Expanding HLA Typing Requirements Resolution 10 Histocompatibility Committee

Expanding HLA Typing Requirements (Resolution 10) Histocompatibility Committee Dolly Tyan, Ph. D Chair

Strategic Plan #6 - Promote the Efficient Management of the OPTN • Prevent unexpected positive crossmatches that result in discards or increased cold ischemia time

The Problem § New KAS takes effect in December § Candidates with CPRA greater than 98% will receive regional and national priority before all other kidney candidates § Literature shows a significant number of sensitized candidates have antibodies to HLA-DQA/DPB § There are currently no fields in Donor. Net® to report these HLA types on donors or in Waitlist℠ as unacceptable antigens for candidates § Without this information, there is a high risk for discards and increased CIT in KAS
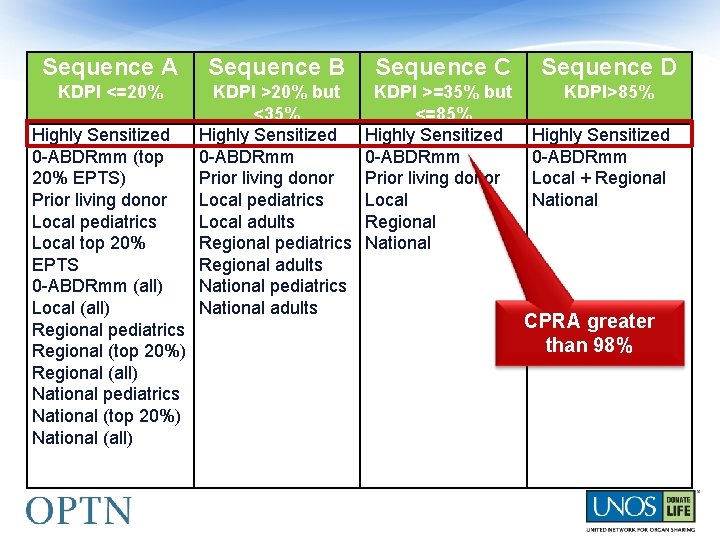
Sequence A Sequence B Sequence C Sequence D KDPI <=20% KDPI >20% but <35% Highly Sensitized 0 -ABDRmm Prior living donor Local pediatrics Local adults Regional pediatrics Regional adults National pediatrics National adults KDPI >=35% but <=85% Highly Sensitized 0 -ABDRmm Prior living donor Local Regional National KDPI>85% Highly Sensitized 0 -ABDRmm (top 20% EPTS) Prior living donor Local pediatrics Local top 20% EPTS 0 -ABDRmm (all) Local (all) Regional pediatrics Regional (top 20%) Regional (all) National pediatrics National (top 20%) National (all) Highly Sensitized 0 -ABDRmm Local + Regional National CPRA greater than 98%

Other Problems Policy has inconsistent HLA reporting requirements for deceased donors (DD) across organ types Molecular typing only required for deceased KI, K-P, and PA donors No HLA typing requirements for deceased islet donors or candidates

Proposed Changes § HLA DQA and –DPB reported when typing is performed on DD. Program DQA and DPB fields in Donor. Net® and Waitlist® as unacceptable antigens § Require consistent and complete list of loci reported on DD § Require molecular methods when performing HLA typing on DD § Align policy requirements for deceased islet donors and candidates with those of pancreas donors and candidates

Overall Project Impact Product Target Population Impact: Policies 2. 11. A - 2. 11. E, 3. 4. D, and 4. 2 All deceased donors, candidates, and recipients. Special emphasis for highly sensitized candidates and all kidney candidates. Total IT Implementation Hours 1, 500/10, 680 Total Overall Implementation Hours 2, 160/17, 885

