EXPLORING THE BINDING OF METHANOL WITH FURANS Mariyam

EXPLORING THE BINDING OF METHANOL WITH FURANS Mariyam Fatima 1, Cristóbal Pérez 1, Daniel A. Obenchain 1, Melanie Schnell 1, 2 1 Deutsche Elektronen Synchrotron (DESY), Hamburg, Germany zu Kiel, Institut für Physikalische Chemie, 2 Christian Albrechts Universität Germany
The First Blind Challenge A theoretical and experimental collaboration • Focus on OH and OD stretching regions • Looking for relative preference of methanol to the O or π docking sites • Furan, methyl furan, and dimethyl furan • The preference was not clear from infrared data alone • The project was double blind • Theoreticians and experimentalists didn’t know each other’s results | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 2

Typical Microwaver Arrogance Yeah, we can solve that problem. Piece of cake! Then the realization hits that we can’t look at a structure before assignments… Wait, three methyl tops at once? !? ! | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 3

Double Blind Study Overview Provided information for the microwave experiment: • Suggestions on carrier gases • For furan methanol complex • Initial assignments were made by pattern recognition • later confirmed to be the furan methanol complex • Note: Any figure we make that shows a structure, we had to generate ourselves. Typically this means we had access to a Cartesian structures there were somehow predicted by computational chemistry. (i. e. we ran some calculations ourselves) For dimethyl furan methanol • Predictions of rotational constants and dipole moments were provided • No information on relative energies of the isomers | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 4
