Examples of nomenclature IUPAC name 2 propen1 ol























- Slides: 28





Examples of nomenclature IUPAC name 2 -propen-1 -ol 2 -Butanol 2 -Methyl-1 -Propanol Common name Allyl alcohol Sec-Butyl alc. Isobutyl alc. Ethyl methyl carbinol Isopropyl carbinol Carbinol method Vinyl carbinol

Classification of alcohols According to number of OH groups • • • Monohydroxy alcohols: which contain one OH group e. g. ethanol Dihydroxy alcohols: which contain two OH group e. g. ethyleneglycol. Polyhydroxy alcohols: which contain more than two OH groups e. g. glycerol and carbohydrate.

n According to number of OH groups Monohydroxy alcohols are classified according to type of carbon bearing OH into three types: • 1 ry alcohols: in which OH attach with 1 ry carbon e. g. RCH 2 OH • 2 nd alcohols: in which OH attach with 2 nd carbon e. g. R 2 CHOH • t alcohols : in which OH attach with tertiary carbon e. g. R 3 COH

Preparation of alcohols n n a) Preparation of 1 ry alcohol 1. 1 - Hydrolysis of alkene 2. Hydrolysis of alkyl halide

n E. g. conversion of benzene to benzyl alcohol 3. Reduction of carbonyl compound Reduction of aldehyde gives 1 ry alcohol, while of ketone gives 2 nd alcohol

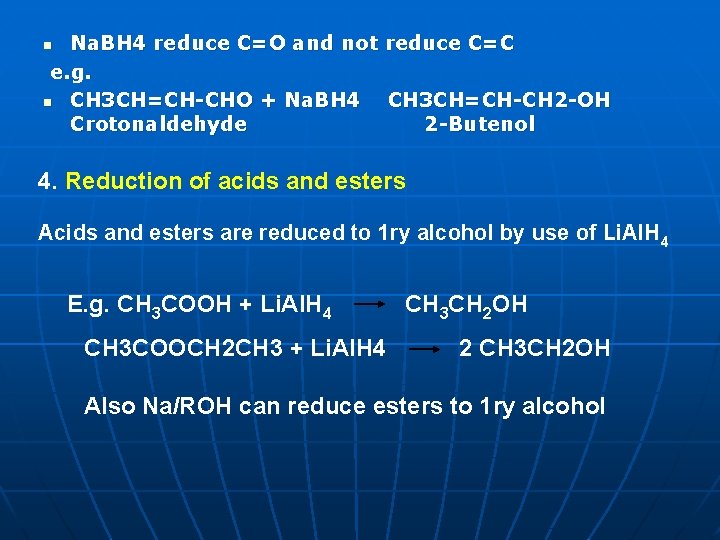
Na. BH 4 reduce C=O and not reduce C=C e. g. n CH 3 CH=CH CHO + Na. BH 4 CH 3 CH=CH CH 2 OH Crotonaldehyde 2 Butenol n 4. Reduction of acids and esters Acids and esters are reduced to 1 ry alcohol by use of Li. Al. H 4 E. g. CH 3 COOH + Li. Al. H 4 CH 3 COOCH 2 CH 3 + Li. Al. H 4 CH 3 CH 2 OH 2 CH 3 CH 2 OH Also Na/ROH can reduce esters to 1 ry alcohol

Grignard reagent can be prepared as follow: Grignard reagent is used to prepare 1 ry, 2 nd, and t-alcohols a) Preparation of 1 ry alcohol is prepared from Grignard reagent and formaldehyde

n b (Preparation of secondary alcohols Secondary alcohols are prepared from Grignard reagent and aldehyde other than formaldehyde.

c (Preparation of tertiary alcohols Tertiary alcohols are prepared from Grignard reagent and ketones.

