Exercise 5 Monte Carlo simulations Bioequivalence and Withdrawal

Exercise 5 Monte Carlo simulations, Bioequivalence and Withdrawal time 1

Objectives of the exercise • To understand regulatory definitions of Bioequivalence and Withdrawal time • To understand the principles and goals of Monte Carlo simulation (MCS) • To simulate a data set using MCS with Crystal Ball (CB) to show that two formulations of a drug (pioneer and generic) can be bioequivalent while having different withdrawal times. • To compute a Withdrawal time using the EMEA software (WT 1. 4 by P Heckman) • To compute a Bioequivalence using WNL (crossover design) 2

Origin of the question 3

A regulatory decision • The new EMEA guideline on bioequivalence (draft 2010) states that it is possible to get a marketing authorization for a new generic without having to consolidate the withdrawal time associated with the pioneer formulation except if there are tissular residues at the site of injection. 4

The EMA guideline 5

Comments on guideline by EMA 6

The question • As an expert, you have to express your opinion on this regulatory decision. • As a kineticist you know that the statistical definitions of Withdrawal time (WT) and bioequivalence (BE) are fundamentally different • So, you decide to demonstrate, with a counterexample, it is not true and for that you have to build a data set corresponding to a virtual trial for which BE exist while WT are different • For that you will use MCS 7

EMA definition of BE (2010) • For two products, pharmacokinetic equivalence (i. e. bioequivalence) is established if the rate and extent of absorption of the active substance investigated under identical and appropriate experimental conditions only differ within acceptable predefined limits. • Rate and extent of absorption are estimated by Cmax (peak concentration) and AUC (total exposure over time), respectively, in plasma. 8

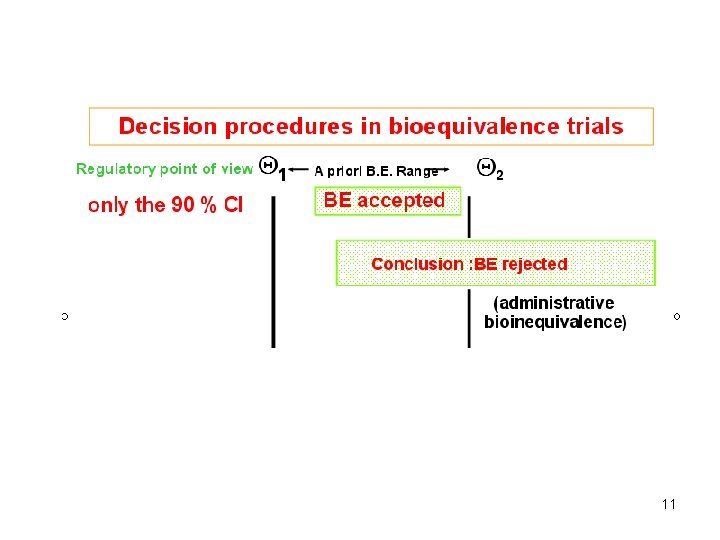
EMA definition of BE • The EMA consider the ratio of the population geometric means (test/reference) for the parameters (Cmax or AUC) under consideration. • For AUC, the 90% confidence intervals for the ratio should be entirely contained within the a priori regulatory limits 80% to 125%. • For Cmax, the a priori regulatory limits 70% to 143% could in rare cases be acceptable 9
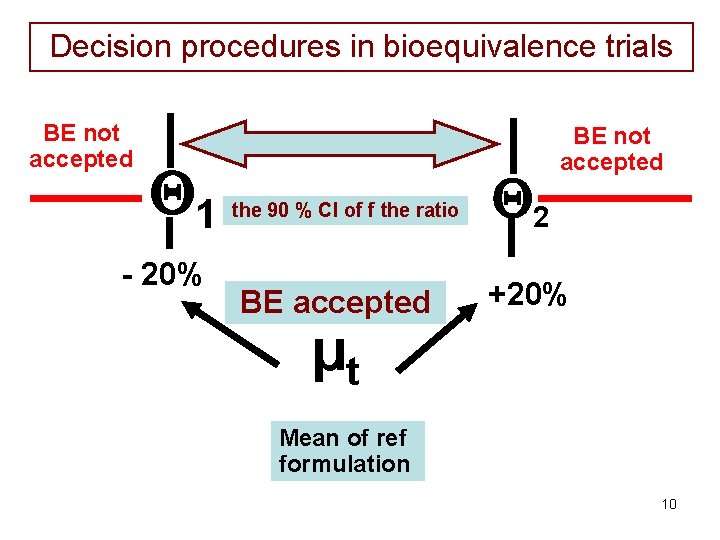
Decision procedures in bioequivalence trials BE not accepted 1 - 20% the 90 % CI of f the ratio BE accepted 2 BE not accepted +20% µt Mean of ref formulation 10
11