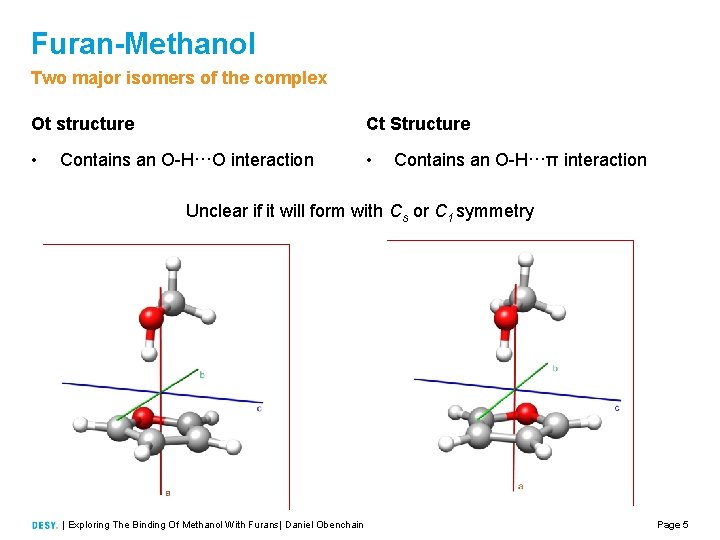
Furan-Methanol Two major isomers of the complex Ot structure Ct Structure • • Contains an O H···O interaction Contains an O H···π interaction Unclear if it will form with Cs or C 1 symmetry | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 5

Instrumentation Chirped-pulse Fourier transform microwave spectroscopy – CP-FTMW Spectrometer in Hamburg COMPACT Methanol Furan Oscilloscope Low Noise Amplifier Arbitrary Waveform Generator Traveling-Wave Tube Amplifier Technical details Frequency range Line resolution Rep. rate Carrier gas Backing pressure T nozzle 2 – 18 GHz 15 k. Hz 8 Hz Neon ≈ 3 bar RT | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 6
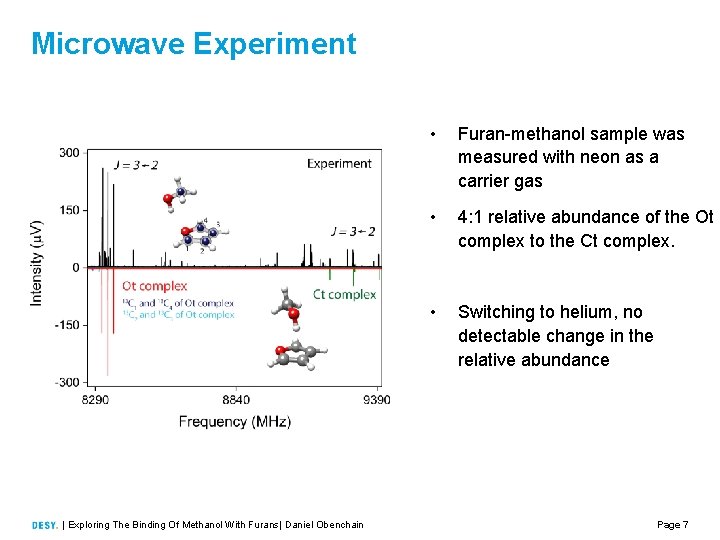
Microwave Experiment | Exploring The Binding Of Methanol With Furans| Daniel Obenchain • Furan methanol sample was measured with neon as a carrier gas • 4: 1 relative abundance of the Ot complex to the Ct complex. • Switching to helium, no detectable change in the relative abundance Page 7
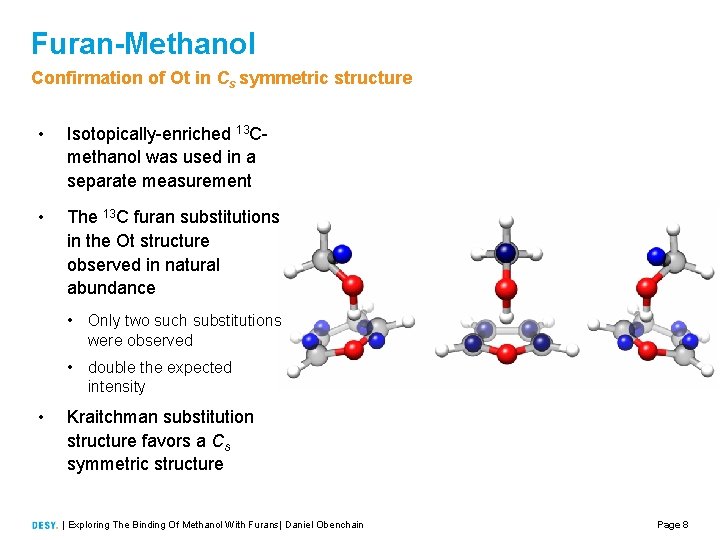
Furan-Methanol Confirmation of Ot in Cs symmetric structure • Isotopically enriched 13 C methanol was used in a separate measurement • The 13 C furan substitutions in the Ot structure observed in natural abundance • Only two such substitutions were observed • double the expected intensity • Kraitchman substitution structure favors a Cs symmetric structure | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 8
