Experiment 14 Enthalpy of Hydration Purposes and Goals

















- Slides: 20

Experiment 14 Enthalpy of Hydration

Purposes and Goals • The purpose of this experiment is to use Hess’s Law to determine the enthalpy change (ΔH) for the hydration of an anhydrous compound.

Introduction • Conservation of energy • Hess’s Law of Heat Summation • Alternative view of Hess’s Law: ΔH does not depend on how I get from reactants to products.

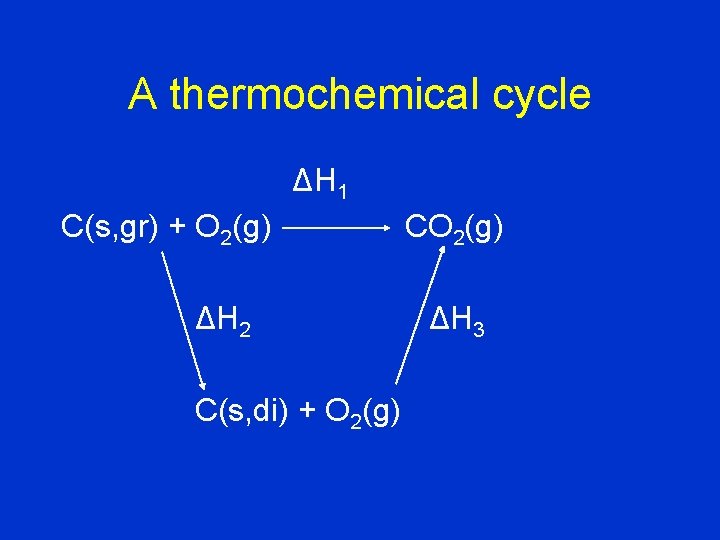
I can use this idea to determine ΔH for a reaction which is hard to study directly. Example: graphite diamond C(s, gr) C(s, di)

A thermochemical cycle ΔH 1 C(s, gr) + O 2(g) ΔH 2 C(s, di) + O 2(g) CO 2(g) ΔH 3

• DH 1 = DH 2 + DH 3 • The direct conversion of graphite to diamond (corresponding to DH 2) is hard to measure, but the two combustions (DH 1 and DH 3) are easy to measure. We can determine DH 2 indirectly from DH 1 and DH 3. • DH 2 = DH 1 - DH 3

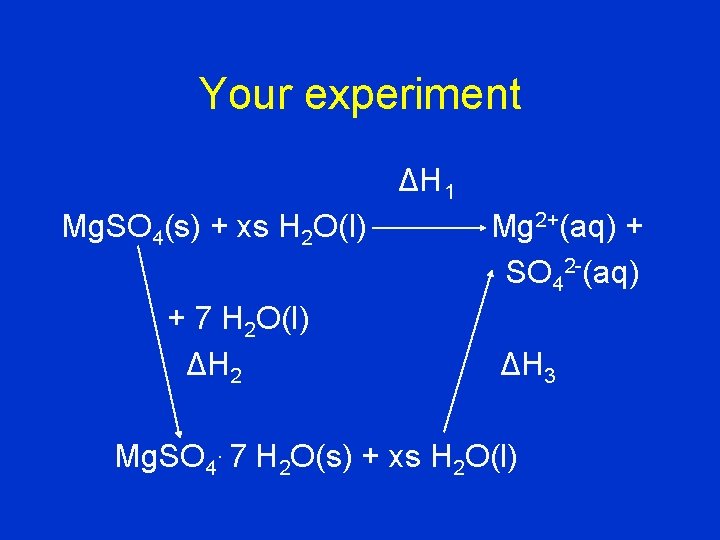
Your experiment ΔH 1 Mg. SO 4(s) + xs H 2 O(l) + 7 H 2 O(l) ΔH 2 Mg 2+(aq) + SO 42 -(aq) ΔH 3 Mg. SO 4. 7 H 2 O(s) + xs H 2 O(l)

Calorimetry • Calorimetry---measuring heat flow • Fundamental idea: Qreaction = -Qsolution • Qsolution = C ΔT • C = (mass of solution) x (specific heat of the solution)

Safety • Aprons and glasses • Thermometers are fragile and expensive; don’t use a thermometer as a stirring rod!

