EXPERIMENT 5 Effect of Temperature on Solubility of

EXPERIMENT (5) Effect of Temperature on Solubility of a Salt

Aim of The Experiment: Study the effect of changing temperature on the amount of solute that will dissolve in a given amount of water For Example: In this experiment, you will completely dissolve different quantities of potassium nitrate, KNO 3, in the same volume of water at a high temperature.

Theory: water solubility: is an important physical property in chemistry, expressed as the mass of solute that dissolve in 100 g of H 2 O at a certain temperature.

Procedure: 1. Label four test tubes 1 -4. Test tube number Amount of KNO 3 used per 5 ml H 2 O Amount of KNO 3 used per 100 g H 2 O 1 2 40 2 4 80 3 6 120 4 8 160 2. Add 5 ml of distilled water to each test tube. 3. Fill a 250 ml beaker with tap water and heat the water bath to about 90 C. 4. Lower the test tube into the water bath and stir the mixture until the KNO 3 is completely dissolved ( HINT: test tubes 3 & 4 need to be heated to a higher temperature than test tubes 1& 2) 5. As each solution cools you will monitor temperature when solid crystals start to form ( T ). 6. Repeat steps 4 & 5 for each of the test tube.
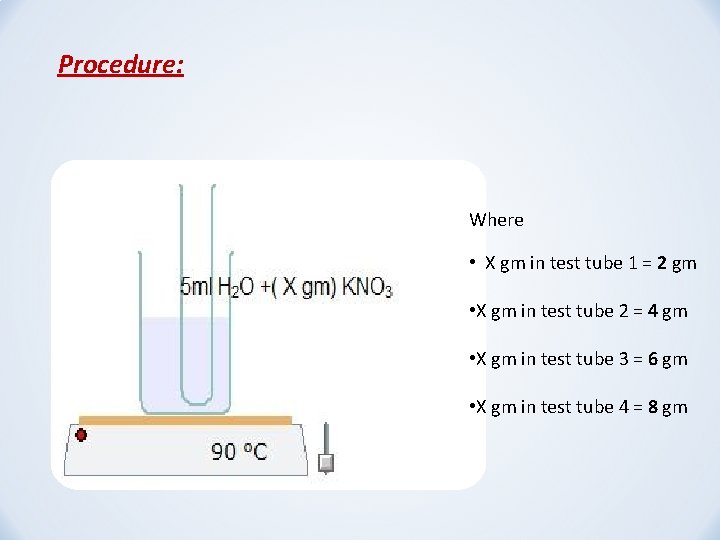
Procedure: Where • X gm in test tube 1 = 2 gm • X gm in test tube 2 = 4 gm • X gm in test tube 3 = 6 gm • X gm in test tube 4 = 8 gm
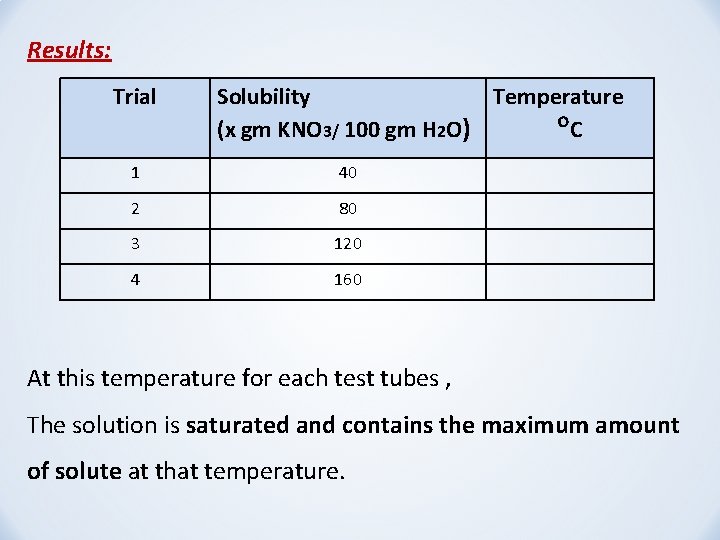
Results: Trial Solubility Temperature (x gm KNO 3/ 100 gm H 2 O) ᴼC 1 40 2 80 3 120 4 160 At this temperature for each test tubes , The solution is saturated and contains the maximum amount of solute at that temperature.
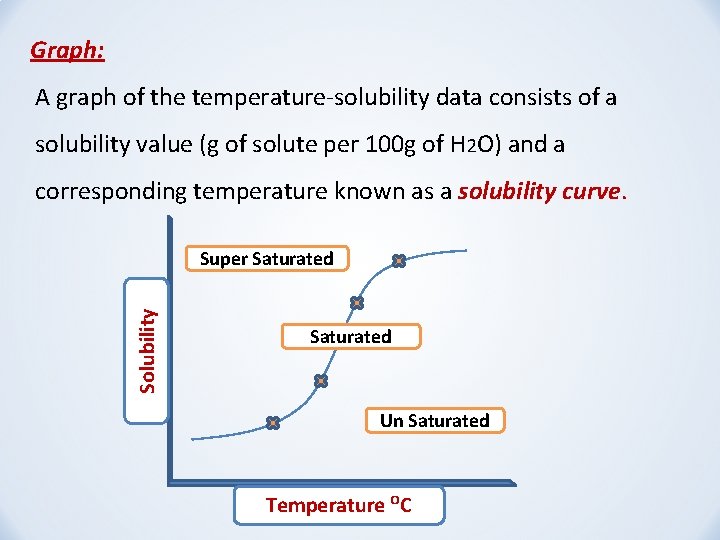
Graph: A graph of the temperature-solubility data consists of a solubility value (g of solute per 100 g of H 2 O) and a corresponding temperature known as a solubility curve. Solubility Super Saturated Un Saturated Temperature ᴼC
- Slides: 7