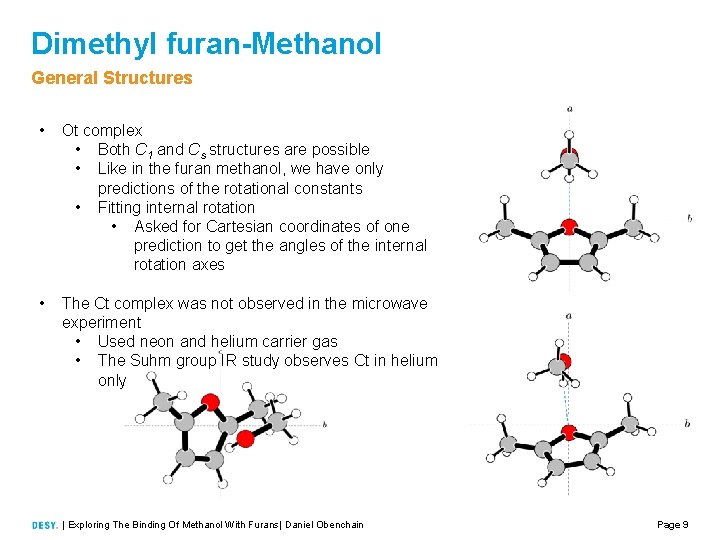
Dimethyl furan-Methanol General Structures • Ot complex • Both C 1 and Cs structures are possible • Like in the furan methanol, we have only predictions of the rotational constants • Fitting internal rotation • Asked for Cartesian coordinates of one prediction to get the angles of the internal rotation axes • The Ct complex was not observed in the microwave experiment • Used neon and helium carrier gas • The Suhm group IR study observes Ct in helium only | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 9
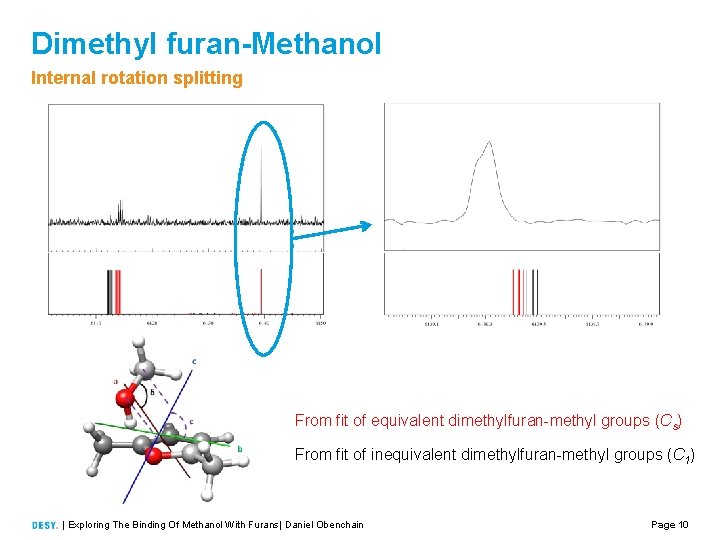
Dimethyl furan-Methanol Internal rotation splitting From fit of equivalent dimethylfuran methyl groups (Cs) From fit of inequivalent dimethylfuran methyl groups (C 1) | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 10
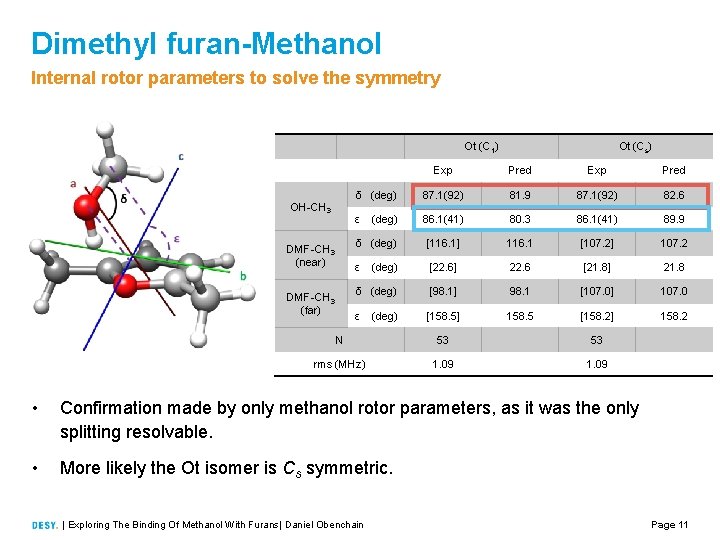
Dimethyl furan-Methanol Internal rotor parameters to solve the symmetry Ot (C 1) OH CH 3 DMF CH 3 (near) DMF CH 3 (far) Ot (Cs) Exp Pred δ (deg) 87. 1(92) 81. 9 87. 1(92) 82. 6 ε 86. 1(41) 80. 3 86. 1(41) 89. 9 δ (deg) [116. 1] 116. 1 [107. 2] 107. 2 ε (deg) [22. 6] 22. 6 [21. 8] 21. 8 δ (deg) [98. 1] 98. 1 [107. 0] 107. 0 ε [158. 5] 158. 5 [158. 2] 158. 2 (deg) N 53 53 rms (MHz) 1. 09 • Confirmation made by only methanol rotor parameters, as it was the only splitting resolvable. • More likely the Ot isomer is Cs symmetric. | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 11
