices API/OSD Production & Pilot Plant Facility & Infrastructure Unit Operations Labs Facility & Infrastructure Unit Operations • Used for new facilities (design) and existing ones (gap analysis) • Unit operation control matrices identify a control approach demonstrated to be effective at controlling exposure below the OEL. • If a desired control is not in line with the matrix recommendations, a documented explanation for how equivalent control levels will be achieved is required for approval

Exposure Control Procedure “Tool Box” • The “Tool Box”, is a library of containment solutions meant to supplement the matrices • The “Tool Box” is organized by: – Unit operation – Control Technology EZI Dock High Shear Mixer Glove Bag
Exposure Control Strategy ROI Can Help Implementation Exposure Control Procedure Management Commitment

Why is ROI Important? § It’s a gauge of managerial effectiveness § "Leaving the question of price aside, the best business to own is one that over an extended period can employ large amounts of incremental capital at very high rates of return. The worst business to own is one that must, or will, do the opposite – that is, consistently employ evergreater amounts of capital at very low rates of return. " - Warren Buffett, 1992 Chairman's Letter § The average ROI for American companies is about 11%

Competing Business Objectives? • EHS is important to everyone in the company – EHS provides technical expertise regarding compliance requirements and assistance with meeting EHS goals – Management is held accountable • Optimal financial performance is essential for the organization’s success – Management is held accountable • Will Objectives be balanced? Budget Schedule Safety Quality

How ROI Helps • “Safety people have their hearts in the right place but seldom understand true business objectives” • “We seldom make a credible business case” • ROI can: – help demonstrate financial benefits – identify the most cost effective compliance solution • Demonstrating the value shows management how EHS projects can be aligned with business objectives instead of competing with them – Above quotes from Dave Ehrets – VP Sikorsky Aircraft

Exposure Control Benefit Highlights • Reliably controlling exposure below the OEL reduces exposure risk and adds business benefits: – Energy Savings • Reduced amount of air changes • Air re-circulation instead of once through air – Less elaborate air locks/decontamination systems – Reduced Medical Surveillance Requirements – PPE cost savings • PPE use reduced • Waste disposal of PPE • Operational Efficiency (Gowning/De-gowning) – Increased yield

ROI Process Steps Identify Opportunity Explore Alternatives Conduct Analysis Recommendation • Step 1 – Understand the Opportunity or Challenge • Step 2 – Identify and Explore Alternative Solutions • Step 3 – Gather Data and Conduct Analysis – Costs and Benefits • Step 4 – Review Results and Make a Recommendation • (ROHSEI Process Steps)

ROI Examples Multi Product Facility Containment Project Single Product Containment Project Rigid or Flexible

