ExamplesApplications of absorption and PL cw spectroscopy Carlo

Examples/Applications of absorption and PL cw spectroscopy Carlo Altucci Course of Atomic and Molecular Physics and Spectroscopy 2014 -2015 Laboratory of Biophotonics and Ultrafast Processes Dipartimento di Fisica Università di Napoli "Federico II " Napoli, Italy Carlo. Altucci@unina. it

www. femto. unina. it 2
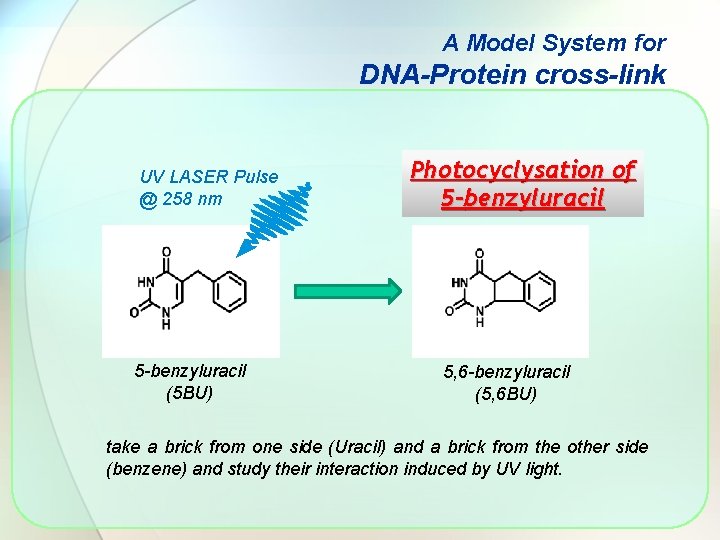
A Model System for DNA-Protein cross-link UV LASER Pulse @ 258 nm 5 -benzyluracil (5 BU) Photocyclysation of 5 -benzyluracil 5, 6 -benzyluracil (5, 6 BU) take a brick from one side (Uracil) and a brick from the other side (benzene) and study their interaction induced by UV light.