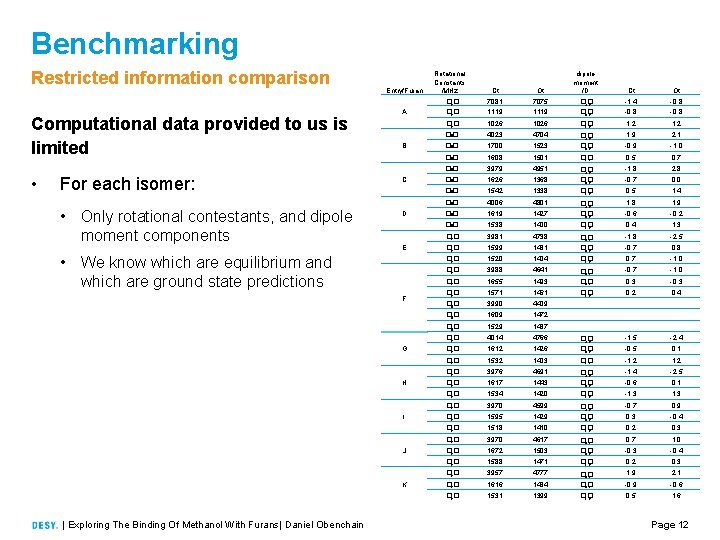
Benchmarking Restricted information comparison Computational data provided to us is limited • For each isomer: • Only rotational contestants, and dipole moment components Entry/Furan A B C D E • We know which are equilibrium and which are ground state predictions F G H I J K | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Rotational Constants /MHz Ct Ot dipole moment /D Ct Ot �� e 7081 1119 7075 1119 �� a, e �� b, e 1. 4 0. 8 1. 2 �� e 1026 �� c, e 1. 2 �� e 4023 4704 1. 9 2. 1 �� e 1700 1523 �� a, e �� b, e 0. 9 1. 0 �� e 1608 1501 �� c, e 0. 5 0. 7 �� e 3979 4951 2. 8 �� e 1626 1368 �� a, e �� b, e 1. 8 0. 7 0. 0 �� e 1542 1338 �� c, e 0. 5 1. 4 �� e 4006 4801 1. 8 1. 9 �� e 1619 1427 �� a, e �� b, e 0. 6 0. 2 �� e 1538 1400 �� c, e 0. 4 1. 3 �� e 3981 4738 1. 8 2. 5 �� e 1599 1431 �� a, e �� b, e 0. 7 0. 8 �� e 1520 1404 �� c, e 0. 7 1. 0 �� e 3988 4641 1. 0 1655 1493 �� a, e �� b, e 0. 7 �� e 0. 3 �� e 1571 1461 �� c, e 0. 2 0. 4 �� 0 3990 4409 �� 0 1609 1472 �� 0 1529 1437 �� e 4014 4766 2. 4 1612 1426 �� a, e �� b, e 1. 5 �� e 0. 5 0. 1 �� e 1532 1403 �� c, e 1. 2 �� e 3976 4691 1. 4 2. 5 �� e 1617 1443 �� a, e �� b, e 0. 6 0. 1 �� e 1534 1420 �� c, e 1. 3 �� e 3970 4599 0. 7 0. 9 �� e 1595 1429 �� a, e �� b, e 0. 3 0. 4 �� e 1518 1410 �� c, e 0. 2 0. 3 �� e 3970 4617 0. 7 1. 0 �� e 1672 1503 �� a, e �� b, e 0. 3 0. 4 �� e 1588 1471 �� c, e 0. 2 0. 3 �� e 3957 4777 1. 9 2. 1 �� e 1616 1434 �� a, e �� b, e 0. 9 0. 6 �� e 1531 1399 �� c, e 0. 5 1. 6 Page 12
