Experimental Study Design RCT Study Designs OBSERVATIONAL Analytic

Experimental Study Design RCT
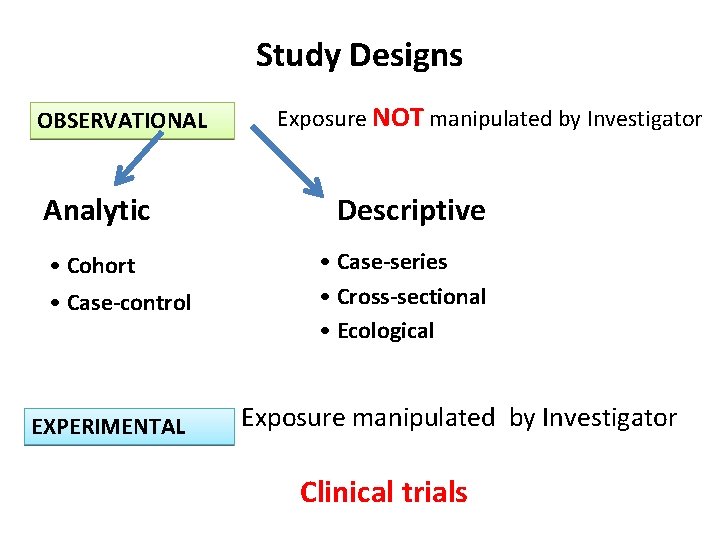
Study Designs OBSERVATIONAL Analytic • Cohort • Case-control EXPERIMENTAL Exposure NOT manipulated by Investigator Descriptive • Case-series • Cross-sectional • Ecological Exposure manipulated by Investigator Clinical trials
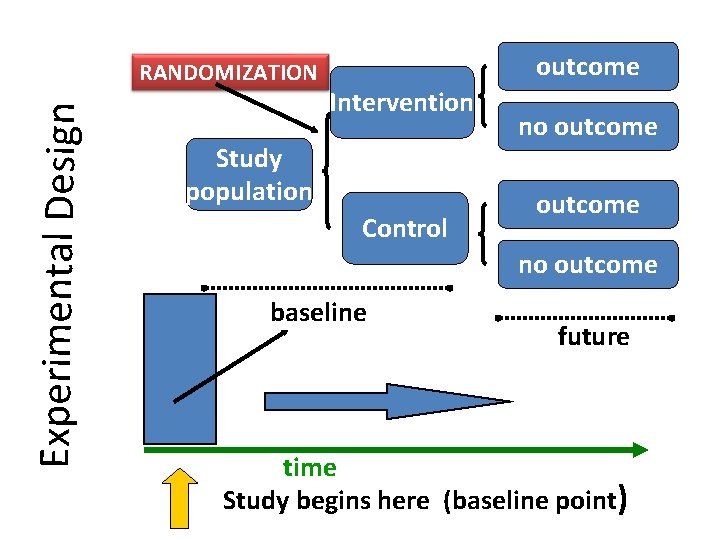
Experimental Design RANDOMIZATION outcome Intervention Study population Control no outcome baseline future time Study begins here (baseline point)
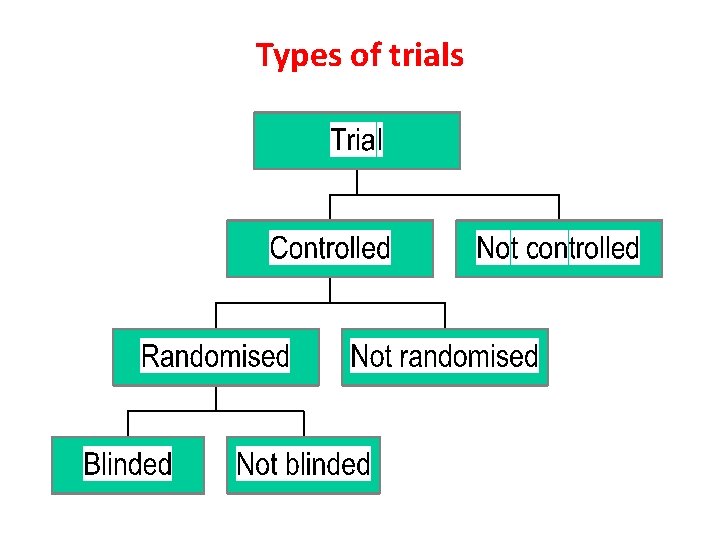
Types of trials

