Exercise 2 How to chose the best property

Exercise 2 How to chose the best property prediction method for simulation ? 9/17/2021 054402 Design & Analysis II 1

Importance of Selecting the Appropriate Prediction Method n n 9/17/2021 Correct predictions of the physical properties of the mixture as a function of temperature and pressure. Each method is suitable only for particular types of components and limited to certain operating conditions. Choosing the wrong method may lead to incorrect simulation results. Particularly important for reliable computations associated with separation operations (distillation, LL extraction, etc. ). 054402 Design & Analysis II 2

Principle Steps in Selecting the Appropriate Thermodynamics Package 1. Choosing the most suitable model/thermo method. 2. Comparing the obtained predictions with data from the literature. 3. Adding estimates for components that not available in the chosen thermo package. Can they be neglected? 4. Generation of lab data if necessary to check thermo model. 9/17/2021 054402 Design & Analysis II 3

Sources of Information 1. 2. 3. 4. 9/17/2021 Publications and professional literature that deal with the process in question or with the components that participate in the process. Simulator reference manual (HELP). DATABANKS Rules of thumb. 054402 Design & Analysis II 4

Issues in Selection of the Appropriate Thermodynamics Package n n n 9/17/2021 Nature of mixture (e. g. , hydrocarbon, polar, electrolyte, etc. ) Pressure and temperature range Availability of data. 054402 Design & Analysis II 5

Recommendations for the Selection of the Appropriate Thermodynamics Package Eric Carlson, “Don’t gamble with physical properties for simulations, ” Chem. Eng. Prog. October 1996, 35 -46 Prof J. D. (Bob) Seader, University of Utah Hyprotech Recommendations 9/17/2021 054402 Design & Analysis II 6
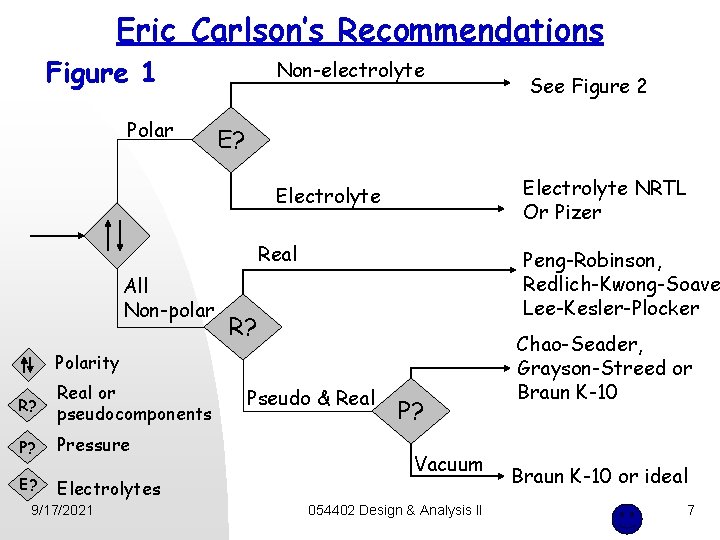
