Experimental Biology 2004 Cellular Homeostasis Refresher Course Generation

Experimental Biology 2004 Cellular Homeostasis Refresher Course Generation of the Membrane Potential Stephen Wright University of Arizona
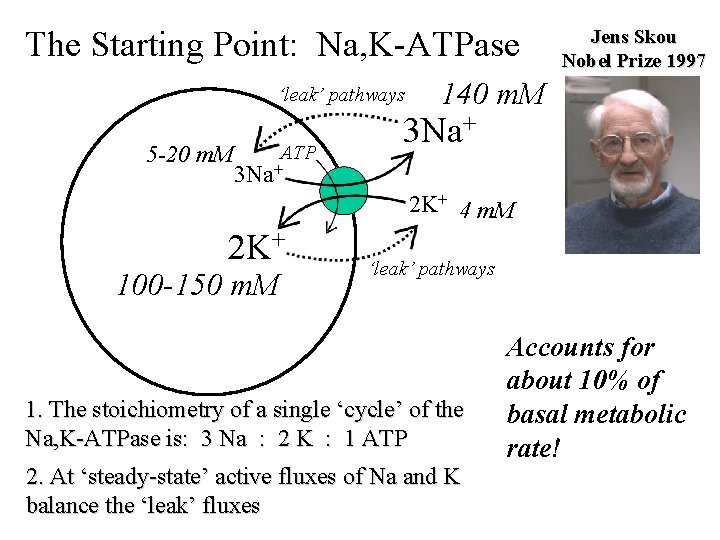
The Starting Point: Na, K-ATPase ‘leak’ pathways 5 -20 m. M ATP Jens Skou Nobel Prize 1997 140 m. M 3 Na+ 2 K+ 4 m. M 2 K+ 100 -150 m. M ‘leak’ pathways 1. The stoichiometry of a single ‘cycle’ of the Na, K-ATPase is: 3 Na : 2 K : 1 ATP 2. At ‘steady-state’ active fluxes of Na and K balance the ‘leak’ fluxes Accounts for about 10% of basal metabolic rate!
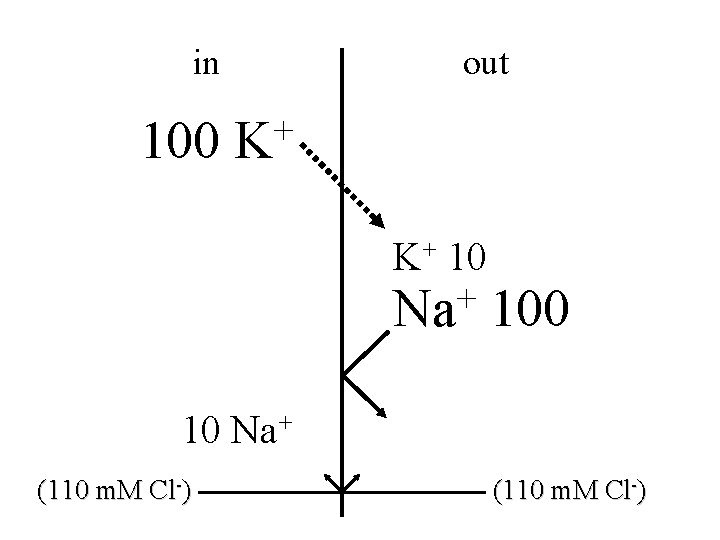
in 100 out + K K+ 10 + Na 100 10 Na+ (110 m. M Cl-)
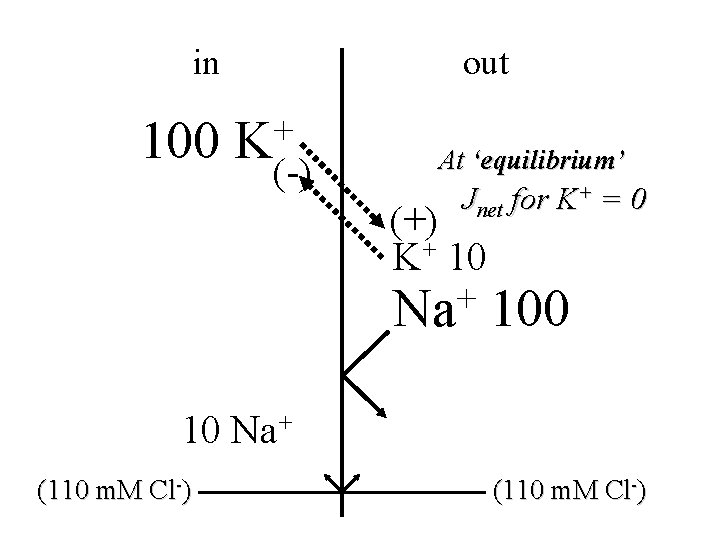
in 100 out + K (-) At ‘equilibrium’ Jnet for K+ = 0 (+) K+ 10 + Na 100 10 Na+ (110 m. M Cl-)