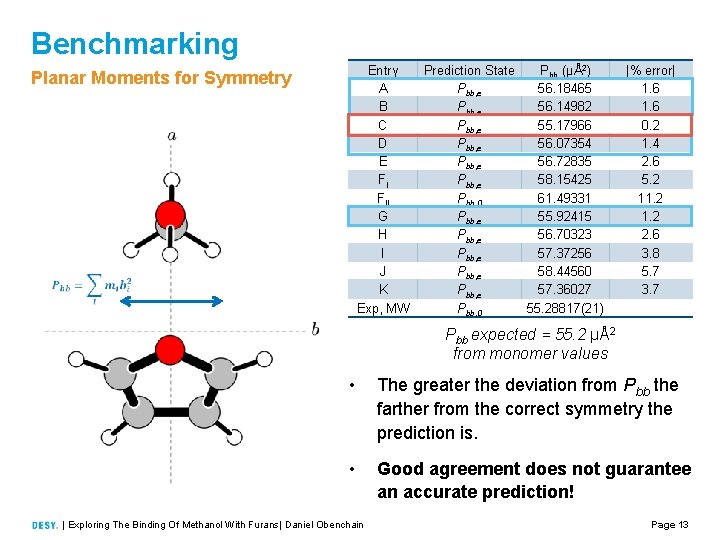
Benchmarking Entry A B C D E FI FII G H I J K Exp, MW Planar Moments for Symmetry Prediction State Pbb (μÅ2) Pbb, e 56. 18465 Pbb, e 56. 14982 Pbb, e 55. 17966 Pbb, e 56. 07354 Pbb, e 56. 72835 Pbb, e 58. 15425 Pbb, 0 61. 49331 Pbb, e 55. 92415 Pbb, e 56. 70323 Pbb, e 57. 37256 Pbb, e 58. 44560 Pbb, e 57. 36027 Pbb, 0 55. 28817(21) |% error| 1. 6 0. 2 1. 4 2. 6 5. 2 11. 2 2. 6 3. 8 5. 7 3. 7 Pbb expected = 55. 2 μÅ2 from monomer values • The greater the deviation from Pbb the farther from the correct symmetry the prediction is. • Good agreement does not guarantee an accurate prediction! | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 13
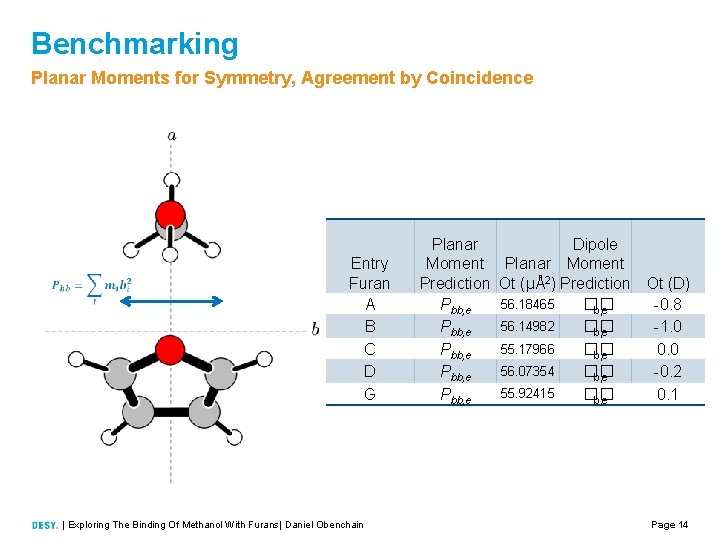
Benchmarking Planar Moments for Symmetry, Agreement by Coincidence Entry Furan A B C D G | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Planar Moment Prediction Pbb, e Pbb, e Dipole Planar Moment Ot (μÅ2) Prediction Ot (D) 56. 18465 �� 0. 8 b, e 56. 14982 �� 1. 0 b, e 55. 17966 �� 0. 0 b, e 56. 07354 �� 0. 2 b, e 55. 92415 �� 0. 1 b, e Page 14
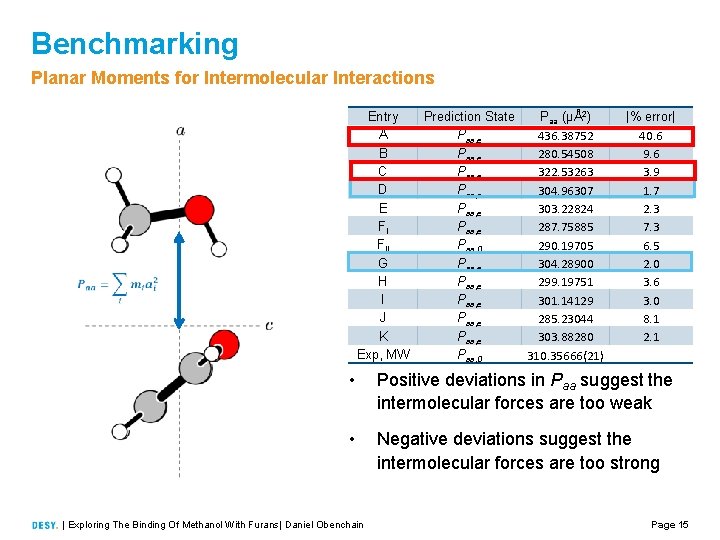
Benchmarking Planar Moments for Intermolecular Interactions Entry A B C D E FI FII G H I J K Exp, MW Prediction State Paa (μÅ2) Paa, e 436. 38752 Paa, e 280. 54508 Paa, e 322. 53263 Paa, e 304. 96307 Paa, e 303. 22824 Paa, e 287. 75885 Paa, 0 290. 19705 Paa, e 304. 28900 Paa, e 299. 19751 Paa, e 301. 