Project Backgrounds (Step 1) • Multi Product Site ROI Performed: – For the benefit of the site to understand the current costs and potential future benefits for containment • Single Product ROI Performed: – For the same purpose as above except with a narrower focus of just the main product at the facility • Rigid or Flexible Compression Containment Performed: – To determine the most cost effective solution between a rigid and a flexible option
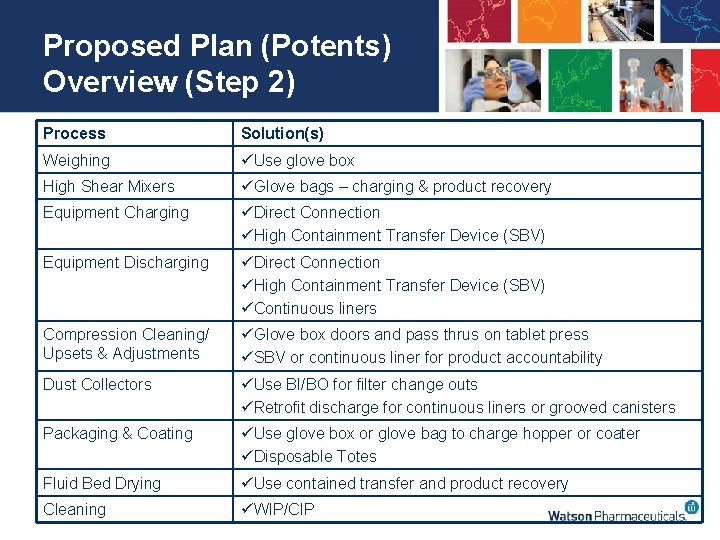
Proposed Plan (Potents) Overview (Step 2) Process Solution(s) Weighing üUse glove box High Shear Mixers üGlove bags – charging & product recovery Equipment Charging üDirect Connection üHigh Containment Transfer Device (SBV) Equipment Discharging üDirect Connection üHigh Containment Transfer Device (SBV) üContinuous liners Compression Cleaning/ Upsets & Adjustments üGlove box doors and pass thrus on tablet press üSBV or continuous liner for product accountability Dust Collectors üUse BI/BO for filter change outs üRetrofit discharge for continuous liners or grooved canisters Packaging & Coating üUse glove box or glove bag to charge hopper or coater üDisposable Totes Fluid Bed Drying üUse contained transfer and product recovery Cleaning üWIP/CIP
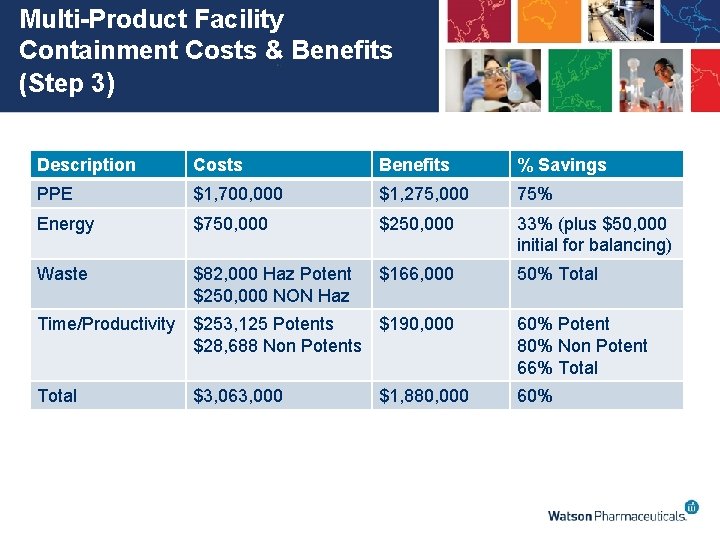
Multi-Product Facility Containment Costs & Benefits (Step 3) Description Costs Benefits % Savings PPE $1, 700, 000 $1, 275, 000 75% Energy $750, 000 $250, 000 33% (plus $50, 000 initial for balancing) Waste $82, 000 Haz Potent $250, 000 NON Haz $166, 000 50% Total Time/Productivity $253, 125 Potents $190, 000 $28, 688 Non Potents 60% Potent 80% Non Potent 66% Total $3, 063, 000 60% $1, 880, 000

Discussion • Assessment does not consider: – decreased production efficiency – decreased cleaning time – increased yields • Costs and benefits are spread over 2 years – estimated to be the implementation time • Capital costs based on 5 potent and 3 non potent simultaneous operations (based on batch projections) – More simultaneous batches would have more costs but also more savings • ROHSEI ROI software makes it easy to change scenarios and look at them from other angles