The a priori BE interval: why 80 -125% • The reference value is 100 thus After a logarithmic transformation: This interval is no longer symmetric around the reference value 12

The a priori BE interval: why 80 -125% After a logarithmic transformation: This interval is no longer symmetric around the reference value This interval is now symmetric around the reference value in the Ln domain 13

Different types of bioequivalence • Average (ABE) : mean • Population (PBE) : prescriptability 14
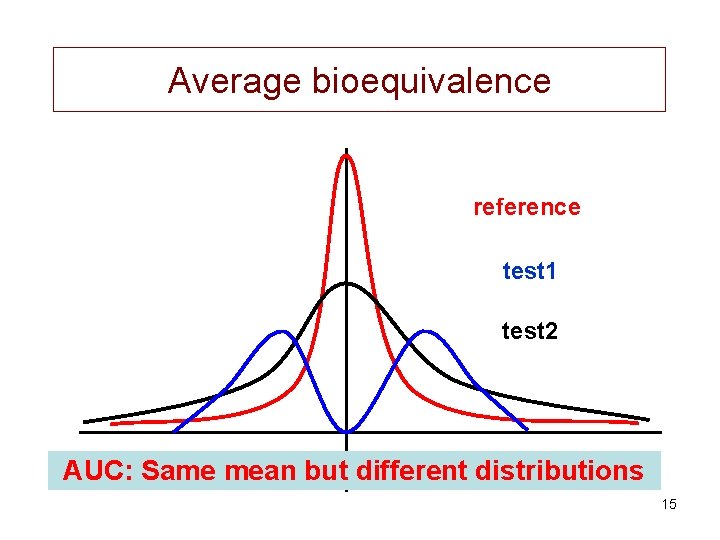