Randomized controlled trials (RCT) An epidemiological experiment in which subjects in a population are randomly allocated into groups, usually called study and control groups to receive and not receive an experimental preventive or therapetuic procedure, maneuver, or intervention John M. Last, 2001

EXPERIMENTAL STUDIES: Are of two types. Randomized Controlled Trials. Non Randomized Controlled Trials 1. Randomized Controlled Trials: Number one method of evaluation, basic steps are Drawing up a Protocol v. Objectives v. Questions to be asked v. Criteria for selection v. Size of sample v. Allocation in study & control groups v. Treatment to be given v. Standardization of Procedures
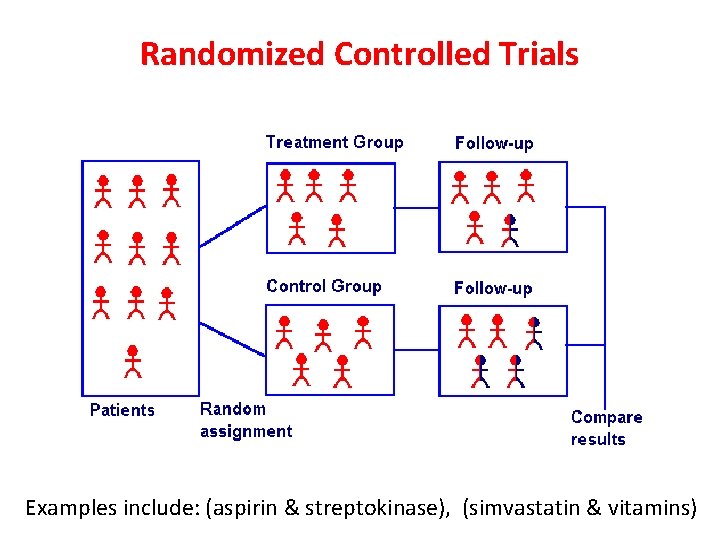
Randomized Controlled Trials Examples include: (aspirin & streptokinase), (simvastatin & vitamins)
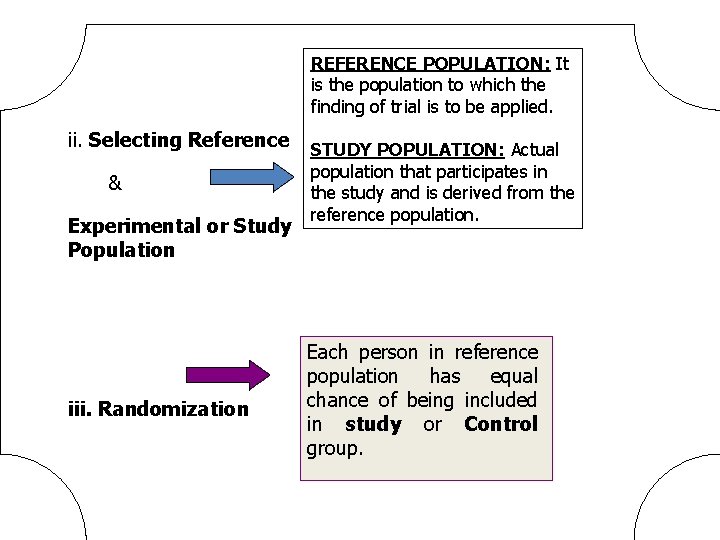
REFERENCE POPULATION: It is the population to which the finding of trial is to be applied. ii. Selecting Reference & Experimental or Study Population iii. Randomization STUDY POPULATION: Actual population that participates in the study and is derived from the reference population. Each person in reference population has equal chance of being included in study or Control group.

iv. Manipulation Deliberate application or withdrawal or reduction of suspected causal factor (vaccine, dietary component, habit). v. Follow Up Examination of the experimental and control groups at a defined interval of time. vi. Assessment of out comes in terms of v. Positive result Benefits v. Negative result Side effects Both are compared in both groups.