Cost Benefit Highlights (Step 4) • Total Capital Costs - $2, 145, 000 (Over 2 years) – ROI also includes initial $50, 000 HVAC balancing and $200, 000 for validation work for implementation (CV, etc. ) • Total Recurring Annual Costs for glove bags (Already being spent) - $ 100, 000 + $50, 000 additional for Non Potent products $150, 000 total • Annual Savings - $1, 880, 000 • Net Annual Savings - $1, 730, 000 • ROI – 71% • IRR – 45 % • DPP – 2. 7 years • NPV - $ 2, 950, 954 • ROI is much higher than Average ROI.
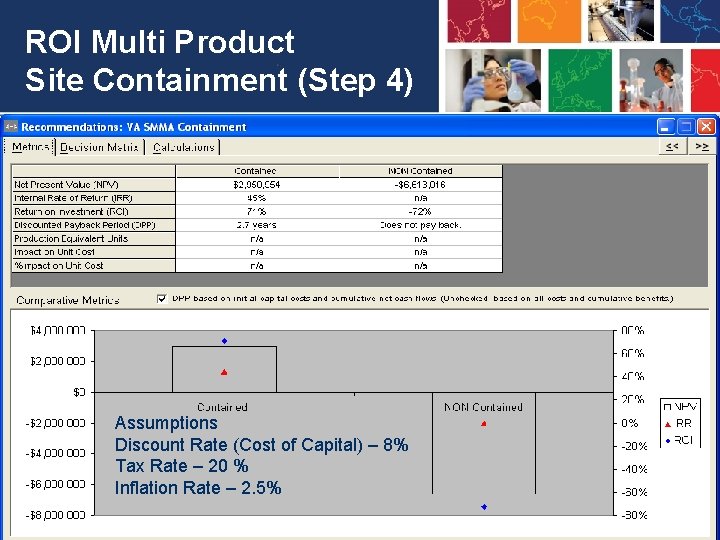
ROI Multi Product Site Containment (Step 4) Assumptions Discount Rate (Cost of Capital) – 8% Tax Rate – 20 % Inflation Rate – 2. 5% 24

Looking at it another way • It’s difficult to test the ROI until after the project is implemented so assumptions may be challenged • ROI Software makes it easy to – Change the assumptions – Look at it from another angle

Cost Benefit Highlights Single Product • • Same facility as previous evaluation Single product with 2 formulations and 9 strengths Cost estimate based on 3 simultaneous operations Product is 30% of manufacturing done at the site • • • Total Initial Capital Costs - $688, 000 Total Recurring Annual Costs - $ 60, 000 Annual Savings - $607, 000 IRR – 55% ROI – 53% DPP – 1. 6 years NPV - $773, 000 Annual Net Savings of $547, 000 ROI is much higher than Average
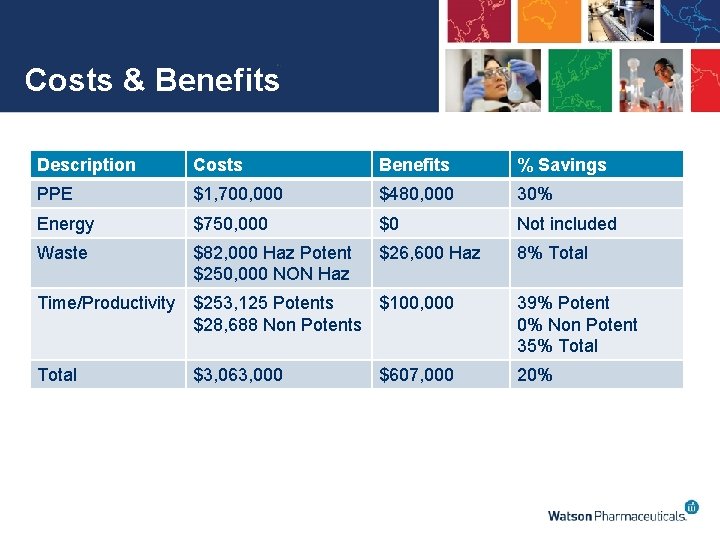
Costs & Benefits Description Costs Benefits % Savings PPE $1, 700, 000 $480, 000 30% Energy $750, 000 $0 Not included Waste $82, 000 Haz Potent $250, 000 NON Haz $26, 600 Haz 8% Total Time/Productivity $253, 125 Potents $100, 000 $28, 688 Non Potents 39% Potent 0% Non Potent 35% Total $3, 063, 000 20% $607, 000

Determining the Most Cost Effective Solution Rigid Assuming each solution is just as effective from a containment and operational perspective; Which is most cost effective? Rigid or Flexible? Where is the tipping point? Flexible