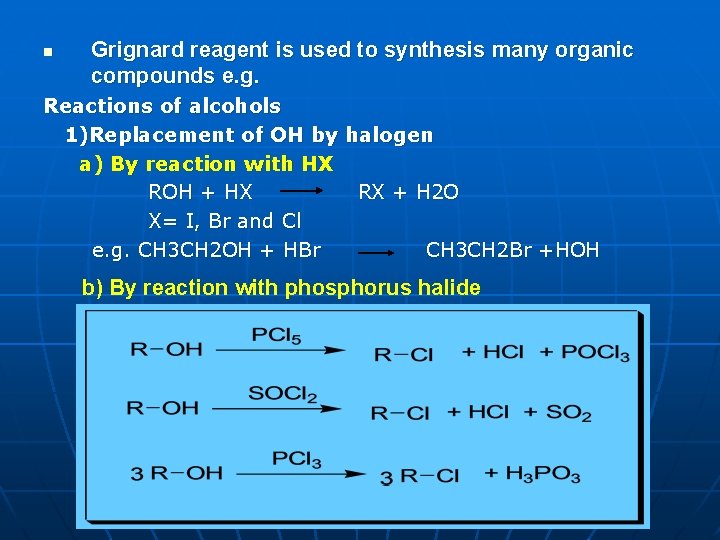
n Grignard reagent is used to synthesis many organic compounds e. g. Reactions of alcohols 1)Replacement of OH by halogen a) By reaction with HX ROH + HX RX + H 2 O X= I, Br and Cl e. g. CH 3 CH 2 OH + HBr CH 3 CH 2 Br +HOH b) By reaction with phosphorus halide

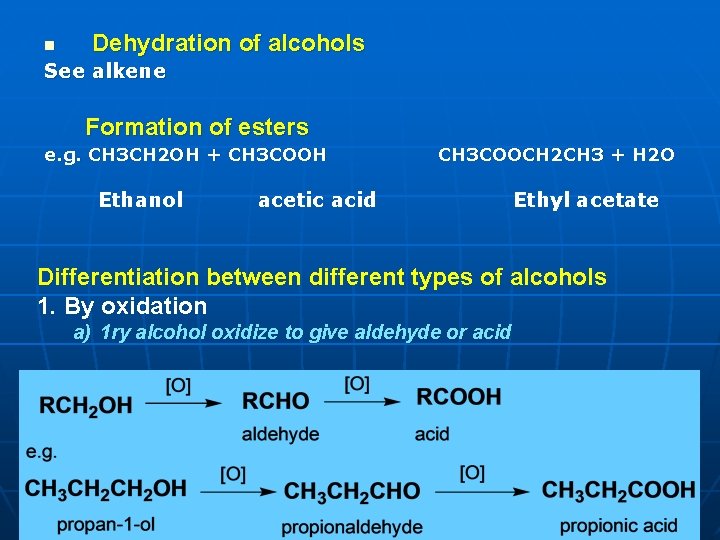
n Dehydration of alcohols See alkene Formation of esters e. g. CH 3 CH 2 OH + CH 3 COOH Ethanol CH 3 COOCH 2 CH 3 + H 2 O acetic acid Ethyl acetate Differentiation between different types of alcohols 1. By oxidation a) 1 ry alcohol oxidize to give aldehyde or acid

Examples of oxidizing agent are K 2 Cr 2 O 7/ H 2 SO 4, KMn. O 4 and Cu/250 o. C b)2 nd alcohol oxidize to give ketone c) Tertiary alcohols do not oxidize Iodoform test This reaction occurs with alcohols containing Also aldehydes or ketones containing CH 3 CO can give iodoform