Average bioequivalence reference test 1 test 2 AUC: Same mean but different distributions 15
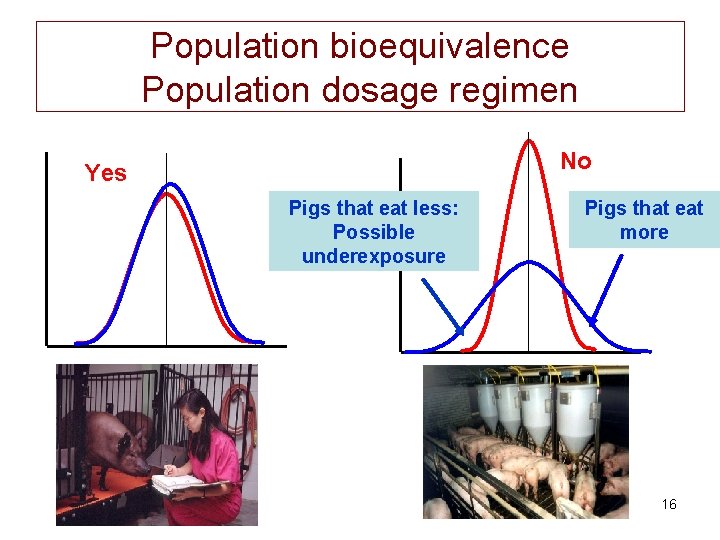
Population bioequivalence Population dosage regimen No Yes Pigs that eat less: Possible underexposure Pigs that eat more 16
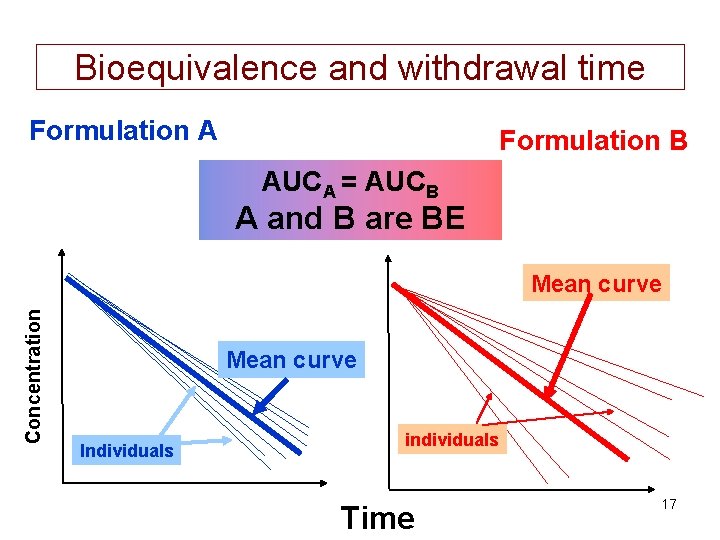
Bioequivalence and withdrawal time Formulation A Formulation B AUCA = AUCB A and B are BE Concentration Mean curve Individuals individuals Time 17
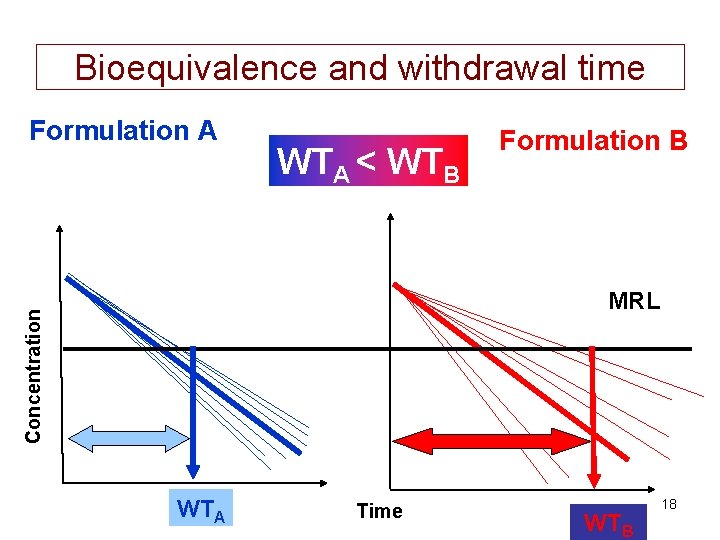
Bioequivalence and withdrawal time Formulation A WTA < WTB Formulation B Concentration MRL WTA Time WTB 18