14129 Paa, e 285. 23044 Paa, e 303. 88280 Paa, 0 310. 35666(21) |% error| 40. 6 9. 6 3. 9 1. 7 2. 3 7. 3 6. 5 2. 0 3. 6 3. 0 8. 1 2. 1 • Positive deviations in Paa suggest the intermolecular forces are too weak • Negative deviations suggest the intermolecular forces are too strong | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 15

Conclusions How much information do we need? • Rotational spectroscopy for both Ct and Ot isomers observed for the furan methanol complex • Ot complex is favored • Overall symmetry of each isomer was determined • Only the Ot complex of dimethyl furan methanol complex was observed • Benchmarking the computational results based on rotational spectroscopy reveals which entries are better for the determination of methanol complexes. | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 16

Pablo Pinacho, TC 05 Weixing Li, RH 07 Ben Arenas, TA 03 Amanda Steber, RA 03 & FC 01 Melanie Schnell, RH 01 Mariyam Fatima, WB 05 Thanks to the other collaborators on the project, with special thanks to the Suhm group. María Mar Quesada Moreno, RH 05

Contact Deutsches Elektronen Synchrotron www. desy. de Daniel Obenchain FS SMP daniel. obenchain@desy. de +49 40 8998 6247
Heading Subheading, optional | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 19
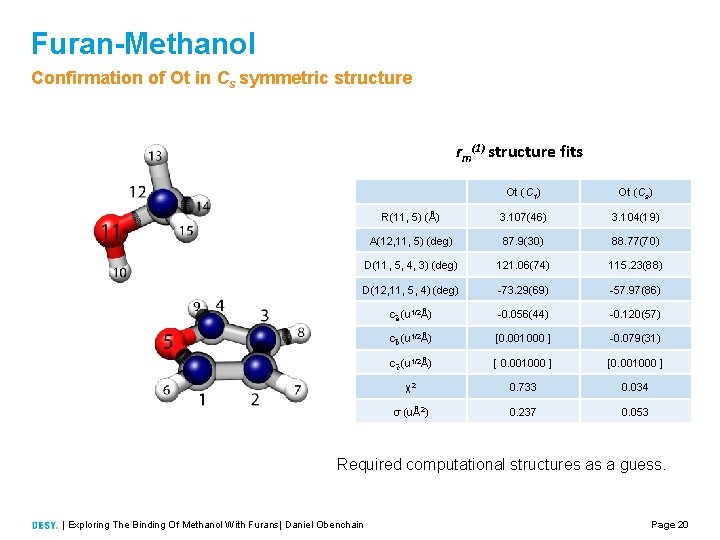
