Exothermic and Endothermic Reactions Teachable Some rights reserved

Exothermic and Endothermic Reactions © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910

Endothermic and exothermic reactions Step 1: Energy must be SUPPLIED to break chemical bonds: Step 2: Energy is RELEASED when new chemical bonds are made: A reaction is EXOTHERMIC if more energy is RELEASED than SUPPLIED. If more energy is SUPPLIED than is RELEASED then the reaction is ENDOTHERMIC © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910
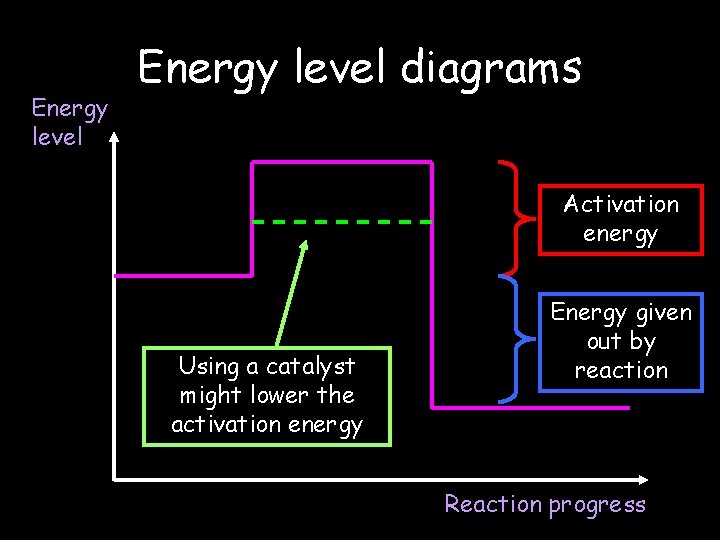
Energy level diagrams Activation energy Using a catalyst might lower the activation energy Energy given out by reaction Reaction progress © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910

Endothermic process: a change (e. g. a chemical reaction) that requires (or absorbs) heat. Photosynthesis is an endothermic reaction (requires energy input from sun) Forming Na+ and Cl- ions from Na. Cl is an endothermic process
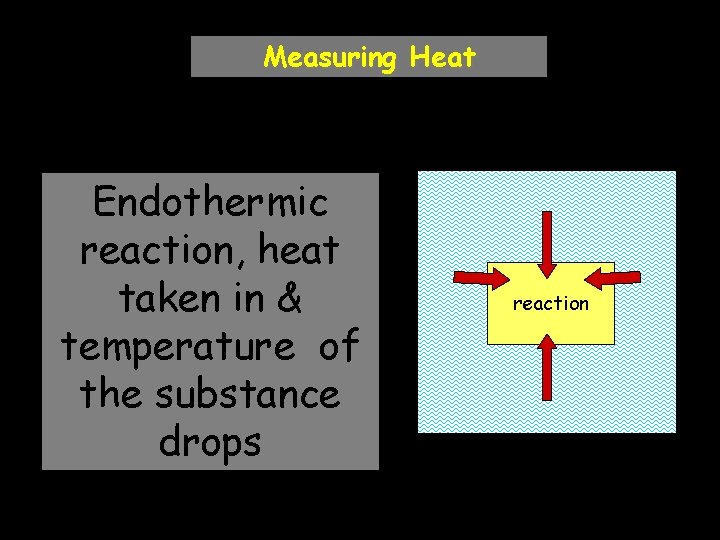
Measuring Heat Endothermic reaction, heat taken in & temperature of the substance drops reaction

EXOTHERMIC & ENDOTHERMIC REACTIONS Exothermic process: a change (e. g. a chemical reaction) that releases heat. Burning fossil fuels is an exothermic reaction
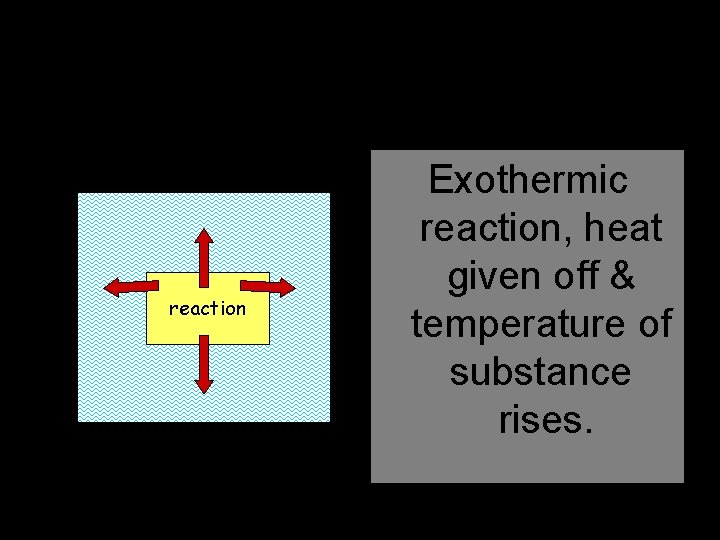
Heat Released reaction Exothermic reaction, heat given off & temperature of substance rises.
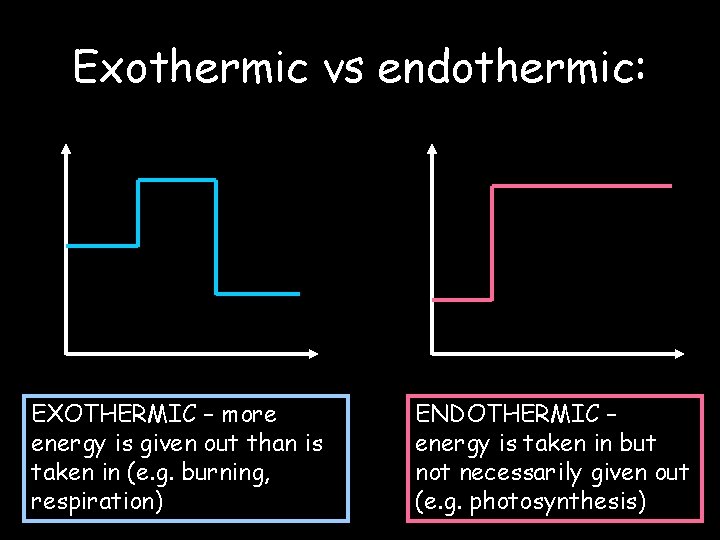
Exothermic vs endothermic: EXOTHERMIC – more energy is given out than is taken in (e. g. burning, respiration) © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910 ENDOTHERMIC – energy is taken in but not necessarily given out (e. g. photosynthesis)

Examples Exothermic • • • Combustion of fuels Epson salts & water Freezing Water Burning sugar Hand warmers © Teachable. Some rights reserved. http: //teachable. net/res. asp? r=1910 Endothermic • • • Photosynthesis Ice packs Melting ice Baking bread Cooking an egg
- Slides: 9