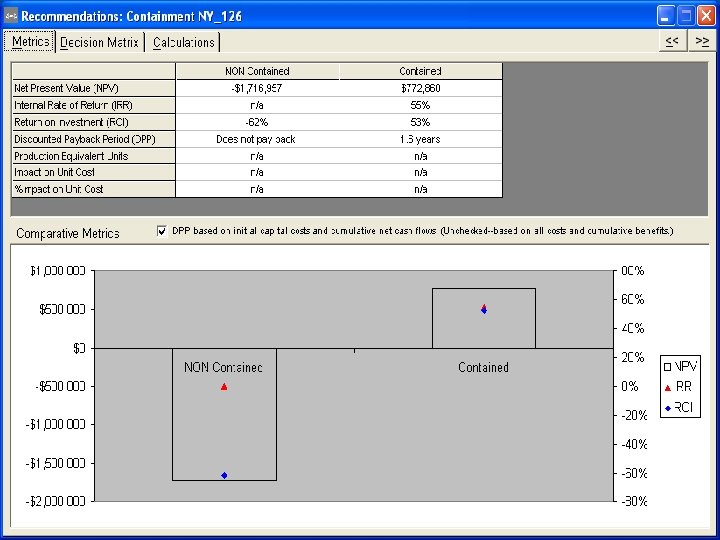
Compression Containment Cost Considerations • 200 -250 batches per press per year • Soft wall tablet press isolator is $1300 -1500 ea • Campaign averages 5 batches per campaign – Average consumable batch cost is $260 -$300 • The cost of consumables for the soft wall will run $52, 000 -$75, 000 per press/yr • Flanges and supports for the soft wall isolator are estimated at $5, 000 per press • A rigid tablet press isolator costs between $50, 000 -75, 000 per press with $5, 000 in consumables/yr
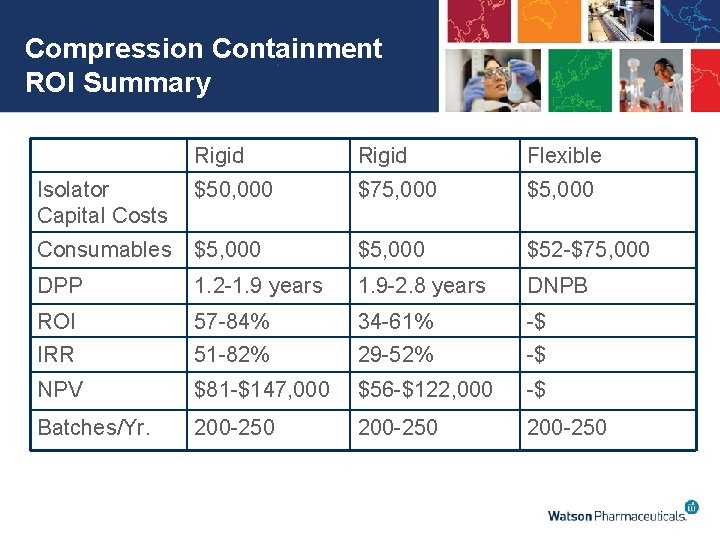
Compression Containment ROI Summary Rigid Flexible Isolator Capital Costs $50, 000 $75, 000 $5, 000 Consumables $5, 000 $52 -$75, 000 DPP 1. 2 -1. 9 years 1. 9 -2. 8 years DNPB ROI 57 -84% 34 -61% -$ IRR 51 -82% 29 -52% -$ NPV $81 -$147, 000 $56 -$122, 000 -$ Batches/Yr. 200 -250

Discussion • Rigid isolator is the most cost effective option based on the number of batches per year • If the number of batches decreases or the price of the rigid Isolator increases this may no longer be the case

Summary • Challenges posed by implementing an exposure control strategy can be dealt with by: – Having an effective exposure control procedure – Using ROI to assist with obtaining management commitment and picking the most cost effective solution • Every company’s EHS mission should include ensuring compliance, protecting the environment and the health & safety of its employees • Financial cost benefit analysis should never be a pre-requisite for EHS compliance

ROI Resources • ROHSEI software and associated training available through Mercer ORC Networks : – http: //www. orc-dc. com/ • AIHA’s Value Strategy Workbook may be purchased through AIHA – http: //www. aiha. org/votp_new/study/index. html • Available Soon from AIHA’s Value Strategy – Software – Exposure Assessment Guideline

Questions? • • Rob Mc Cafferty, CIH Associate Director Global H&S Watson Pharmaceuticals Morris Corporate Center III 400 Interpace Parkway Parsippany, NJ 07054 (862) 262 -7420 Robert. Mc. Cafferty@Watson. com
- Slides: 35