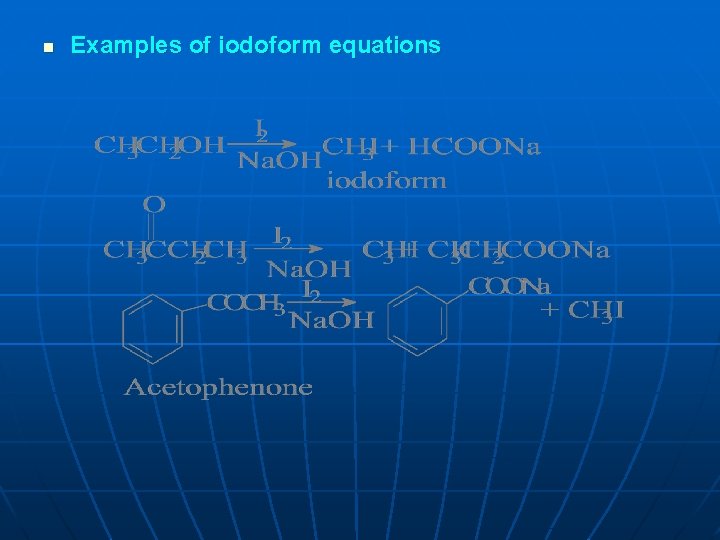
n Examples of iodoform equations

n Mechanism of iodoform n (a) For alcohols

n n n (b) For aldehyde and ketones Only one aldehyde can give iodoform, this aldehyde is acetaldehyde, the mechanism of aldehyde and ketones are step 2, and 3 in alcohol. E. g. acetone

n n Ethers Nomenclature of ethers IUPAC nomenclature ﻓﺮﻉ ﻓﻲ ﺳﻠﺴﻠﺔ ﺍﻻﻟﻜﺎﻥ RO ﻭ ﺗﺘﻢ ﺍﻟﺘﺴﻤﻴﺔ ﺑﺎﻋﺘﺒﺎﺭ Alkoxy ﺏ RO ﺗﺴﻤﻰ E. g. CH 3 OMethoxy CH 3 CH 2 OEthoxy CH 3 CH 2 CH 2 OButoxy Examples of ethers CH 3 OCH 2 CH 3 1 -Methoxypropane CH 3 CH 2 OCH 2 CH 3 Ethoxyethane n

n Comman Nomenclature ether ﻧﺬﻛﺮ ﺃﺴﻤﺎﺀ ﺍﻷﻠﻜﻴﻞ ﺍﻟﻤﺘﺼﻠﺔ ﺑﺬﺭﺓ ﺍﻷﻮﻛﺴﺠﻴﻦ ﺛﻢ ﻧﻜﺘﺐ ﻛﻠﻤﺔ n Examples CH 3 CH 2 OCH 2 CH 3 C 6 H 5 OCH 3 (CH 3) 3 C-OCH 2 CH 3 Diethyl ether methyl phenyl ether t-Butyl ether (Anisole) n

Preparation of ethers n From alcohols Wiliamson synthesis It is reaction between alkoside and alkyl halide E. g. preparation of diethyl ether

Question n Show could you prepare the following ethers: (a) Anisole (phenyl methyl ether) Answer (b) t butyl ethyl et

n Observation: Alkyl halide must be primary or secondary because t alkyl halide undergoes loss of HCl (elimination) so it gives alkene not ether e. g.

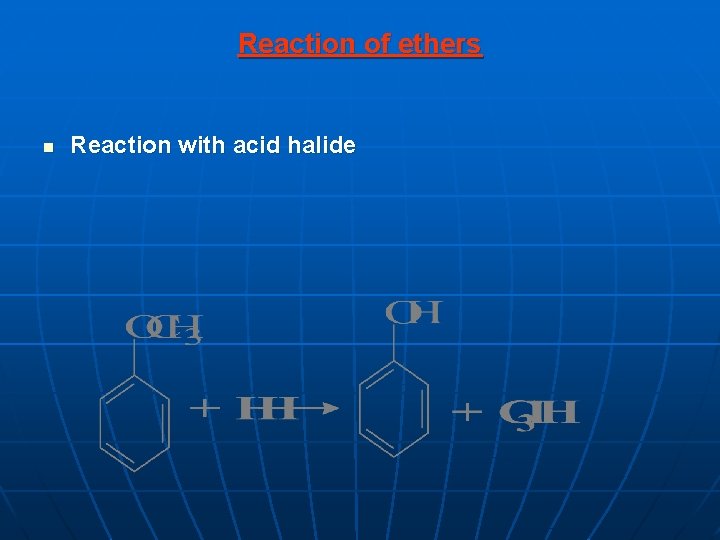
Reaction of ethers n Reaction with acid halide


Preparation of cyclic ethers In case of unsymmetrical cyclic ethers, the product depends on the reaction condition In case of acidic medium, nucleophile goes to more steric carbon i. e. carbon containing less number of hydrogen. In case of basic medium, nucleophile goes to less steric carbon i. e. carbon containing great number of hydrogen.

Examples