Bioequivalence and the problem of drug residues • Bioequivalence studies in foodproducing animals are not acceptable in lieu of residues data: why? 19

Definitions ans statistics associated to (average) bioequivalence and withdrawal time are fundamentally different 20

EMA guidance for WT 21

Definition of WT by EMA 22

The EU statistical definition of the Withdrawal Time "WT is the time when the upper onesided 95% tolerance limit for residue is below the MRL with 95% confidence" Tolerance limits: Limits for a percentage of a population 23

WT definition is related to a tolerance interval (limits) • Tolerance interval is a statistical interval within which a specified proportion of a population falls (here 95%) with some confidence (here 95%) • To compute a WT you have to specify two different percentages. – The first expresses what fraction (percentage) of the values (animals) the interval will contain. – The second expresses how sure you want to be – If you set the second value (how sure) to 50%, then a tolerance interval is the same as a prediction interval (see our first exercise). 24
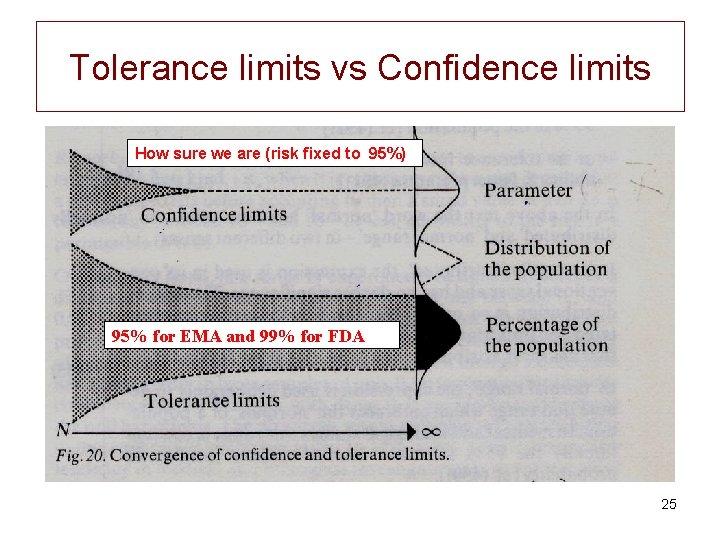
Tolerance limits vs Confidence limits How sure we are (risk fixed to 95%) 95% for EMA and 99% for FDA 25

Bioequivalence and withdrawal time • Bioequivalence is related to a confidence interval for a parameter (e. g. mean AUC-ratio for 2 formulations) • Withdrawal time is related to a tolerance limit (quantile 95% EU or 99% in US) and it is define as the time when the upper one- sided 95% tolerance limit for residue is below the MRL with 95% confidence“ • The fact to guarantee that the 90% confidence interval for the AUC-ratio of the two formulations lie within an acceptance interval of 0. 80 -1. 25 do not guarantee that the upper one- sided 95% tolerance limit for residue is below the MRL with 95% confidence for both formulations“ 30

Who is affected by an inadequate statistical risk associated to a WT • It is not a consumer safety issue • It is the farmer that is protected by the statistical risk associated to a WT – It is the risk, for a farmer, to be controlled positive while he actually observe the WT. – When the WT is actually observed, at least 95% of the farmers in an average of 95% of cases should be negative 31

How to build a counterexample to show that it is possible to have two formulations complying with BE requirements while their WTs largely differ. 32

Counterexample • In mathematics, counterexamples are often used to show that certain conjectures are false, 33

How to build a counter example • Considering that BE is demonstrated using plasma concentration over a rather short period of time (e. g. 24 or 48 h) but that WT is generally much longer (e. g. 12 days), you can expect that two bioequivalent formulations exhibiting a so-called very late terminal phase could have different WT. 34
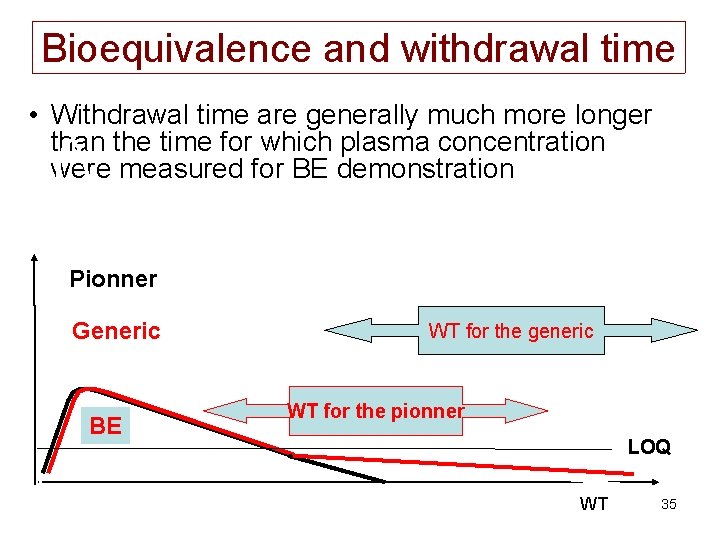
Bioequivalence and withdrawal time • Withdrawal time are generally much more longer than the time for which plasma concentration were measured for BE demonstration Pionner Generic BE WT for the generic WT for the pionner LOQ WT 35