Eric Carlson’s Recommendations Figure 1 Polar Non-electrolyte E? Electrolyte NRTL Or Pizer Electrolyte Real All Non-polar Peng-Robinson, Redlich-Kwong-Soave Lee-Kesler-Plocker R? Polarity R? Real or pseudocomponents P? Pressure E? Electrolytes 9/17/2021 See Figure 2 Pseudo & Real P? Vacuum 054402 Design & Analysis II Chao-Seader, Grayson-Streed or Braun K-10 or ideal 7
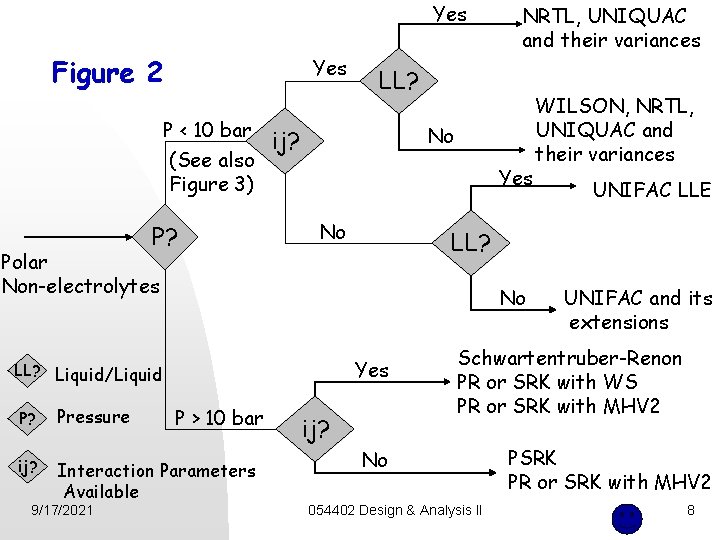
Yes Figure 2 Yes P < 10 bar (See also Figure 3) P? LL? No ij? Yes No No Yes LL? Liquid/Liquid P > 10 bar P? Pressure ij? Interaction Parameters Available WILSON, NRTL, UNIQUAC and their variances UNIFAC LLE LL? Polar Non-electrolytes 9/17/2021 NRTL, UNIQUAC and their variances ij? UNIFAC and its extensions Schwartentruber-Renon PR or SRK with WS PR or SRK with MHV 2 No 054402 Design & Analysis II PSRK PR or SRK with MHV 2 8
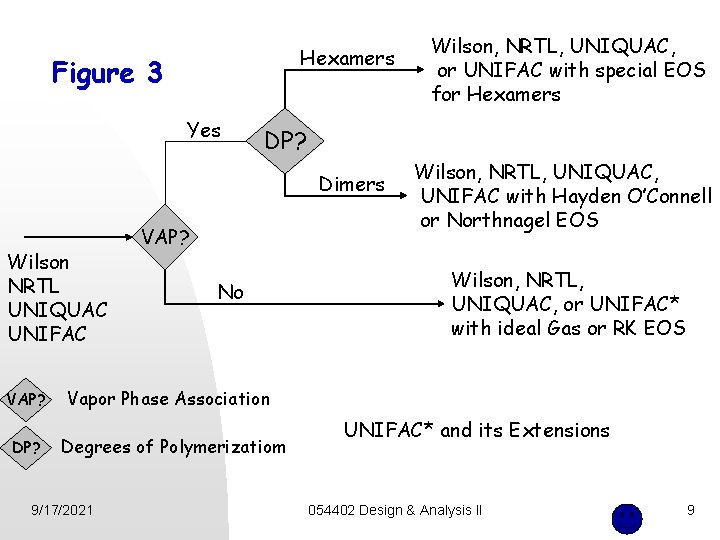