![at Equilibrium: Electrical Force = Chemical Force (electrical ‘voltage’ = chemical ‘gradient’) [K+]in VKz. at Equilibrium: Electrical Force = Chemical Force (electrical ‘voltage’ = chemical ‘gradient’) [K+]in VKz.](http://slidetodoc.com/presentation_image/5590ef1f09d736ebb60a28c9375161f1/image-5.jpg)
at Equilibrium: Electrical Force = Chemical Force (electrical ‘voltage’ = chemical ‘gradient’) [K+]in VKz. KF = -RT ln + [K ]out +] [K -RT in VK = ln + z KF [K ]out Nernst Equation

Walther Hermann Nernst Nobel Prize: 1920
![+] [K -RT in VK = ln + z KF [K ]out R = +] [K -RT in VK = ln + z KF [K ]out R =](http://slidetodoc.com/presentation_image/5590ef1f09d736ebb60a28c9375161f1/image-7.jpg)
+] [K -RT in VK = ln + z KF [K ]out R = 8. 31 J/(mol-deg) T = 310 o K F= 96, 500 coul/mol ln to log (x 2. 303) +] [K -61. 5 m. V in VK = log + z. K [K ]out +] [K -60 m. V VK log + in z. K [K ]out [K+]in = 100 m. M and [K+]out = 10 m. M VK -60 m. V
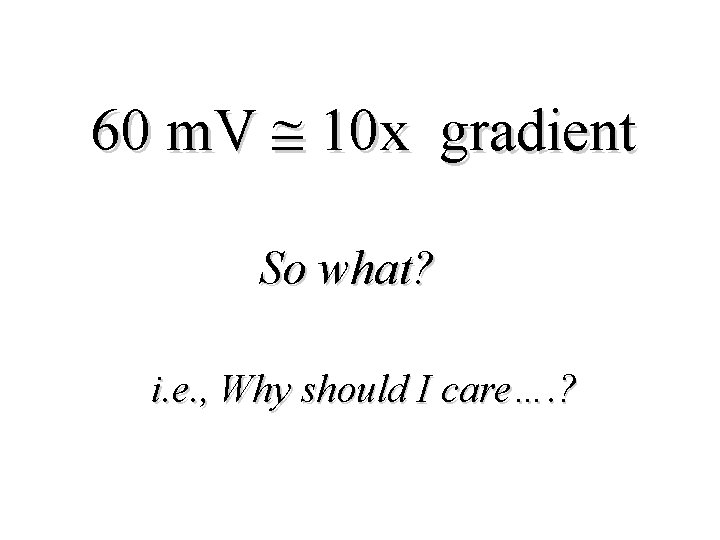
60 m. V 10 x gradient So what? i. e. , Why should I care…. ?