Furan-Methanol Confirmation of Ot in Cs symmetric structure rm(1) structure fits Ot (C 1) Ot (Cs) R(11, 5) (Å) 3. 107(46) 3. 104(19) A(12, 11, 5) (deg) 87. 9(30) 88. 77(70) D(11, 5, 4, 3) (deg) 121. 06(74) 115. 23(88) D(12, 11, 5, 4) (deg) 73. 29(69) 57. 97(86) c a (u 1/2Å) 0. 056(44) 0. 120(57) c b (u 1/2Å) [0. 001000 ] 0. 079(31) c c (u 1/2Å) [ 0. 001000 ] [0. 001000 ] χ2 0. 733 0. 034 σ (uÅ2) 0. 237 0. 053 Required computational structures as a guess. | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 20
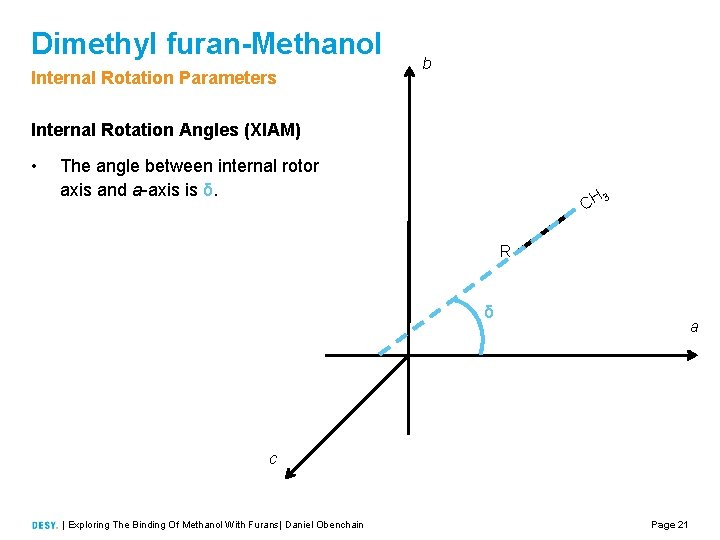
Dimethyl furan-Methanol Internal Rotation Parameters b Internal Rotation Angles (XIAM) • The angle between internal rotor axis and a axis is δ. CH 3 R δ a c | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 21
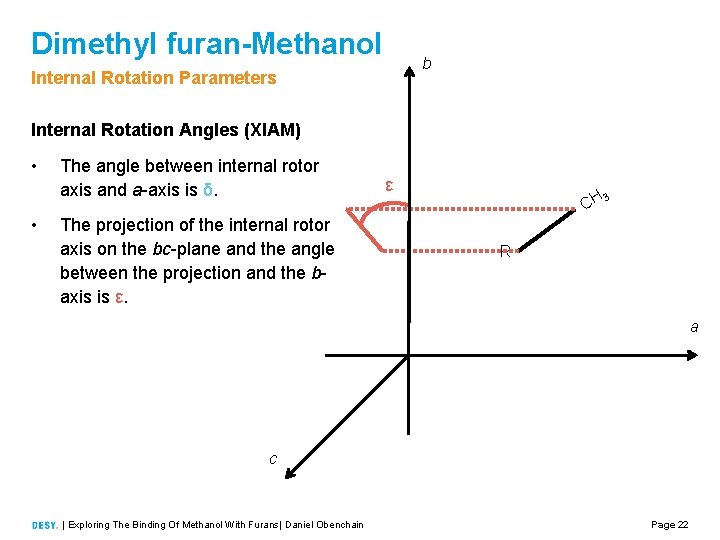
Dimethyl furan-Methanol b Internal Rotation Parameters Internal Rotation Angles (XIAM) • • The angle between internal rotor axis and a axis is δ. The projection of the internal rotor axis on the bc plane and the angle between the projection and the b axis is ε. ε CH 3 R a c | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 22
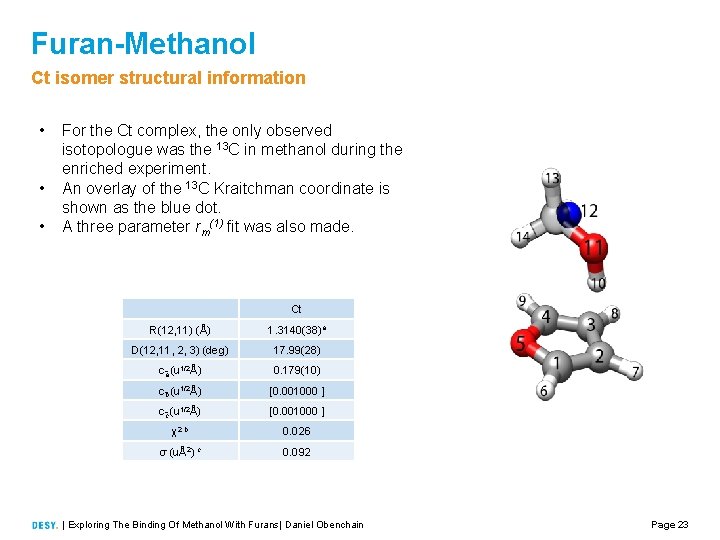
Furan-Methanol Ct isomer structural information • • • For the Ct complex, the only observed isotopologue was the 13 C in methanol during the enriched experiment. An overlay of the 13 C Kraitchman coordinate is shown as the blue dot. A three parameter rm(1) fit was also made. Ct R(12, 11) (Å) 1. 3140(38)a D(12, 11, 2, 3) (deg) 17. 99(28) c a (u 1/2Å) 0. 179(10) c b (u 1/2Å) [0. 001000 ] c c (u 1/2Å) [0. 001000 ] χ2 b 0. 026 σ (uÅ2) c 0. 092 | Exploring The Binding Of Methanol With Furans| Daniel Obenchain Page 23
- Slides: 23