Hexamers Figure 3 Yes DP? Dimers Wilson NRTL UNIQUAC UNIFAC VAP? DP? Wilson, NRTL, UNIQUAC, or UNIFAC with special EOS for Hexamers VAP? No Wilson, NRTL, UNIQUAC, UNIFAC with Hayden O’Connell or Northnagel EOS Wilson, NRTL, UNIQUAC, or UNIFAC* with ideal Gas or RK EOS Vapor Phase Association Degrees of Polymerizatiom 9/17/2021 UNIFAC* and its Extensions 054402 Design & Analysis II 9
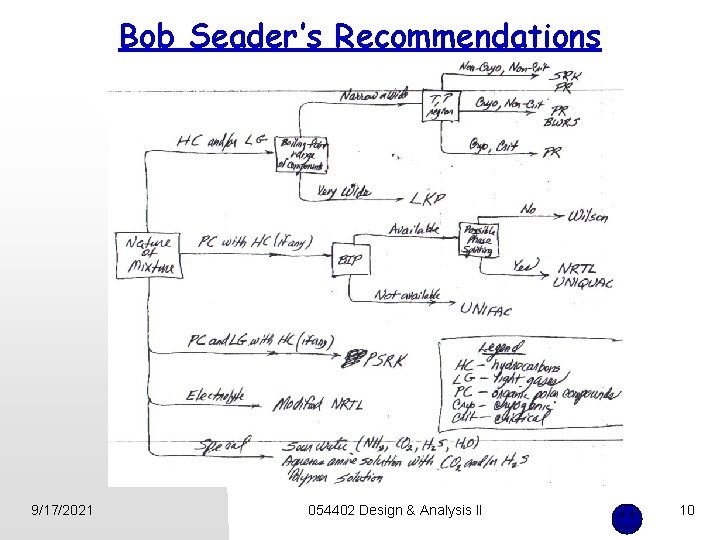
Bob Seader’s Recommendations 9/17/2021 054402 Design & Analysis II 10
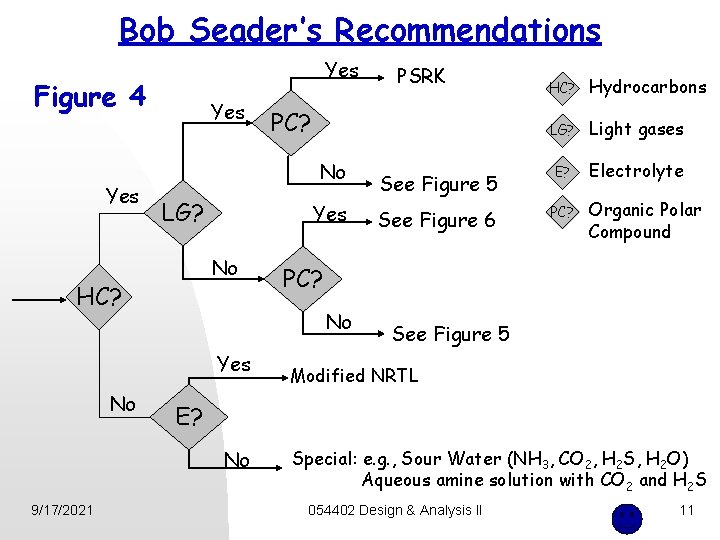
Bob Seader’s Recommendations Yes Figure 4 Yes Yes No HC? No LG? Light gases See Figure 5 E? Electrolyte See Figure 6 PC? Organic Polar Compound See Figure 5 Modified NRTL E? No 9/17/2021 Hydrocarbons PC? No Yes HC? PC? No LG? PSRK Special: e. g. , Sour Water (NH 3, CO 2, H 2 S, H 2 O) Aqueous amine solution with CO 2 and H 2 S 054402 Design & Analysis II 11
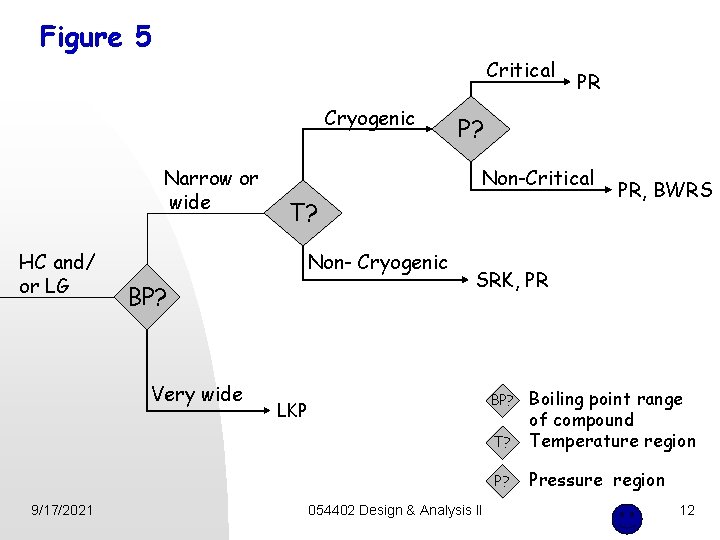
Figure 5 Critical Cryogenic Narrow or wide HC and/ or LG 9/17/2021 P? Non-Critical T? Non- Cryogenic BP? Very wide PR PR, BWRS SRK, PR T? Boiling point range of compound Temperature region P? Pressure region BP? LKP 054402 Design & Analysis II 12
