Exercise 6 Electrowinning Sipi Seisko Benjamin Wilson CHEME

Exercise 6: Electrowinning Sipi Seisko & Benjamin Wilson CHEM-E 6180 Product recovery exercise

Exercise 3, Product recovery with electrowinning • Electrowinning of product solution (from solution purification) in order to recover Cu • Simulation of electrowinning process with HSC 8. 0. http: //www. redtigermining. com/s/Photo. Gallery. asp http: //www. tankhousetechnology. co. za/ http: //www. palabora. com/copper. asp 13. 6. 2021 2

Electrowinning of product solution • Electowinning and smelting are the final process stages in the copper production • Copper ions from concentrated Cu solution are turned into solid copper • Electric current is passed through the conductive electrolyte and causes the precipitation of copper on cathode • Electrons pass from cathode (negative) to anode (positive) since external power supply is used 13. 6. 2021 3

Electrowinning of product solution Electowinning system includes cell, anode, cathode and conductive electrolyte solution • As a cathode: copper (thin) or stainless steel plate • As an anode: lead (alloy) plate • As a electricity conductor: Cu concentrated solution http: //www. weiku. com/products/12070134/Pb_Lead_Alloy_Rolled_Anode_For_Nonferrous_Electrowinning. html Laitinen, I. S. , 2009. Modelling, simulation and optimization of a copper electrolysis cell group. Doctoral thesis, TKY 13. 6. 2021 4
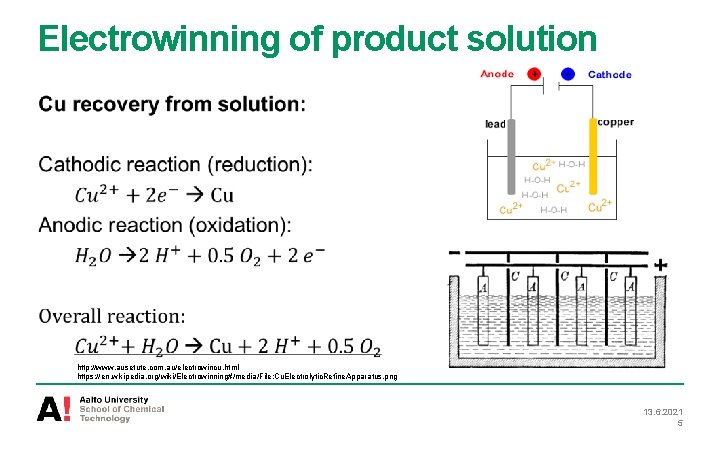
Electrowinning of product solution http: //www. ausetute. com. au/electrowincu. html https: //en. wikipedia. org/wiki/Electrowinning#/media/File: Cu. Electrolytic. Refine. Apparatus. png 13. 6. 2021 5
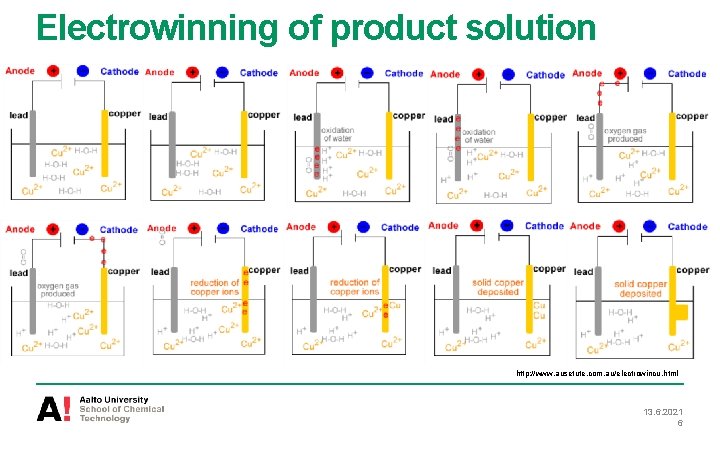
Electrowinning of product solution http: //www. ausetute. com. au/electrowincu. html 13. 6. 2021 6

Objective http: //www. redtigermining. com/s/Photo. Gallery. asp 13. 6. 2021 7

Calculations

Important factors for the process Specific factors • Cell current density • Cell voltage • Energy consumption • Copper production • Cell Scale • Plant Scale 13. 6. 2021 9

Which factors you can get from tests • Current efficiency • Cell voltage specific energy consumption • How material of anode affects the cell voltage • Quality of copper • Analysis required 13. 6. 2021 10

Cell current density (1/2) 13. 6. 2021 11

Cell current density (2/2) 13. 6. 2021 12

Calculation of cell voltage (1/3) 13. 6. 2021 13

Calculation of cell voltage (2/3) 13. 6. 2021 14

Calculation of cell voltage (3/3) 13. 6. 2021 15

Factors affecting to the cell voltage Affecting factors and their average share: • • Resistivity of electrolyte (50%) Polarization of cathode (25%) Polarization of anode (19%) Drop in voltage because of connections (6%) = usually 10% resistivity of electrolyte 13. 6. 2021 16

Potential drop 13. 6. 2021 17

Polarization of electrode (overpotential) (1/3) 13. 6. 2021 18

Polarization of electrode (overpotential) (2/3) 13. 6. 2021 19

Polarization of electrode (overpotential) (3/3) 13. 6. 2021 20

Calculation of copper production 13. 6. 2021 21

Factors affecting to the current efficiency Examples about factors that reduce the current efficiency: • Dissolution of copper from cathode back to the solution • Leakage current • Short circuit between anode and cathode 13. 6. 2021 22

Current efficiency 13. 6. 2021 23

Calculation of energy consumption 13. 6. 2021 24
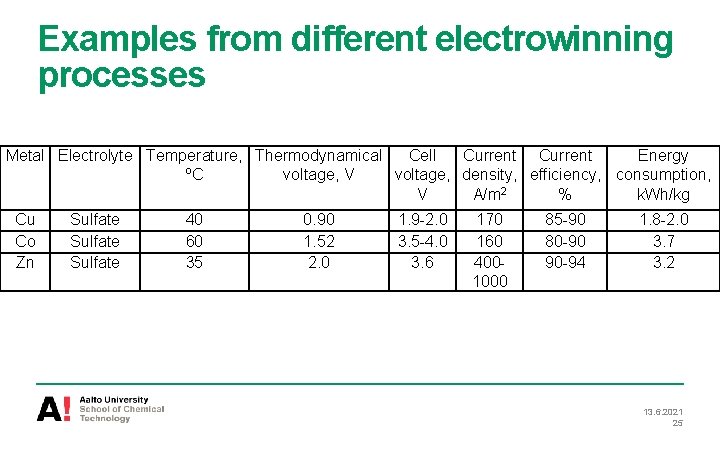
Examples from different electrowinning processes Metal Electrolyte Temperature, Thermodynamical Cell Current Energy ºC voltage, V voltage, density, efficiency, consumption, V A/m 2 % k. Wh/kg Cu Co Zn Sulfate 40 60 35 0. 90 1. 52 2. 0 1. 9 -2. 0 3. 5 -4. 0 3. 6 170 160 4001000 85 -90 80 -90 90 -94 1. 8 -2. 0 3. 7 3. 2 13. 6. 2021 25
- Slides: 25