Well. . . The Nernst Equation: 1. Underscores the equality that exists between chemical and electrical forces 2. Provides a tool for predicting if ‘active’ transport processes are present. . . 3. Conceptual ‘starting point’ for understanding the physiological basis of all bioelectric activity

Summary to this point. . Chemical force and Electrical force are ‘two sides of the same coin’ energy (driving forces) Nernst equation displays theoretical balance between these forces Goldman equation reflects the real world situation of finite and variable ionic permeabilities
![2 parameters of Goldman eq: Ion Permeabilities rapidly variable Ion Gradients ‘stable’ PK[K+]in + 2 parameters of Goldman eq: Ion Permeabilities rapidly variable Ion Gradients ‘stable’ PK[K+]in +](http://slidetodoc.com/presentation_image/5590ef1f09d736ebb60a28c9375161f1/image-11.jpg)
2 parameters of Goldman eq: Ion Permeabilities rapidly variable Ion Gradients ‘stable’ PK[K+]in + PNa[Na+]in + PCl [Cl-]out Vm -60 m. V log PK[K+]out + PNa[Na+]out + PCl [Cl-]in ‘Goldman Equation’
![Factors influencing the Membrane Potential at Rest A. Ion Gradients Ion [in] Na+ [out] Factors influencing the Membrane Potential at Rest A. Ion Gradients Ion [in] Na+ [out]](http://slidetodoc.com/presentation_image/5590ef1f09d736ebb60a28c9375161f1/image-12.jpg)
Factors influencing the Membrane Potential at Rest A. Ion Gradients Ion [in] Na+ [out] 12 m. M 140 m. M Veq (Nernst) +64 m. V Pi 10 -9 cm/sec K+ 135 4 -92 10 -7 cm/sec Cl+ 4 116 -88 10 -8 cm/sec B. Membrane Permeability Goldman prediction of Vm = -84 m. V