Use of Monte Carlo simulations 36
What is the origin of the word Monte Carlo? Toulouse Monte-Carlo (Monaco) 37
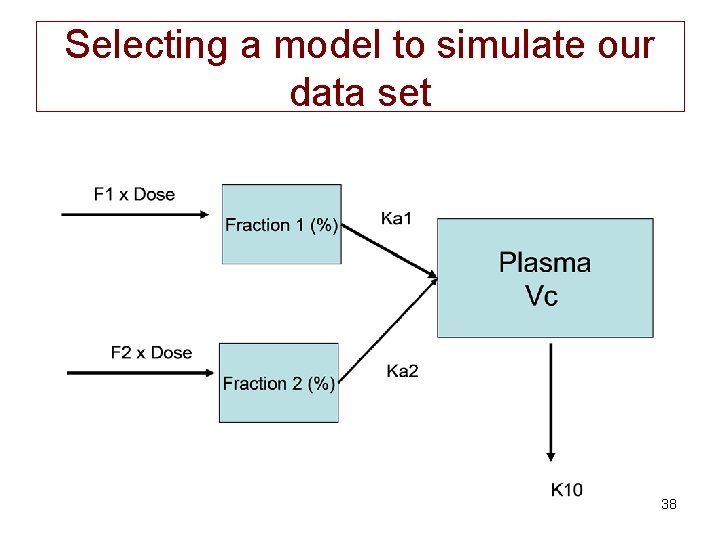
Selecting a model to simulate our data set 38

Equation describing our model 39

Step 1: Implementation of the selected model in an Excel sheet • We have to write this equation in Excel and to solve it for: – times ranging from 0 to 144 h for plasma concentrations for BE – from 144 to 1440 h for tissular concentrations for WT. 40
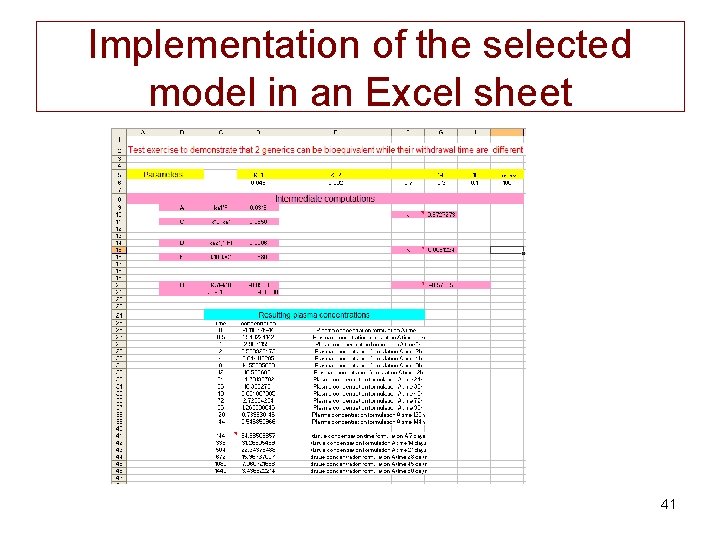
Implementation of the selected model in an Excel sheet 41

Step 2: Monte Carlo simulation to establish a data set using CB • For the BE trial, we need 12 animals to carry out a 2 x 2 crossover design i. e. 24 vectors of plasma concentrations from time 0 to 144 h (12 vectors formulation 1 and 12 vectors formulation 2). • For the WT we are planning a trial with 4 different slaughter times (14, 21, 28 and 45 days) and for each slaughter time, we need 6 samples i. e. 24 animals per formulation; thus the total number of animals to simulate for the WT is 48. • The total number of vectors to simulate is of n=72 i. e. 36 formulation 1 and 36 formulation 2. 42