NON-RANDOMIZED TRIALS • When RCT is not possible on ethical, adm grounds ·When preventive measures can be applied on community basis ·When disease frequency is low and natural history is long ·When cost and logistic is limited
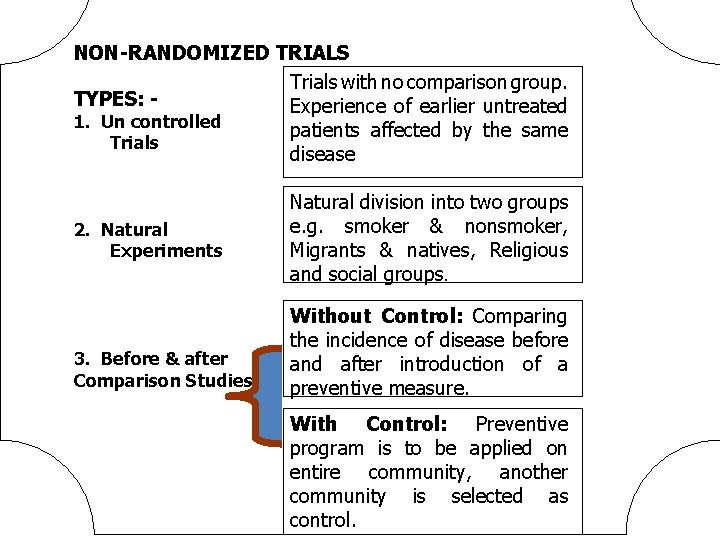
NON-RANDOMIZED TRIALS Trials with no comparison group. TYPES: Experience of earlier untreated 1. Un controlled patients affected by the same Trials disease 2. Natural Experiments 3. Before & after Comparison Studies Natural division into two groups e. g. smoker & nonsmoker, Migrants & natives, Religious and social groups. Without Control: Comparing the incidence of disease before and after introduction of a preventive measure. With Control: Preventive program is to be applied on entire community, another community is selected as control.

Experimental Study Examples • Randomized clinical trial to determine if giving magnesium sulfate to pregnant women in preterm labor decreases the risk of their babies developing cerebral palsy • Randomized community trial to determine if fluoridation of the public water supply decreases dental cavities

Types Experimental Study Design 1. Randomized control trials/Clinical trials • • Parallel design Cross over design 2. Field trials 3. Community trials or Quasi study design 13
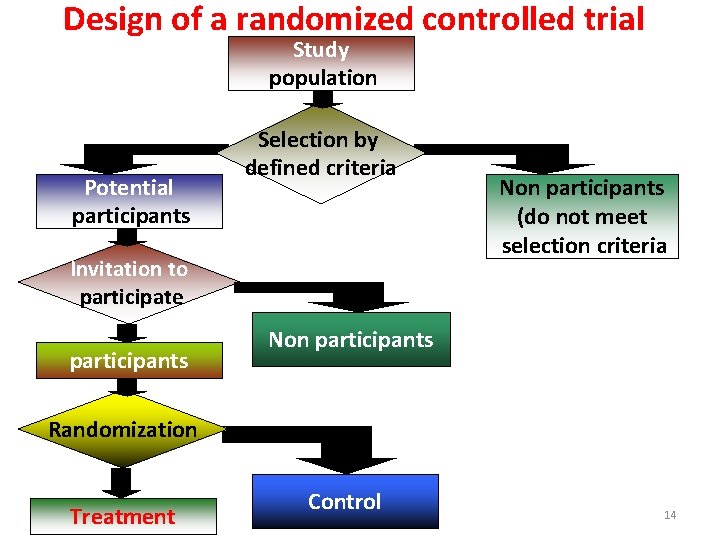
Design of a randomized controlled trial Study population Potential participants Selection by defined criteria Invitation to participate participants Non participants (do not meet selection criteria Non participants Randomization Treatment Control 14

