Example of Tertiary and Quaternary Structure of Protein


![Bohr effect • ↑[H+] – protonation of N terminal in Hb • Create a Bohr effect • ↑[H+] – protonation of N terminal in Hb • Create a](https://slidetodoc.com/presentation_image_h/d02e4a82f44065503a4df69897dee593/image-12.jpg)
- Slides: 14

Example of Tertiary and Quaternary Structure of Protein Myoglobin and Hemoglobin

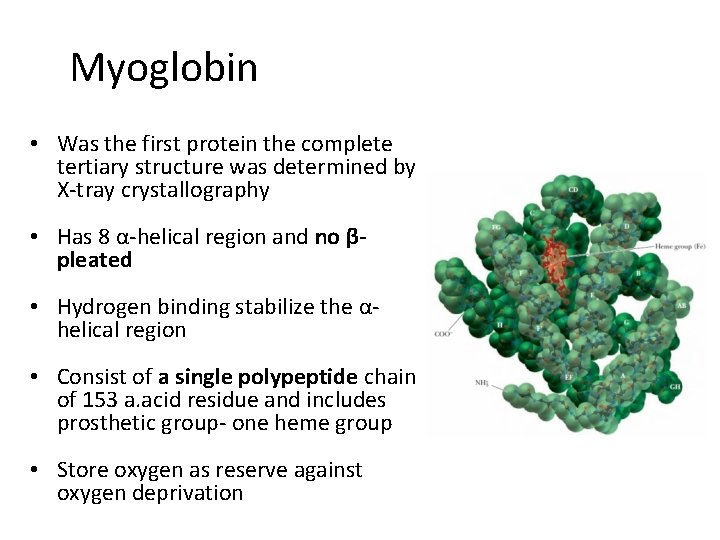
Myoglobin • Was the first protein the complete tertiary structure was determined by X-tray crystallography • Has 8 α-helical region and no βpleated • Hydrogen binding stabilize the αhelical region • Consist of a single polypeptide chain of 153 a. acid residue and includes prosthetic group- one heme group • Store oxygen as reserve against oxygen deprivation

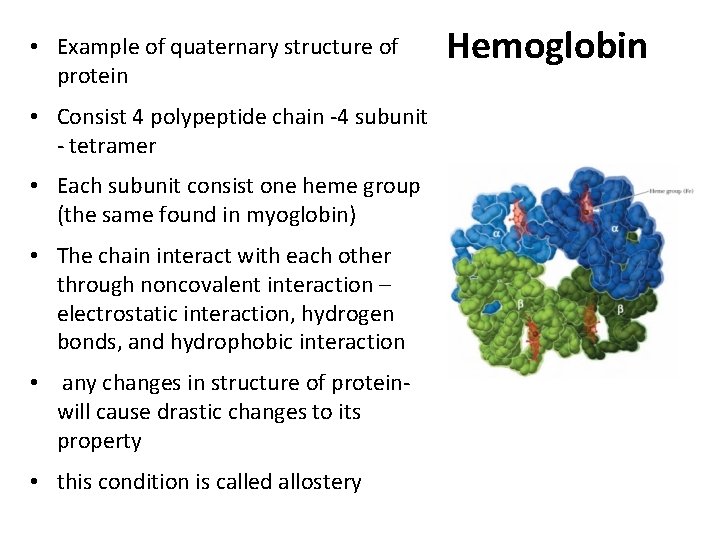
• Example of quaternary structure of protein • Consist 4 polypeptide chain -4 subunit - tetramer • Each subunit consist one heme group (the same found in myoglobin) • The chain interact with each other through noncovalent interaction – electrostatic interaction, hydrogen bonds, and hydrophobic interaction • any changes in structure of proteinwill cause drastic changes to its property • this condition is called allostery Hemoglobin