Step 2: Monte Carlo simulation to establish a data set using CB • Only variability is introduced in F 1; – for the first formulation F 1 is normally distributed with a mean F 1=0. 7 associated with a relatively low inter-animal variability of 10%. – For the second formulation we also fixed F 1=0. 7 but with an associated CV of 30% meaning more variability between animals for this second formulation but the same average PK parameters. 43
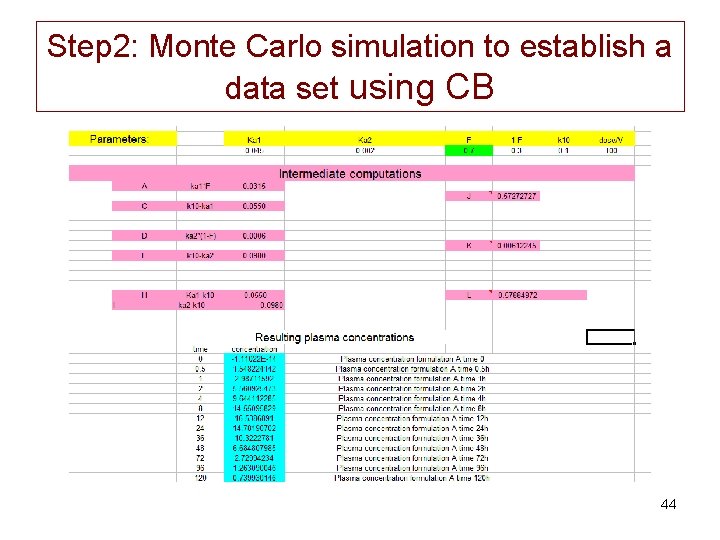
Step 2: Monte Carlo simulation to establish a data set using CB 44
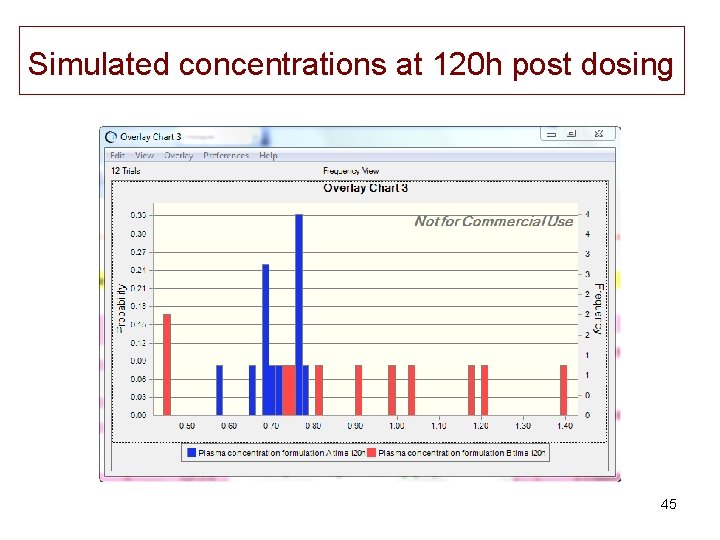
Simulated concentrations at 120 h post dosing 45
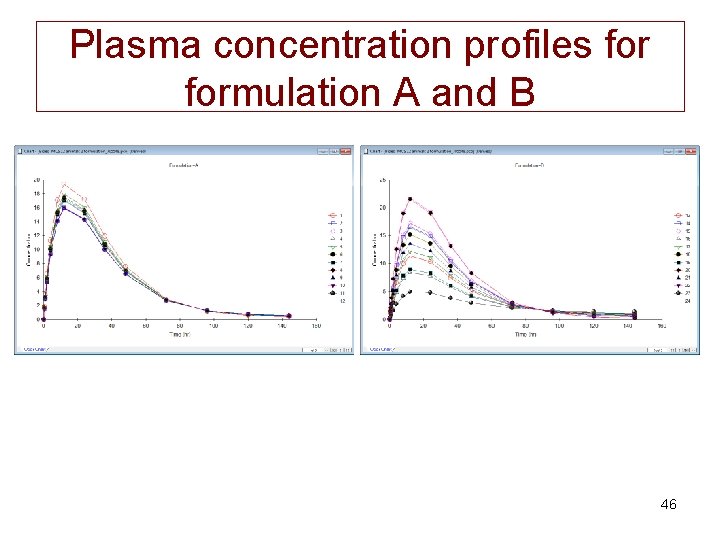
Plasma concentration profiles formulation A and B 46