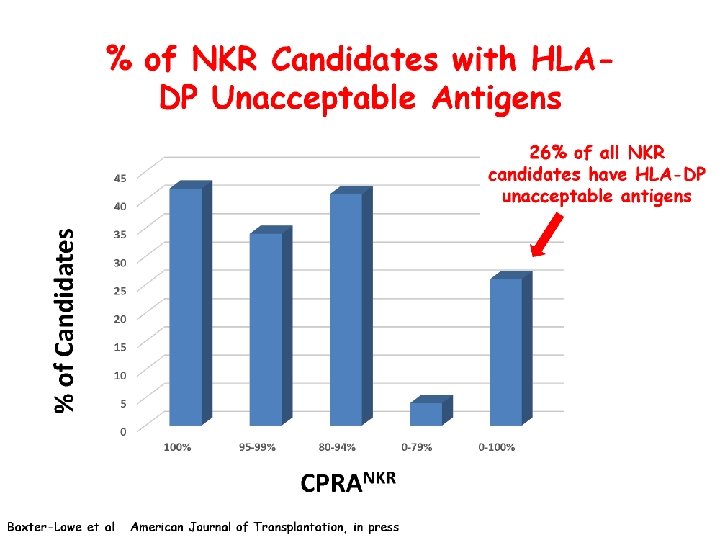
Frequency of DQA/DPB UAs Kidney WL data • 10, 556 (10%) have 98%+ CPRA • 2, 351 have 99% and 6, 900 have 100% CPRA Bray et al, AJT, in press • Data for 6 labs/10 tx centers • 98%+ CPRA candidates (N=593): 63% had DQA/DPB UAs
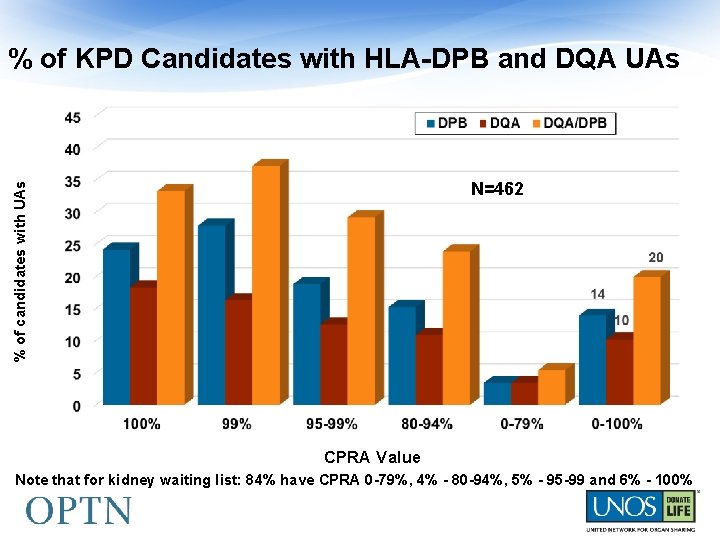
% of KPD Candidates with HLA-DPB and DQA UAs % of candidates with UAs N=462 CPRA Value Note that for kidney waiting list: 84% have CPRA 0 -79%, 4% - 80 -94%, 5% - 95 -99 and 6% - 100%

Two Programming Options #1 • Donor. Net® Only #2 • Donor. Net® and Waitlist℠

Pro: • Allows deceased donor HLA info to be reported for these types • Slight reduction in IT programming estimate (still in large category) Con: • Patient safety concerns • Burdensome for transplant programs • Inconsistent with programming for all other HLA reporting and the KPD program • Committees expressed patient safety concerns

Pro: • Eliminates patient safety concerns • Less burdensome to transplant programs • Consistent with KPD programming • Supported by community (based on feedback from regions, Kidney Committee, OPO Committee, Pancreas Committee, Operations and Safety Committee, and ASHI) Con: • Higher IT programming cost (very large category)
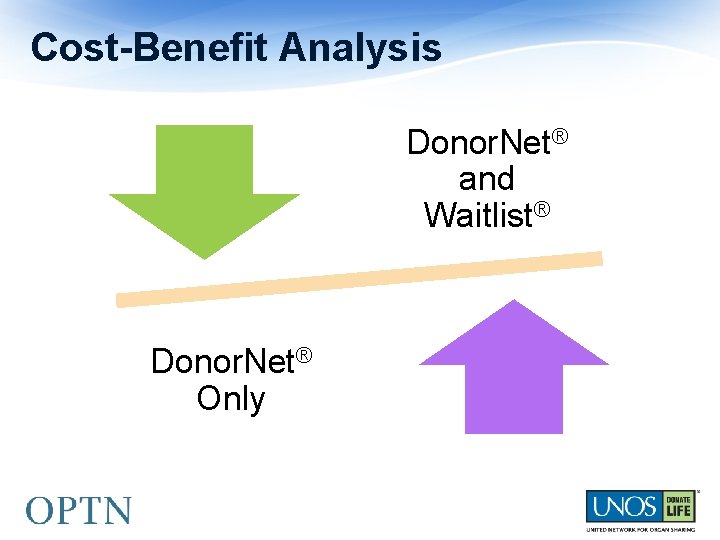
Cost-Benefit Analysis Donor. Net® and Waitlist® Donor. Net® Only