4
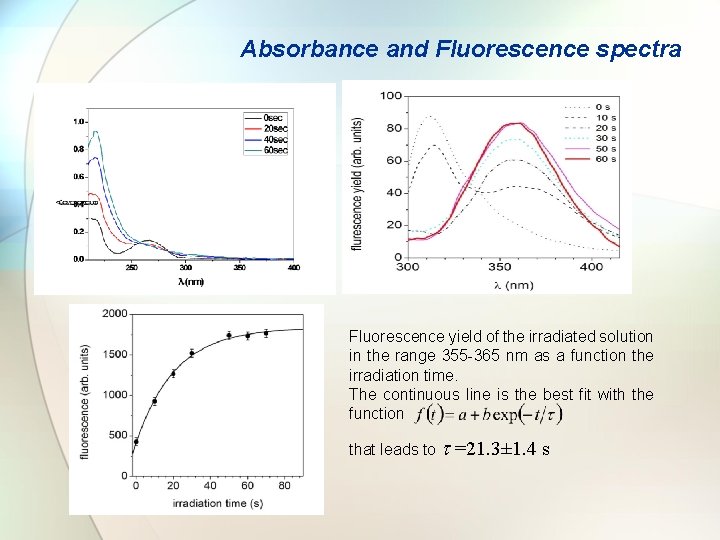
Absorbance and Fluorescence spectra Fluorescence yield of the irradiated solution in the range 355 -365 nm as a function the irradiation time. The continuous line is the best fit with the function that leads to t =21. 3± 1. 4 s
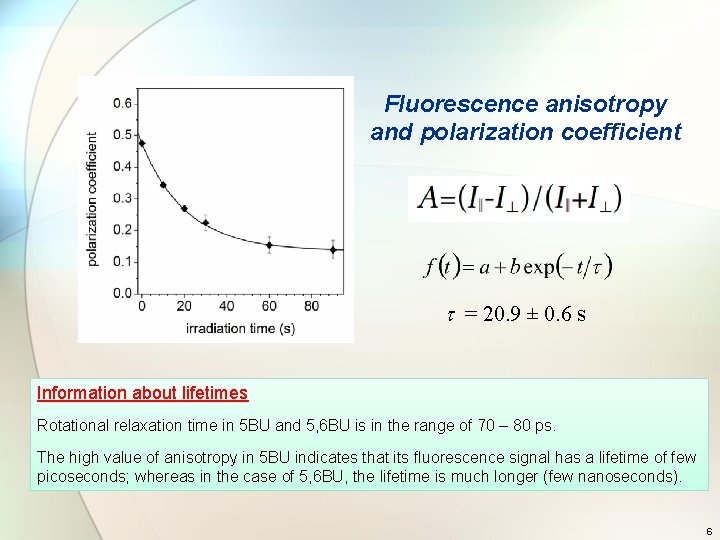
Fluorescence anisotropy and polarization coefficient t = 20. 9 ± 0. 6 s Information about lifetimes Rotational relaxation time in 5 BU and 5, 6 BU is in the range of 70 – 80 ps. The high value of anisotropy in 5 BU indicates that its fluorescence signal has a lifetime of few picoseconds; whereas in the case of 5, 6 BU, the lifetime is much longer (few nanoseconds). 6
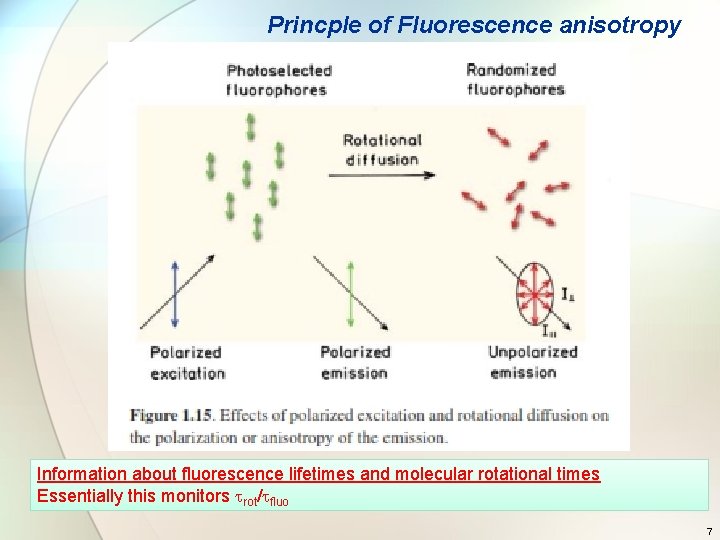
Princple of Fluorescence anisotropy Information about fluorescence lifetimes and molecular rotational times Essentially this monitors rot/ fluo 7

How to work out the photo-cyclization probability from steady state fluorescence anisotropy t =21. 3± 1. 4 s To confirm we independently measured also the behavior of the anisotropy t = 20. 9 ± 0. 6 s 8

The photo-cyclization reaction t : the characteristic time for the transformation of 5 BU into 5, 6 BU where f. L is the repetition rate of the laser, 2 k. Hz, pabs is the probability that a molecule absorbs one photon in a single laser pulse, and ppc is the probability that the excited molecule will photocyclize. λ=258 nm pabs is measured by measuring abs which is proportional to the measure absorptivity, (mol-1 cm-1)= abs (cm 2)/(3. 82 x 10 -21) 9
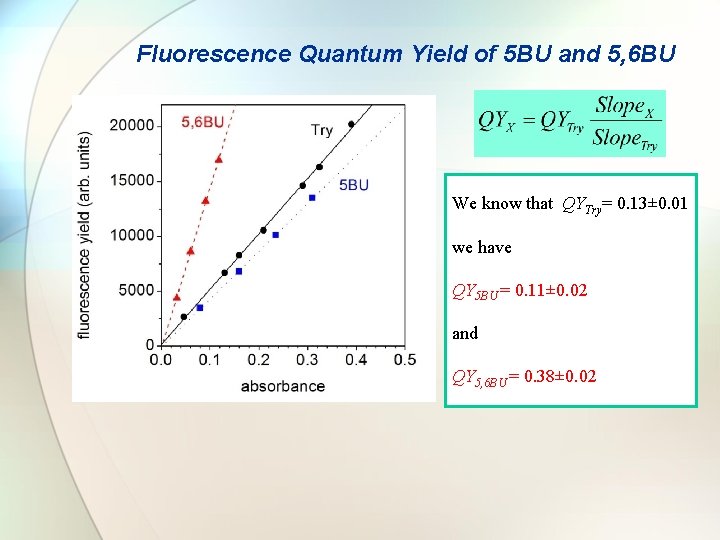
Fluorescence Quantum Yield of 5 BU and 5, 6 BU We know that QYTry= 0. 13± 0. 01 we have QY 5 BU = 0. 11± 0. 02 and QY 5, 6 BU = 0. 38± 0. 02