NCA of the data set • Computation of AUC using the NCA by WNL • Computation of some statistics using the statistical tool of WNL. – It appeared that the ratio of the AUC (geometric means) was 0. 83 (659. 2/788. 8). – This point estimate is to close to the a priori lower bound of the a priori confidence interval for a BE trial (lower bound is 0. 8) thus I know a priori that it will be impossible to conclude to a BE with this data set 47

New simulation • So, I decided to re-run my simulations but with a lower variances for the second formulation • Second simulation CV=10 and 20% formulations A and B respectively; same mean F=0. 70. 48
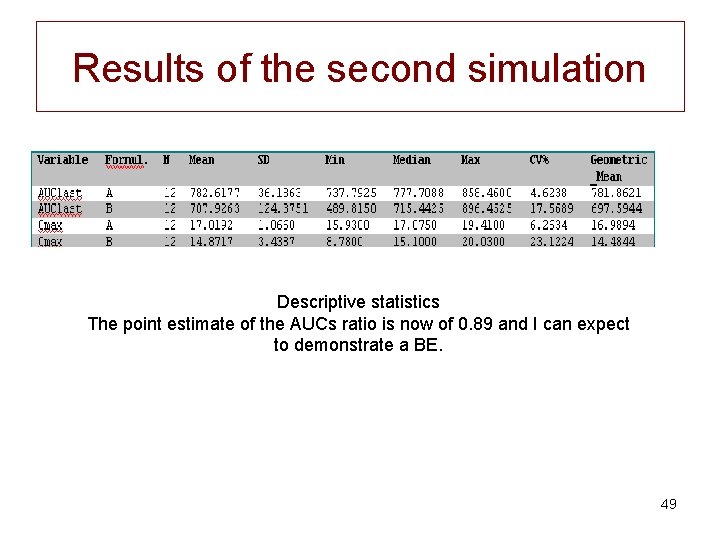
Results of the second simulation Descriptive statistics The point estimate of the AUCs ratio is now of 0. 89 and I can expect to demonstrate a BE. 49
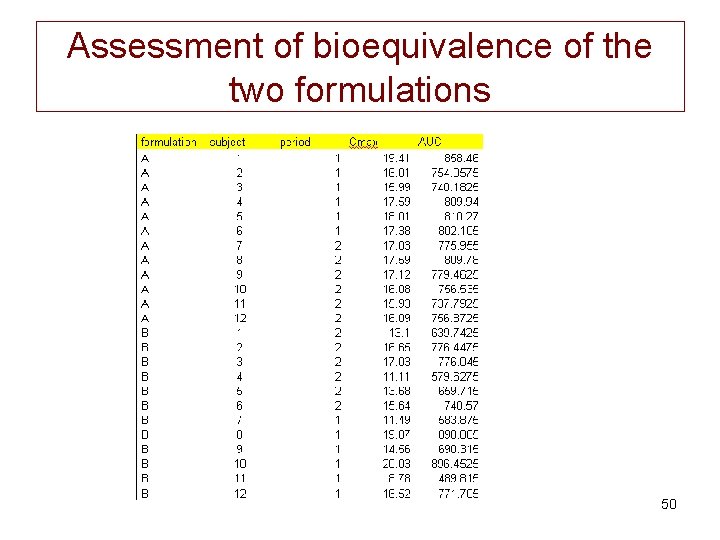
Assessment of bioequivalence of the two formulations 50
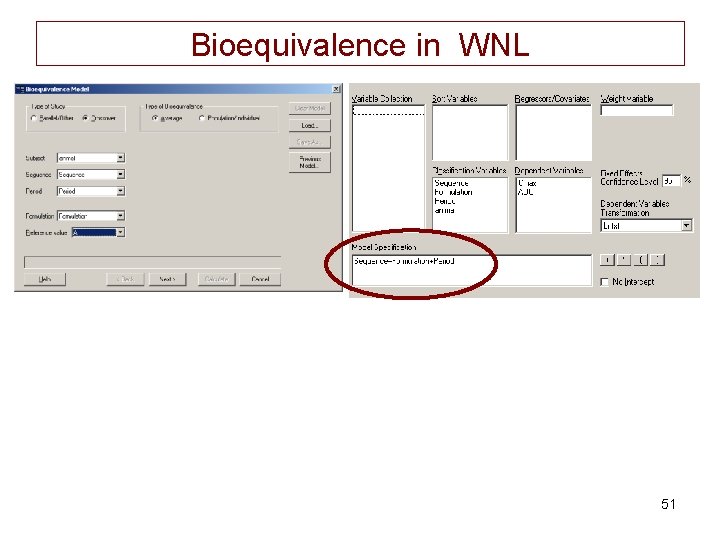
Bioequivalence in WNL 51