Public Comment/Professional Society Feedback Regions Committees Individual • Unanimous Support • Feedback from 6 • Unanimous Support Professional Societies • ASTS, AST, ASHI, CAP • Unanimous Support

RESOLUTION 20 The Board is asked to approved additions and changes to Policies 2. 11. A (Required information for Deceased Kidney Donors); 2. 11. B (Required information for Deceased Liver Donors); 2. 11. C (Required Information for Deceased Heart Donors); 2. 11. D (Required Information for Deceased Lung Donors); 2. 11. E (Required Information for Deceased Pancreas Donors); 3. 4. D (Candidate Human Leukocyte Antigen (HLA Information); and 4. 2 (Requirements for Performing and Reporting HLA Typing) to provide greater consistency in HLA typing requirements across organ types. *Page 74 of Board book

Extra Slides

Background Fall 2012 • Committee noticed inconsistencies in HLA typing requirements Summer 2012 • BOD did not approve original request to program DQA and DPB Dec. 2014 • Policy changes recommended • Outreach to organ specific committees Spring 2014 • Approved for public comment • Update presented to BOD (June 2014)
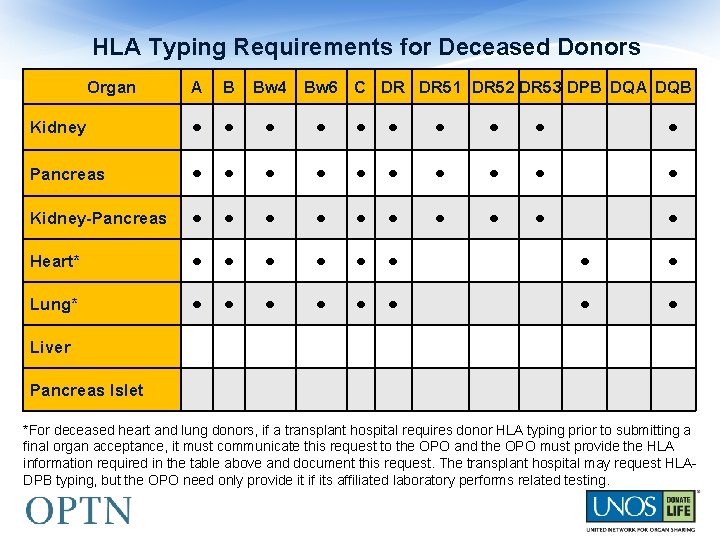
HLA Typing Requirements for Deceased Donors Organ A B Bw 4 Bw 6 C DR DR 51 DR 52 DR 53 DPB DQA DQB Kidney • • • Pancreas • • • Kidney-Pancreas • • • Heart* • • • Lung* • • • Liver Pancreas Islet *For deceased heart and lung donors, if a transplant hospital requires donor HLA typing prior to submitting a final organ acceptance, it must communicate this request to the OPO and the OPO must provide the HLA information required in the table above and document this request. The transplant hospital may request HLADPB typing, but the OPO need only provide it if its affiliated laboratory performs related testing.
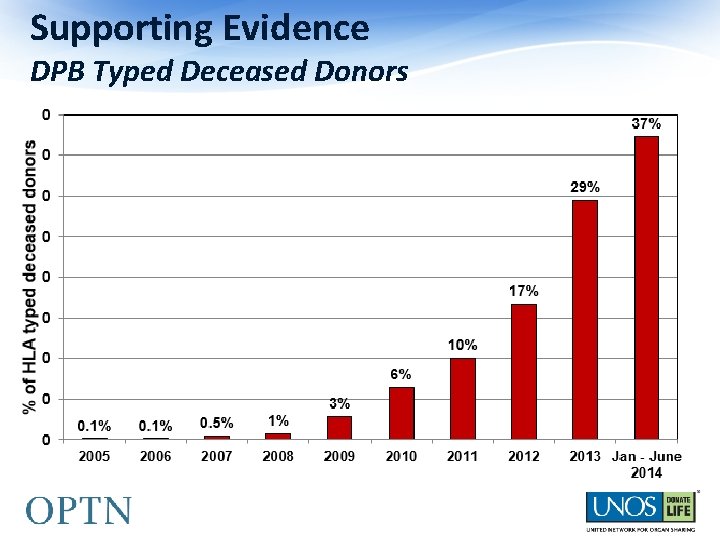
Supporting Evidence DPB Typed Deceased Donors
- Slides: 20