Experiment Observations Conclusions Model DOUBLE SLIT EXPERIMENT Observations
Experiment: Observations: Conclusions: Model:

DOUBLE SLIT EXPERIMENT Observations: Experiment: Light passes through a double-slit experiment Conclusions: • Light it is making an interference pattern, similar to interference patterns made by sound waves and water waves. • Light must be a wave. • A diffraction pattern is made with bright spots and dark nodes. Model: • Light must be a wave. • c = λ×v • c = the speed of light = 3. 00 x 108 m/s • λ = wavelength (in m) • v = frequency (in s-1 or Hertz (Hz))

He emission spectrum Frequency (v) = Hertz (Hz) or cycles/s (S-1) Wavelength (λ) = m or nm Speed of light(c) = 3. 00 x 108 m/s or 1. Calculate the frequency of the light in the yellow 3. 00 x 1017 portion of this spectra. nm/s 2. How does the frequency of the yellow spectral line compare to the frequency of the blue-green spectral line?
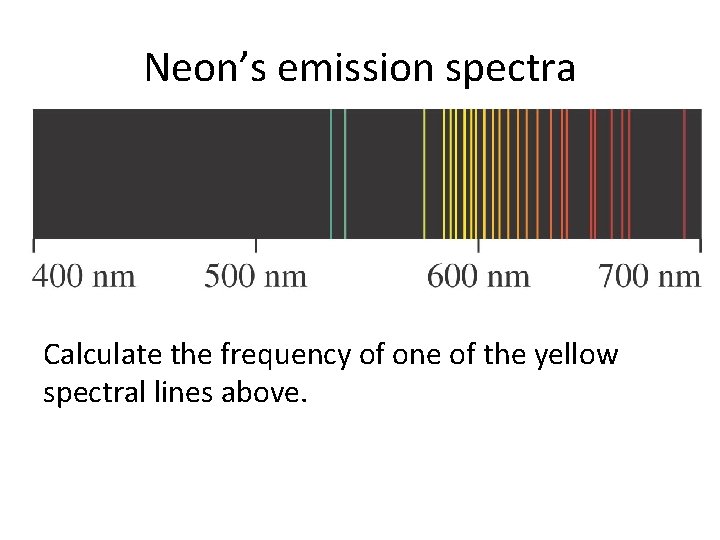
Neon’s emission spectra Calculate the frequency of one of the yellow spectral lines above.

PHOTOELECTRIC EFFECT Observations: Experiment: Light ejects electrons off of a piece of metal. Beginning Question: What variables affect the ejection of electrons? Conclusions: • Light must come in packets of ENERGY. • Changing wavelength does not have a direct relationship • Changing intensity does not necessarily have a relationship • Only wavelengths with a minimum frequency could eject electrons. Model: • Light acts like a particle, which we call a “photon. ” • Equation to solve for the energy of a wavelength is E = h × ν • E = Energy (in J) • h = Planck’s constant = 6. 626 x 10 -34 J·s • v = frequency (in Hz)

Each color of light corresponds to a specific wavelength… How does this relate to transitions within an atom? • A photon is a packet of energy released when an electron returns to ground state. E = hn (n = frequency) (h= Planck’s constant, 6. 626 x 10 -34 J/s 1) • This equation calculates the amount of energy emitted from the photon.
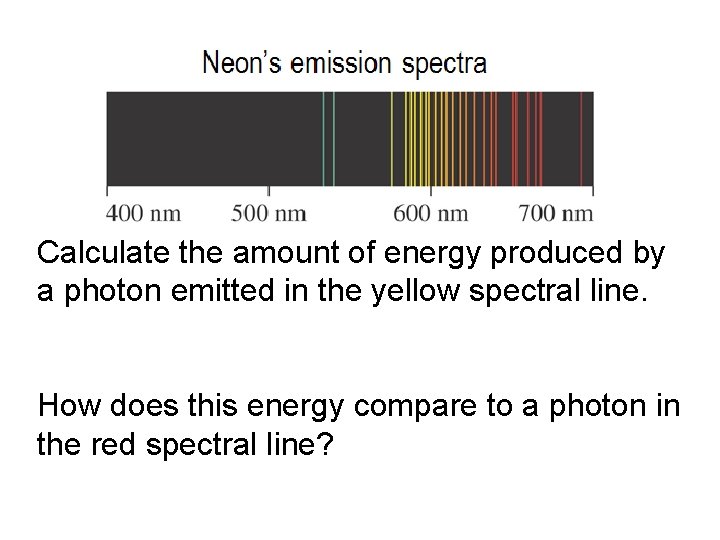
Calculate the amount of energy produced by a photon emitted in the yellow spectral line. How does this energy compare to a photon in the red spectral line?
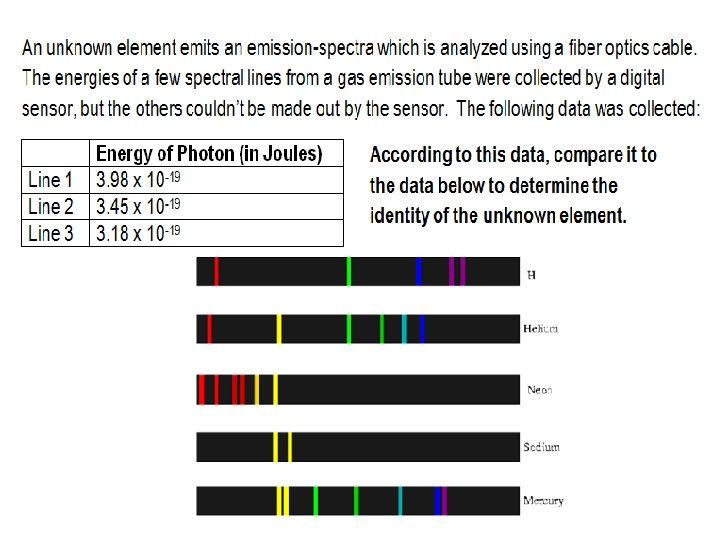
- Slides: 8