![PK[K+]in + PNa[Na+]in + PCl [Cl-]out Vm -60 m. V log PK[K+]out + PNa[Na+]out PK[K+]in + PNa[Na+]in + PCl [Cl-]out Vm -60 m. V log PK[K+]out + PNa[Na+]out](http://slidetodoc.com/presentation_image/5590ef1f09d736ebb60a28c9375161f1/image-13.jpg)
PK[K+]in + PNa[Na+]in + PCl [Cl-]out Vm -60 m. V log PK[K+]out + PNa[Na+]out + PCl [Cl-]in VNa Membrane Potential (m. V) +50 0 ‘boundary conditions’ -50 Vm V -100 Cl VK
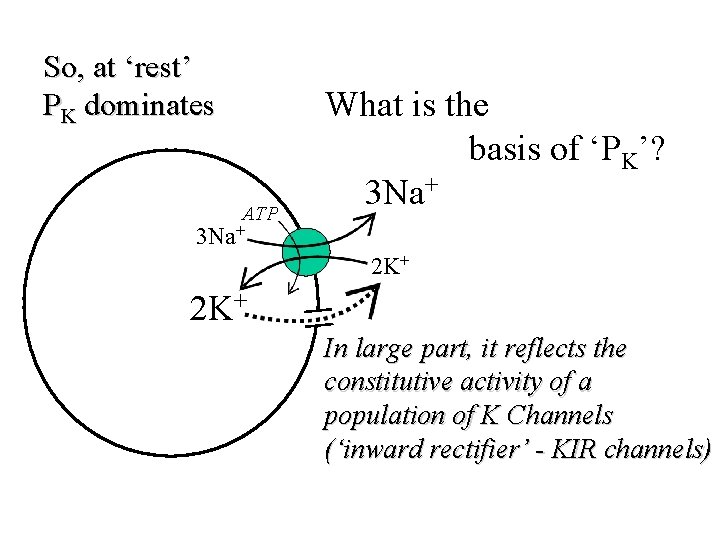
So, at ‘rest’ PK dominates ATP What is the basis of ‘PK’? 3 Na+ 2 K+ In large part, it reflects the constitutive activity of a population of K Channels (‘inward rectifier’ - KIR channels)
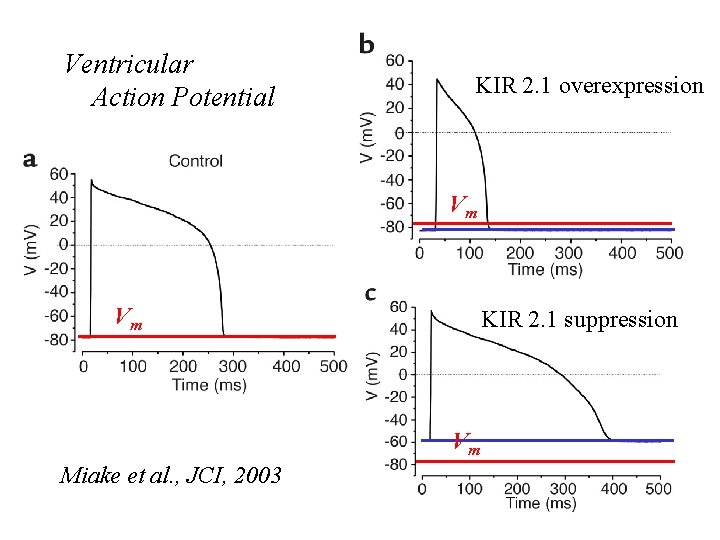
Ventricular Action Potential KIR 2. 1 overexpression Vm Vm KIR 2. 1 suppression Vm Miake et al. , JCI, 2003
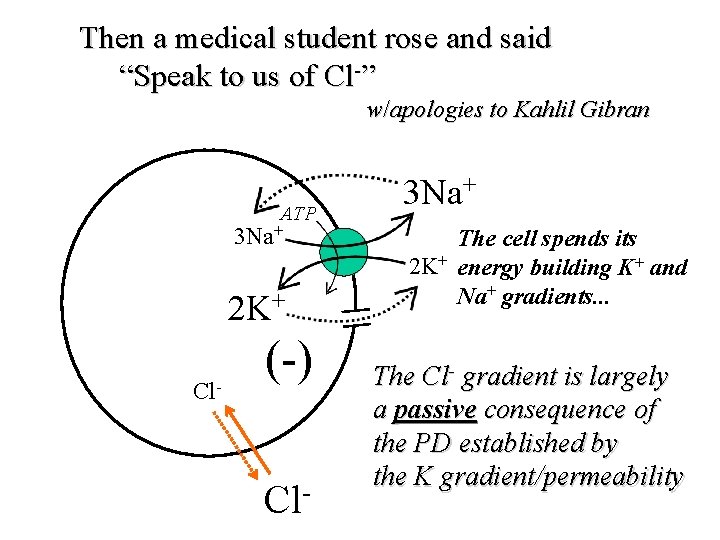
Then a medical student rose and said “Speak to us of Cl-” w/apologies to Kahlil Gibran ATP 3 Na+ 2 K+ Cl- (-) Cl- 3 Na+ The cell spends its 2 K+ energy building K+ and Na+ gradients. . . The Cl- gradient is largely a passive consequence of the PD established by the K gradient/permeability
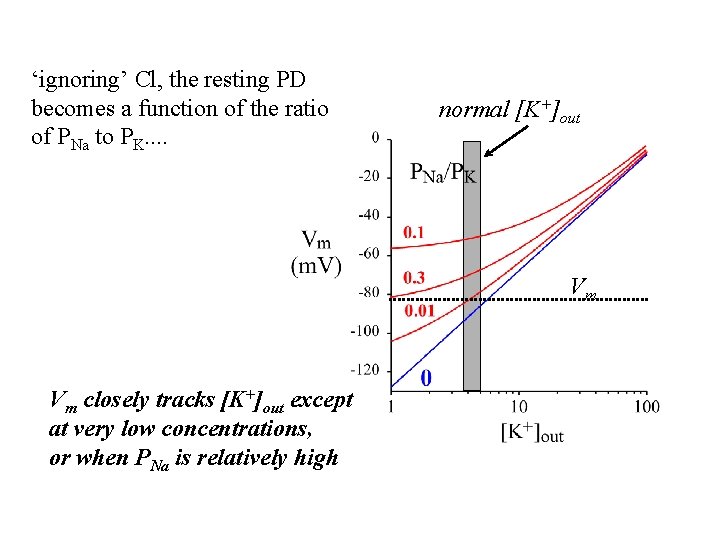
‘ignoring’ Cl, the resting PD becomes a function of the ratio of PNa to PK. . normal [K+]out Vm Vm closely tracks [K+]out except at very low concentrations, or when PNa is relatively high

In Conclusion. . . A. Active transport establishes K+ and Na+ gradients these tend to be stable B. Electrical Potential Difference is a consequence of these gradients and the relative permeability of the membrane to these ions C. At ‘rest’ the PD is dominated by the combined influence of: 1. the outwardly direct K+ gradient 2. large PK
- Slides: 18