52

Bioequivalence output • Cmax: – the regulatory 90% CI was from 73. 9 -: 98. 3 • AUC – the regulatory CI 90% was : 80. 2 - 98. 9 I permuted the 2 CI in the document? ) 53

Computation of withdrawal time for the two formulations 54

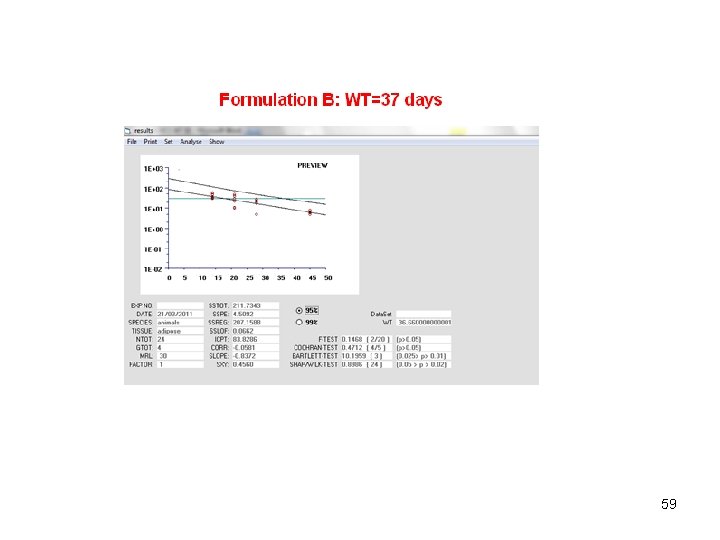
How to obtain raw data • Simulate the model between 144 and 1440 h (i. e. from 6 to 60 days) • You need 6 tissular samples for each of the four sampling times • Sampling times: 14, 21 , 28 and 45 days • Tissular concentrations are 100 times plasma concentrations • Thus for the 6 first animals, you select forecasts at day 14, then forecasts at day 21 for animals 7 to 12 and so on. 55

MRL=30 56
Computation of a WT 57
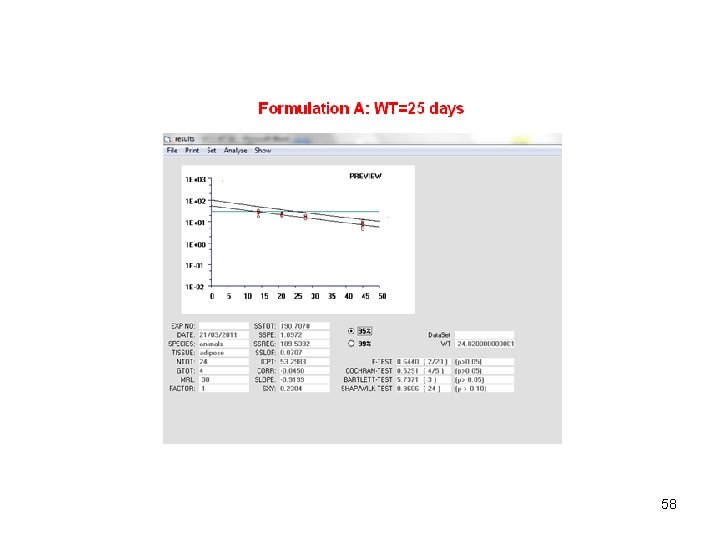
58
59

Conclusion • This example shows that we are in position to demonstrate that an average bioequivalence between two formulations is not a proof to guarantee that the formulations have identical withdrawal times. 60
- Slides: 56