Pump and Probe Technique Femtosecond spectroscopy is the most powerful technique to obtain information on ultrashort time scales !! The laser pulse for excitation ('pump') modulates the initial state. Then, the time-delayed pulse measures ('probes') the optical properties of the sample. Doing so for different time delays one obtains snap-shots of the dynamics on a femtosecond time scale.

Pump and Probe Technique Fluorescence (induced by the probe pulse) • At one delay time, many pump-probe cycles are collected for sufficient S/N level in the data; • Changing the delay time step-by-step to cover the entire kinetics trace; • Time resolution determined by the laser pulse duration (~200 fs); • Time window determined by the optical delay length (i. e. several ns).

13

The PIT (Photonic Immobilization Technique) 14

15

Absorption and PL spectra to characterize antibodies immuno-recognition function Intermediate treatment area where PIT mechanism is active but immunorecognition preserved. This area is appearing only with fs treatment. Ns pulses, in fact, damage more due to thermal effects 16

Example: Absorbance spectra of direct bandgap semiconductors Transition rate by Fermi’s Golden Rule Parabolic approximation of Conduction and Valence Band in direct bandgap semiconductors III-V semiconductor In. As E(k) CB Eg k VB for direct bandgap semiconductors close to the bandgap the density of states can be approximater as From M. Fox, Optical properties of solids (Oxford University Press , 2001)

Absorbance spectra of Cd. S and Zn. S nanoparticlesassembled films on a glass substrate produced by femtosecond laser deposition Nanostructures of II-VI semiconductor materials such as Cd. S and Zn. S attract interest because they show significant quantum confinement effects which influence their electrical and optical properties. The possibility to tune the properties of the nanostructures also motivates research into their application in photovoltaic, photonic, and optoelectronic devices and sensors. Cd. S and Zn. S are direct bandgap semiconductors with reported bandgap energy of 2. 25 -2. 45 and 3. 5 -3. 8 e. V, respectively From Sanz, Amoruso et al, J. Phys. Chem. C 115, 3203– 3211, (2011)

In a real case (II-IV semiconductor nanostructures): • there can be impurities and defect states with energy within the bandgap • as the photon energy hn increaseas above the bandgap the density of states does not obey the dependence on frequency derived above in parabolic approximation is not completely valid Absorbance spectra of Cd. S and Zn. S nanoparticles-assembled films Tauc plot method (see inset) Close to badgap energy (a hn)2 hn for hn > Eg Eg is obtained by the intersection between two linear dependences below and above hn* = Eg (inset) Eg (Cd. S) 2. 4 e. V Eg (Zn. S) 3. 5 e. V

