Experiment Objectives Preparing Staining and observing human metaphase


Experiment Objectives • Preparing • Staining and observing human metaphase chromosomes.

Chromosome Morphology • Chromosomes are not visible under the light microscope in non-dividing cells (interphase cells). • As the cell begins to divide, the threads of chromatin (DNA-protein complex) in the nucleus begin to condense into multiple levels of coiled structures recognizable as chromosomes. • There are two modes of cell division: • Mitosis: is responsible for the proliferation of body (somatic) cells, • Meiosis: is responsible for the production of gametes. • Because mitotic cells are easy to obtain, morphological studies are generally based on mitotic metaphase chromosomes.
Cell division • Cell division can be divided into: • Interphase • Mitosis üProphase üMetaphase üAnaphase üTelophase • Cytokinesis
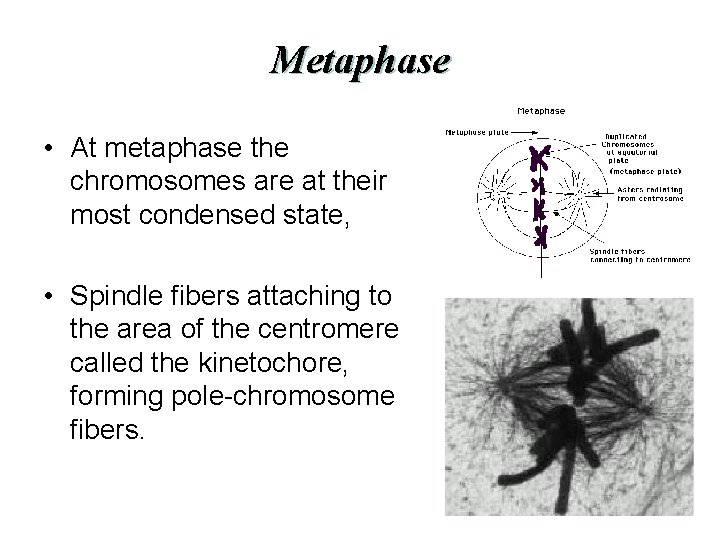
Metaphase • At metaphase the chromosomes are at their most condensed state, • Spindle fibers attaching to the area of the centromere called the kinetochore, forming pole-chromosome fibers.

• Anaphase begins with the division of the centromere and the separation of chromatids. • Once separated, each chromatid is known as a chromosome. • The kinetochore: is the protein structure on chromatids where the spindle fibers attach during cell division to pull sister chromatids apart.

Chromosome Analysis • The best mitotic stage for chromosome analysis is prometaphase or metaphase. • A typical metaphase chromosome consists of two arms separated by a primary constriction or centromere. • Each of the two sister-chromatids contains a highly coiled double helix of DNA. • Often the sister chromatids are so close to each other that the whole chromosome appears as a single rod-like structure • A chromosome may be characterized by its total length and the position of its centromere.
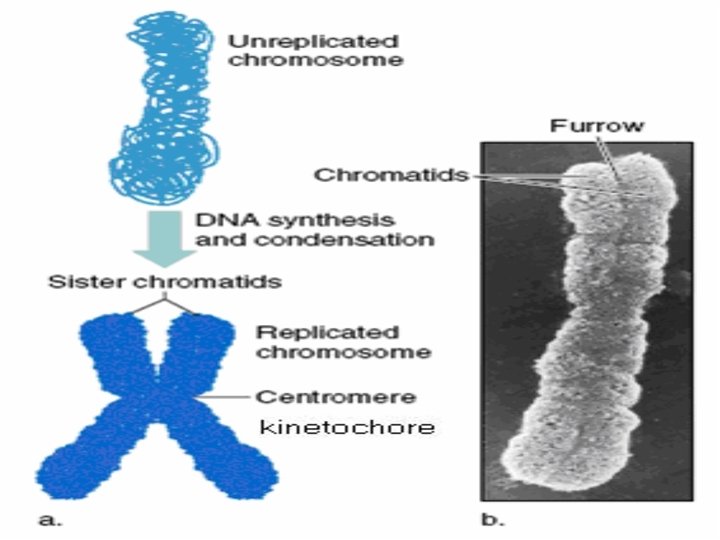
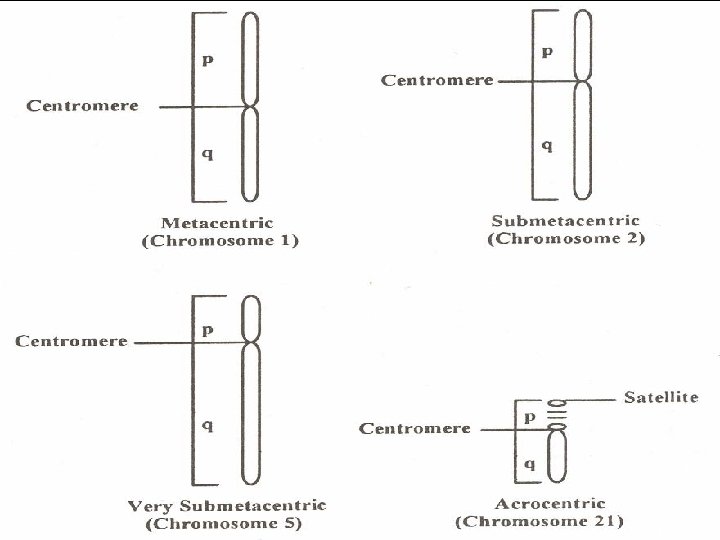