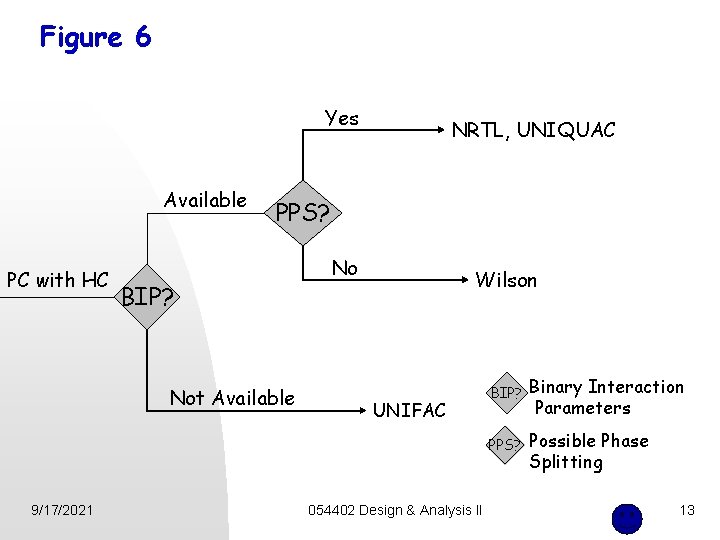
Figure 6 Yes Available PC with HC PPS? BIP? Not Available 9/17/2021 NRTL, UNIQUAC No Wilson UNIFAC 054402 Design & Analysis II BIP? Binary Interaction Parameters PPS? Possible Phase Splitting 13
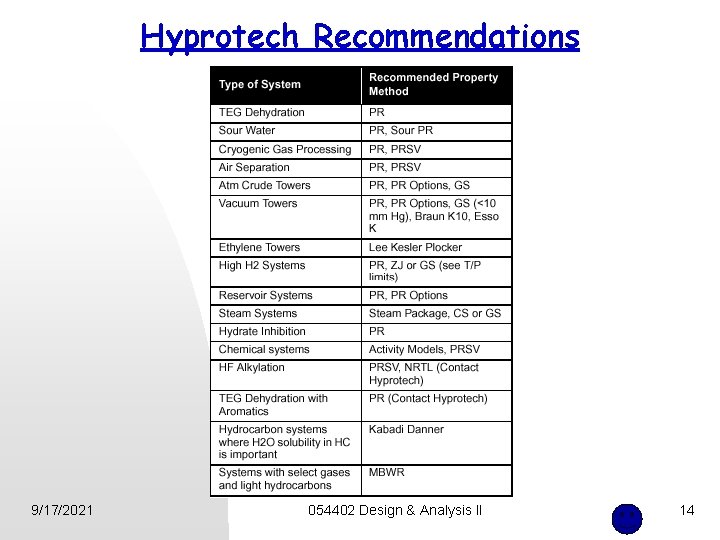
Hyprotech Recommendations 9/17/2021 054402 Design & Analysis II 14

Example n Find the best thermodynamic package for 1 -Propanol , H 2 O mixture. Eric Carlson, “Don’t gamble with physical properties for simulations, ” Chem. Eng. Prog. October 1996, 35 -46 Prof J. D. (Bob) Seader, University of Utah 9/17/2021 054402 Design & Analysis II 15
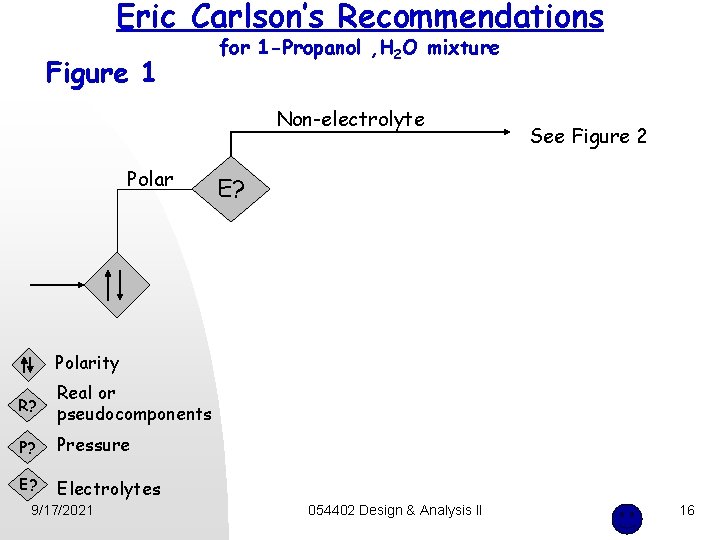
Eric Carlson’s Recommendations Figure 1 for 1 -Propanol , H 2 O mixture Non-electrolyte Polar See Figure 2 E? Polarity R? Real or pseudocomponents P? Pressure E? Electrolytes 9/17/2021 054402 Design & Analysis II 16
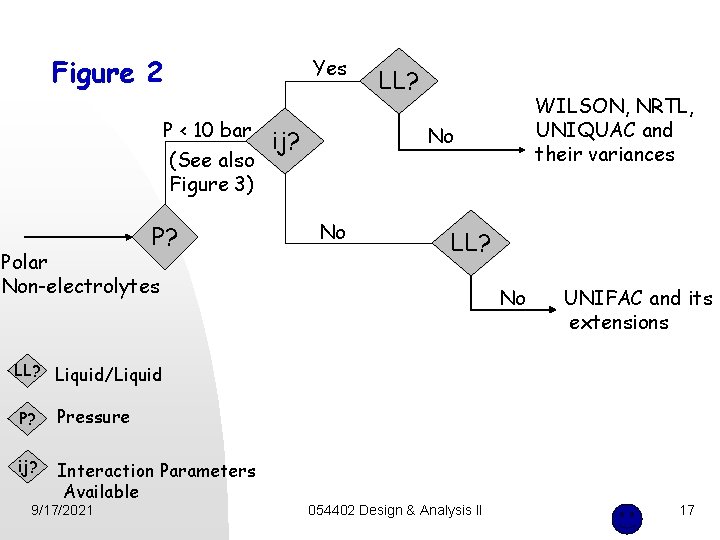