Absorption and Emission to characterize low-dimension structures for several applications (energetics, photonics, bio-photonics) – contribution from Felice Gesuele 20
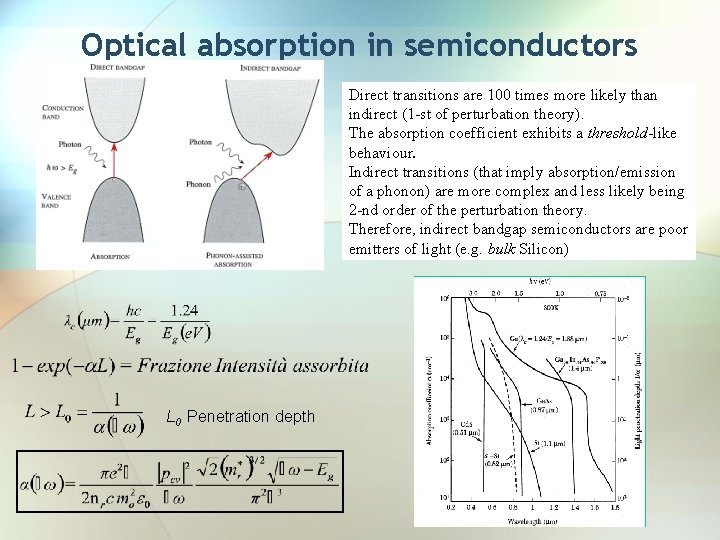
Optical absorption in semiconductors Direct transitions are 100 times more likely than indirect (1 -st of perturbation theory). The absorption coefficient exhibits a threshold-like behaviour. Indirect transitions (that imply absorption/emission of a phonon) are more complex and less likely being 2 -nd order of the perturbation theory. Therefore, indirect bandgap semiconductors are poor emitters of light (e. g. bulk Silicon) L 0 Penetration depth

Low-dimension nanostructures Structures where one or more sizes are as short as the electron wavefunction extension. This modifies the band structure (as compared to the bulk material) and the Density of States (DOS) that becomes discrete from continuous.

Quantum Dots: tuning size and optical properties A bulk semiconductor has continuous valence and conduction bands, separated by the energy gap Eg, 0. A Quantum Dot is characterized by discrete atomic-like levels having values determined by its radius R. The energy separation between the lowest electron and the highest hole level depends on R (quantum confinement ). The absorption spectrum of a QDs ensemble contains the bulk spectrum with the overlap of a series of peaks in correspondence of discrete transitions.
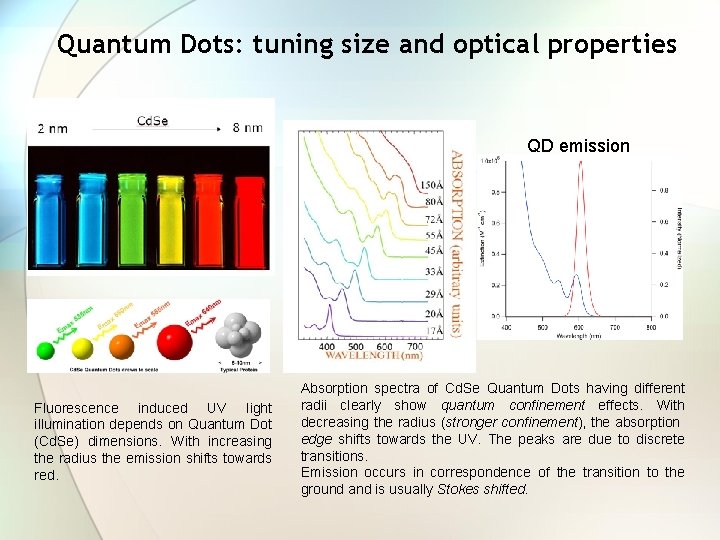
Quantum Dots: tuning size and optical properties QD emission Fluorescence induced UV light illumination depends on Quantum Dot (Cd. Se) dimensions. With increasing the radius the emission shifts towards red. Absorption spectra of Cd. Se Quantum Dots having different radii clearly show quantum confinement effects. With decreasing the radius (stronger confinement), the absorption edge shifts towards the UV. The peaks are due to discrete transitions. Emission occurs in correspondence of the transition to the ground and is usually Stokes shifted.

Carbon Nanotubes (CNT) A CNT can thought of as a graphene rolled plane. In practice, each CNT is characterized by two indeces (n, m) that define the Ch vector. This vector describes how to roll the graphene plane. T represents the tube axis. Depending on the (n, m) values the CNT can be either semiconductor or metal

Carbon Nanotubes: excitation induced photoluminescence PLE is PL obtained at a variable excitation wavelength. Each line corresponds to an emission spectrum at a fixed excitation wavelength. In practice this technique allows one to identify differenty (n, m) values in a subset of CNTs containing several (n, m) couples. PL of a selected CNT (n, m) will be at maximum when the excitation wavelength will match the second transition energy E 22.