Chromosome Number • The diploid chromosome number is the number of chromosomes in the somatic cell and is designated by the symbol 2 N. • The gametes, which have one half the diploid number, have the haploid number N. • Thus, there are 23 pairs of chromosomes in human cells.

• Of these, 22 pairs are not directly involved in sex determination, and are known as autosomes. • The remaining chromosome pair consists of the sex chromosomes, and is directly involved in sex determination. • In females the two sex chromosom es are identical (XX), whereas in males the two sex chromosomes are not identical (XY).

Types of Tissue • A variety of tissue types can be used to obtain chromosome preparations. • Some examples include peripheral blood, bone marrow, amniotic fluid and products of conception. • In the case of blood cell culture only cells that are actively dividing can be used for cytogenetic studies. • Normally only white blood cells are used for cytogenetic analysis. • Specific techniques differ according to the type of tissue used.

Overview of Procedure 1. Collection of blood 2. Cell culture 3. Harvesting: stopping the cell division at metaphase 4. Hypotonic treatment of red & white blood cells 5. Fixation 6. Slide preparation 7. Staining
1 - Collection of blood • Draw 5 ml of venous blood into a sterile heparinized tube containing 0. 1 ml of sodium heparin (500 units/ml).

2 - Cell Culture • Sterile technique must be used throughout the cell culture preparation, because it is possible to cause major contamination during this procedure, • 70% of the problems are due to a lack of good sterile technique. • Antibiotics do not eliminate problems of gross contamination which result from poor sterile technique or antibiotic-resistant mutants. • Autoclaving renders pipettes, glassware, and solutions sterile.

2 - Cell Culture Medium • Pipette 10 ml RPMI 1640 medium with LGlutamine into a 15 ml labeled sterile culture tube • Supplement the medium with the following: Penicillin-Streptomycin Stock solution 10 µl (100000 u penicillin/ml – 100 mg/ml Streptomycin Phytohemagglutinin 0. 3 ml 20 µg/ml Fetal bovine Serum 20% 2 ml
2 - Cell Culture Incubation • Add 1 ml of whole heparinized blood into the tube containing the supplemented medium • Mix contents of tube with gentle inversion • Incubate in 5% CO 2 incubator at 37 o. C for 72 hours

3 - Harvesting • Harvesting: mitotic spindle formation is blocked: usually by adding colcemide to the culture, and the cell division is stopped at the metaphase level. • Pre-warm the Colchicine (0. 04 mg/ml) in incubator at 37 o. C. • Add 25 µl of pre-warmed Colchicine to the culture. • Mix gently and incubate at 37 o. C for 30 -60 minutes. • Note: Colchicine inhibits microtubule polymerization by binding to tubulin, one of the main constituents of microtubules

4 - Hypotonic treatment of red & white blood cells • Centrifuge for 10 minutes at 2000 rpm. • Discard supernatant without disturbing the cells leaving 0. 5 ml of fluid. • Add 1 ml of pre-warmed hypotonic solution (0. 075 M KCl) at 37 o. C. • Mix and then add 9 ml of hypotonic solution. • Mix well by Pasteur pipette. o • Incubate at 37 C incubator for 17 minutes, • hypotonic solution should not be in contact with cells more than 27 minutes (may cause rupture of WBCs).