Procedure • Work in pairs. • Needed equipment: 100 -m. L graduated cylinder, stirring rods, thermometers, 2 thermometer clamps, 2 calorimeter cups, ring stand, ring. • Same setup as Expt. 6 (CHEM 1031 lab).

• One run with Mg. SO 4, two with hydrate. • Weigh empty calorimeter cup. Record mass on data sheet. • Weigh solute (about 7. 50 g of Mg. SO 4, 15. 35 g of hydrate). Idea: equal moles of solute in samples.

• Add 100 m. L water to cup; measure temperature every 30 seconds for five minutes. • Add Mg. SO 4; use stirring rod (NOT THERMOMETER) to dissolve solute quickly. • Measure temperature every minute for fifteen minutes. After run is over, measure and record mass of cup plus solution.

• Weigh second calorimeter cup; add water and measure temperature for five minutes as before. • After 5 minutes, add Mg. SO 4. 7 H 2 O. Measure temperature for 15 minutes as before. Measure and record mass of cup and solution.

• Discard solution (sink); add 100 m. L of water and repeat run. After 5 minutes, add second portion of Mg. SO 4. 7 H 2 O. Measure temperature for 15 minutes as before. Measure and record mass of cup plus solution; you will use the same mass of empty cup for both runs.

Calculations • Draw a graph of temperature vs time for your experimental data. Each run is a separate graph. • Extrapolate the temperature lines before and after adding solute to the time at which solute was added.



• DT = Tfinal – Tinitial • C = mass of solution x specific heat of solution (specific heat of solution = 3. 837 J g-1 deg-1) • Qreaction = - C DT • (Good model: p. 14 -3 of lab manual!)

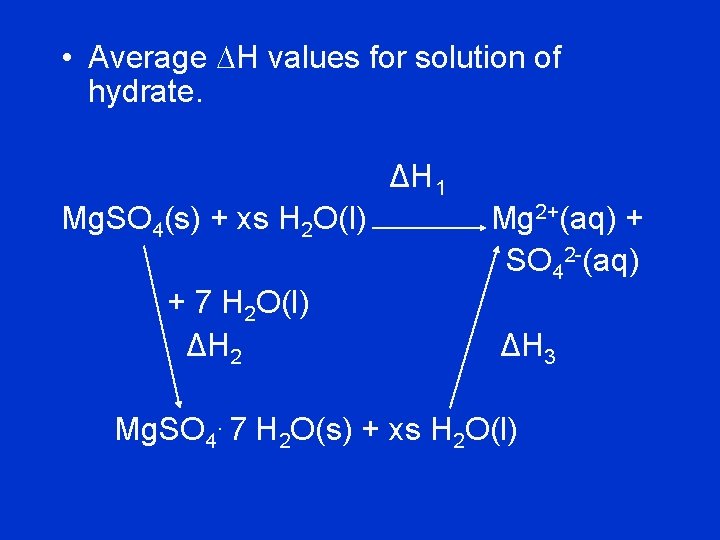
• Average DH values for solution of hydrate. ΔH 1 Mg. SO 4(s) + xs H 2 O(l) + 7 H 2 O(l) ΔH 2 Mg 2+(aq) + SO 42 -(aq) ΔH 3 Mg. SO 4. 7 H 2 O(s) + xs H 2 O(l)

• You have calculated the heats of solution of Mg. SO 4 (DH 1 ) and of Mg. SO 4. 7 H 2 O (DH 3). • The enthalpy of hydration (DH 2) is DH 2 = DH 1 - DH 3.