Bi-dimensional materials A A calss of crystals made of a transition element (Mo, W) and an element of the 8 -th group exhibits the property to be layered. In practice in plane bonds are very strong, whereas out of plane bonds are weak. These materials are easily esfoliatied until a single atomic layer. The bulk material is an indirect gap semiconductor The single layer is a direct gap one. Practically, the indirect gap increases in energy until it exceeds the direct one. K. F. Mak (T. Heinz), Physical Review Letters 105 (13), 136805
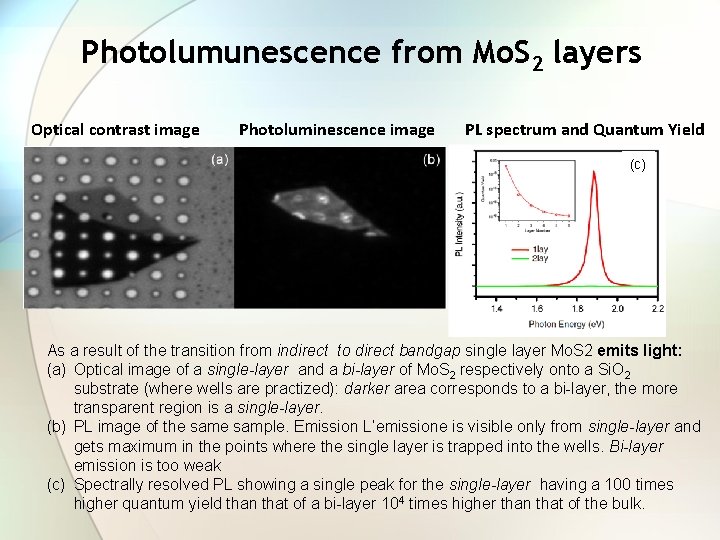
Photolumunescence from Mo. S 2 layers Optical contrast image Photoluminescence image PL spectrum and Quantum Yield (c) As a result of the transition from indirect to direct bandgap single layer Mo. S 2 emits light: (a) Optical image of a single-layer and a bi-layer of Mo. S 2 respectively onto a Si. O 2 substrate (where wells are practized): darker area corresponds to a bi-layer, the more transparent region is a single-layer. (b) PL image of the sample. Emission L’emissione is visible only from single-layer and gets maximum in the points where the single layer is trapped into the wells. Bi-layer emission is too weak (c) Spectrally resolved PL showing a single peak for the single-layer having a 100 times higher quantum yield than that of a bi-layer 104 times higher than that of the bulk.
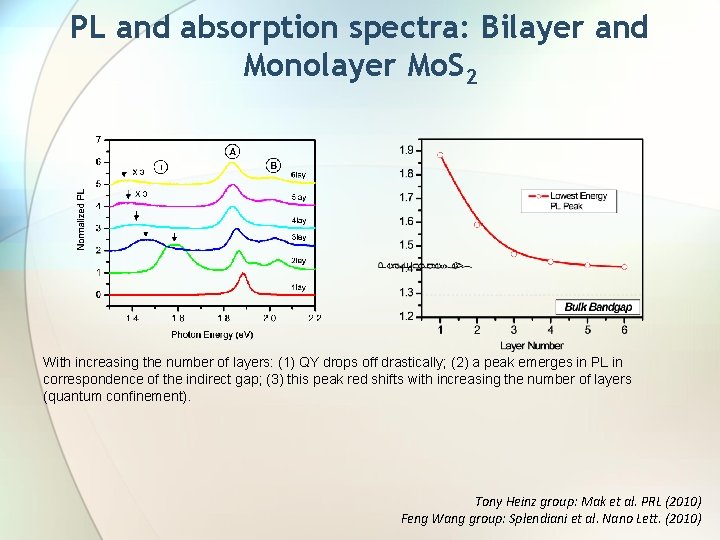
PL and absorption spectra: Bilayer and Monolayer Mo. S 2 With increasing the number of layers: (1) QY drops off drastically; (2) a peak emerges in PL in correspondence of the indirect gap; (3) this peak red shifts with increasing the number of layers (quantum confinement). Tony Heinz group: Mak et al. PRL (2010) Feng Wang group: Splendiani et al. Nano Lett. (2010)
- Slides: 29