5 - Fixation • Fixative must be prepared fresh • Add 3 parts of chilled absolute methanol: 1 part glacial acetic acid. • Centrifuge for 10 minutes at 1000 – 1500 rpm. • Remove supernatant leaving about 0. 5 ml of fluid on top of cells. • At this time there is probably a small whitish or reddish film at the bottom of the tube. • The film contain red blood cell debris and enlarged WBCs.

5 - Fixation • • • Add 5 ml of fixative to the tube. Mix with a Pasteur pipette 3 -4 times. Place in refrigerator for 30 minutes. Centrifuge the tube for 10 minutes at 1000 -1500 rpm. Remove supernatant and add another 6 ml of cold fixative, & mix well. Centrifuge the tube for 10 minutes at 1000 -1500 rpm. Repeat the last two steps. Remove the supernatant leaving 1 ml of fluid at the bottom. The remaining material will be used to make the slides.

6 - Slides Preparation • The slide must be exceptionally clean • Lay slides on a paper towel • Withdraw a few drops of cell suspension into a pipette • From a height of 20 cm, drop 2 or 3 drops of fluid on each slide • Allow the slides to dry

7 - Staining • Stain the slides by immersion in fresh Giemsa stain for 7 -10 minutes • Remove slides from stain & rinse in distilled water • Observe under microscope 40 X then under oil immersion
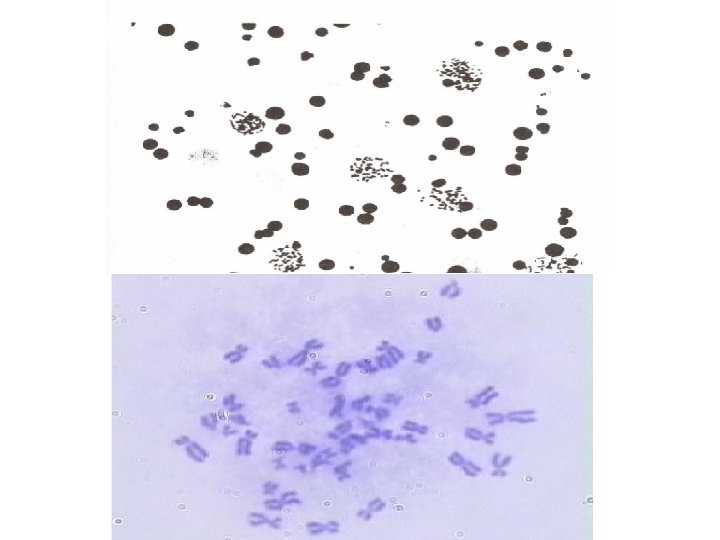
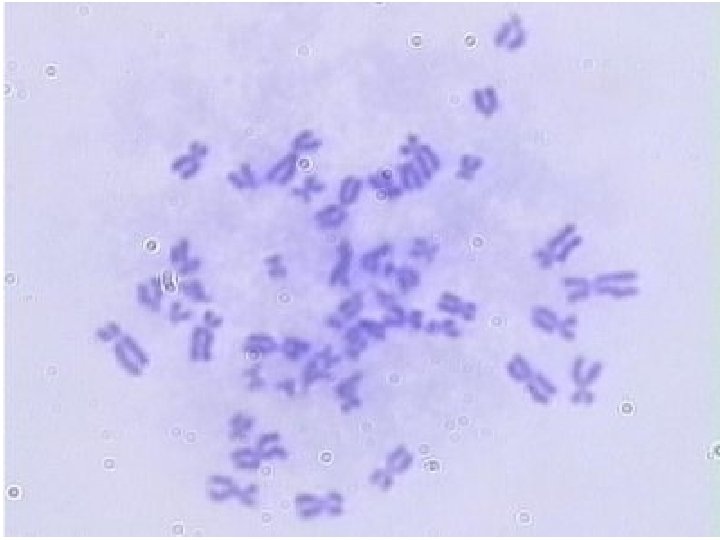

• http: //www. biology. arizona. edu/human_bio/activities /karyotyping/patient_a. html • http: //www. youtube. com/watch? v=E 0 Wk. Zr 819 UU
- Slides: 26