Designs Used in Experimental Studies • Parallel Design • Cross Over Design 15
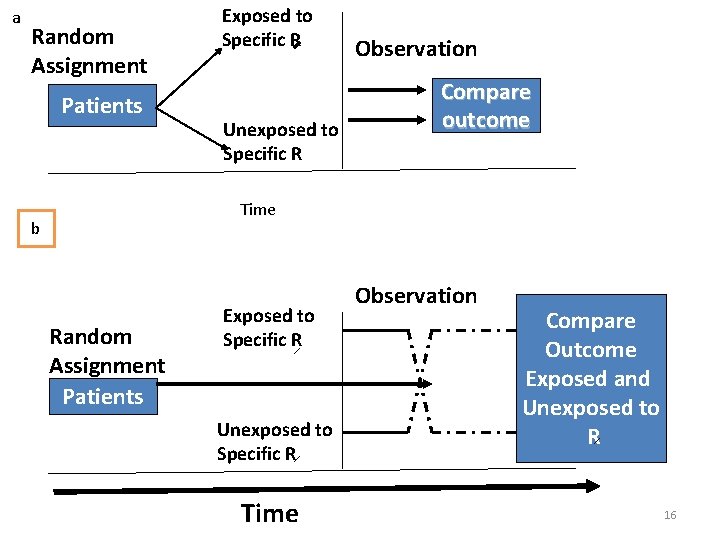
a Random Assignment Patients Exposed to Specific R Unexposed to Specific R Observation Compare outcome Time b Random Assignment Patients Exposed to Specific R Unexposed to Specific R Time Observation Compare Outcome Exposed and Unexposed to R 16

Advantages of Experimental Studies Exposure is under the control of investigator Randomization Blinding eliminates bias Control on time span Confounding factors can be controlled Best method to study causal relationship We can confirm or refute etiological hypothesis on evidence. • Evaluate effectiveness and efficiency of Health services • • 17

Disadvantages of Experimental Studies • Subject exclusion may limit ability to generalize findings to other patients. • A long period of time is often required to reach a conclusion. • A large number of participants may be required. • Financial costs are typically high. • Ethical concerns may arise. • Subjects may not comply with treatment assignments. • Exposure or treatment alternatives should be acceptable to both groups 18

Introduction • Recall the definition of an experiment • Trials - from the French trier (to try) • Clinical trial – apply therapeutic interventions to sick individuals (e. g. , chemotherapy trial) • Field trial – apply preventive interventions to healthy individuals (e. g. , vaccine trial) • Community trial – apply interventions to aggregate units (e. g. , fluoridation of public water) 19
Selected Concepts 1. 2. 3. 4. 5. Control group Randomization Admissibility criteria Outcome ascertainment Ethics All except #2 apply to observational designs as well 20

Element 1. Control Group The effects of an exposure can only be judged in comparison to what would happen in its absence The control group provides this comparison Exposed Not exposed 21

Illustration: “MRFIT” • Multiple Risk Factor Intervention Trial • Exposure: Health education vs. no special intervention • Outcome: CVD • Treatment group experienced dramatic declines in CVD • But so did the control group • Rates were declining in all groups in the 1970 s • Effect of the intervention was negligible 22

Effects from inert interventions Placebo effect – improvement associated with inert interventions Placebo effects are a scientific mystery 23

Hawthorne Effect subjects improve an aspect of their behavior being experimentally measured simply in response to the fact that they are being studied, not in response to any particular experimental manipulation. 24

Element 2. Randomization works by balancing extraneous determinants in the groups being compared, thus mitigating confounding 25

How randomization works Suppose you want to determine whether a particular diet (the exposure) is associated with improved weight gain in lab animals (outcome) Randomization encourages equal numbers of fast-growing rats in each group 26

Polio Field Trial (1954) Polio rates (per 100, 000) Placebo 69 Refusers 46 Vaccinated 28 Note: Had refusers been used as the control group, the effects of the intervention would have been underrated (Am J Pub Health, 1957, 47: 283 -7) Dr. Jonas Salk, 1953 27

3. Admissibility Criteria • Restrict participants to those with uniform characteristics • This too mitigates confounding • Example: Excluding smokers from a study base would prevent confounding from smoking 28

4. Outcome Ascertainment • Outcome ascertainments must be valid • Without valid outcome ascertainment, we have GIGO (garbage in, garbage out) 29

5. Ethics – Respect for individuals – Beneficence – Justice – oversight – Informed consent 30

Unique Problems of Intervention Studies • Ethics – Sufficient doubts to withold from half the population – Sufficient believes to expose half the population – Requires high scientific standards • Feasibility – Widespread adaption of measures by community – Problems of finding sufficiently large eligible sample size • Costs – Expensive

Summary • Gold standard in epidemiological research • Makes study groups comparable – Random allocation – Sufficient sample size • Unique problems of ethics, feasibility and costs • Ensure transparency of all trials
- Slides: 32