Figure 2 Yes P < 10 bar (See also Figure 3) P? Polar Non-electrolytes LL? WILSON, NRTL, UNIQUAC and their variances No ij? No LL? No UNIFAC and its extensions LL? Liquid/Liquid P? Pressure ij? Interaction Parameters Available 9/17/2021 054402 Design & Analysis II 17
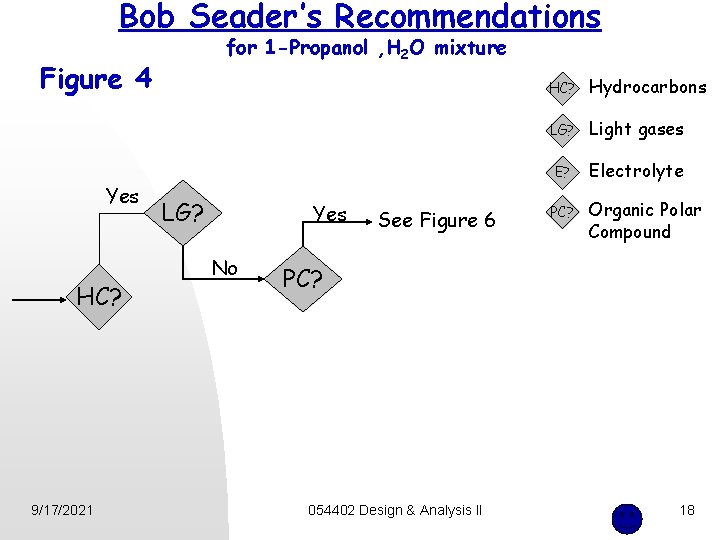
Bob Seader’s Recommendations for 1 -Propanol , H 2 O mixture Figure 4 Yes HC? 9/17/2021 LG? Yes No See Figure 6 HC? Hydrocarbons LG? Light gases E? Electrolyte PC? Organic Polar Compound PC? 054402 Design & Analysis II 18
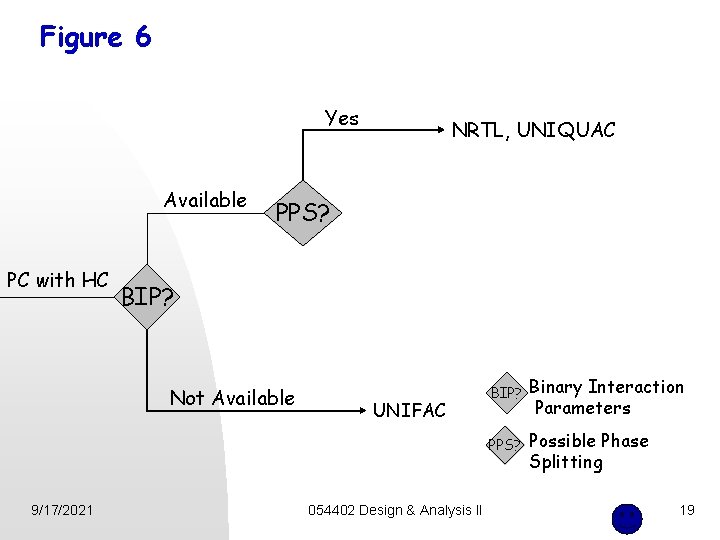
Figure 6 Yes Available PC with HC PPS? BIP? Not Available 9/17/2021 NRTL, UNIQUAC UNIFAC 054402 Design & Analysis II BIP? Binary Interaction Parameters PPS? Possible Phase Splitting 19
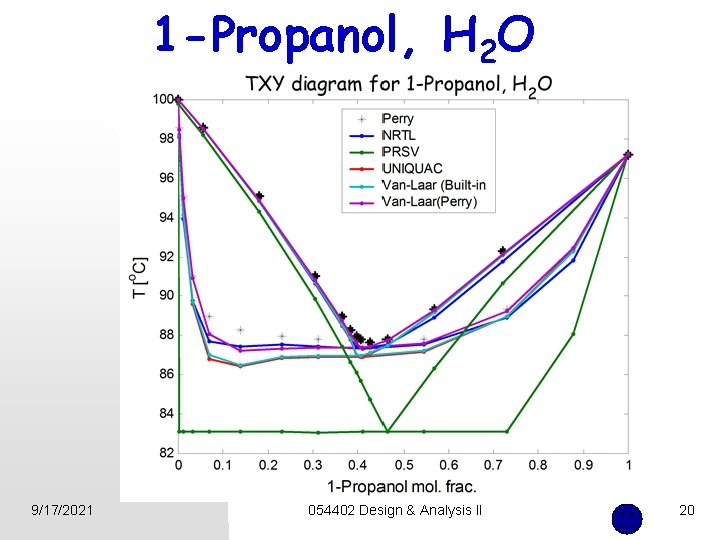
1 -Propanol, H 2 O 9/17/2021 054402 Design & Analysis II 20
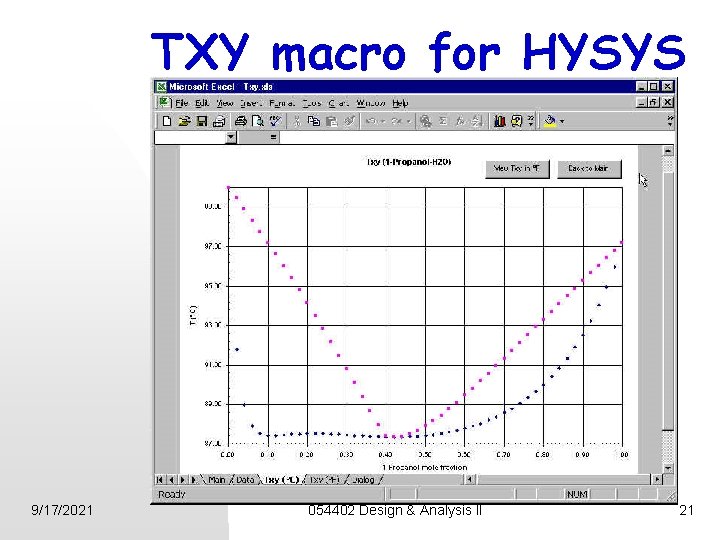
TXY macro for HYSYS 9/17/2021 054402 Design & Analysis II 21
- Slides: 21