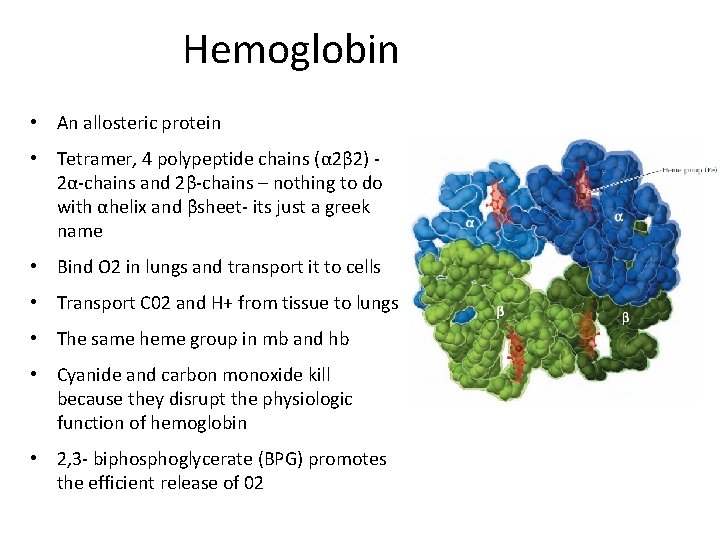
Hemoglobin • An allosteric protein • Tetramer, 4 polypeptide chains (α 2β 2) 2α-chains and 2β-chains – nothing to do with αhelix and βsheet- its just a greek name • Bind O 2 in lungs and transport it to cells • Transport C 02 and H+ from tissue to lungs • The same heme group in mb and hb • Cyanide and carbon monoxide kill because they disrupt the physiologic function of hemoglobin • 2, 3 - biphosphoglycerate (BPG) promotes the efficient release of 02

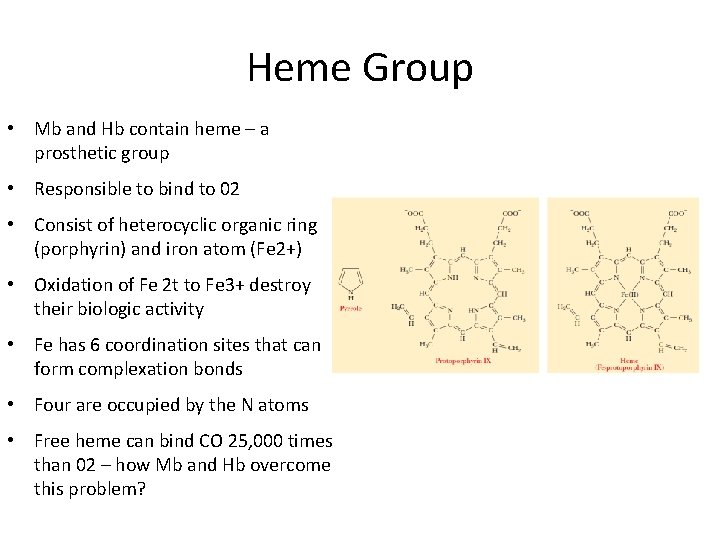
Heme Group • Mb and Hb contain heme – a prosthetic group • Responsible to bind to 02 • Consist of heterocyclic organic ring (porphyrin) and iron atom (Fe 2+) • Oxidation of Fe 2 t to Fe 3+ destroy their biologic activity • Fe has 6 coordination sites that can form complexation bonds • Four are occupied by the N atoms • Free heme can bind CO 25, 000 times than 02 – how Mb and Hb overcome this problem?

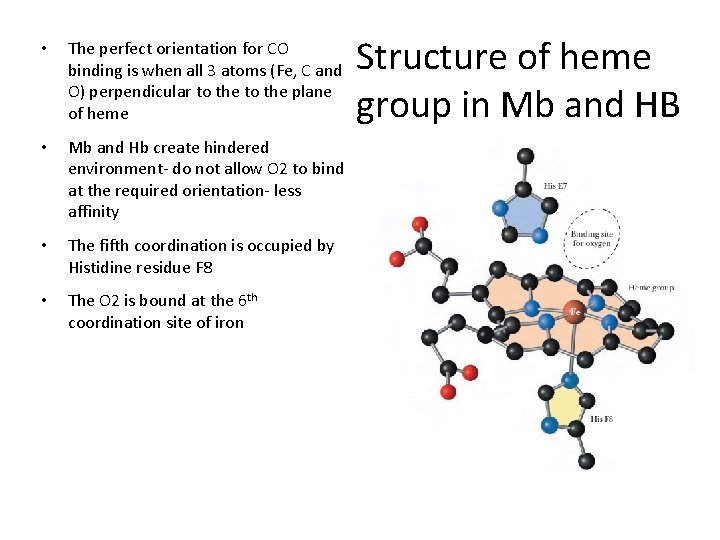
• The perfect orientation for CO binding is when all 3 atoms (Fe, C and O) perpendicular to the plane of heme • Mb and Hb create hindered environment- do not allow O 2 to bind at the required orientation- less affinity • The fifth coordination is occupied by Histidine residue F 8 • The O 2 is bound at the 6 th coordination site of iron Structure of heme group in Mb and HB

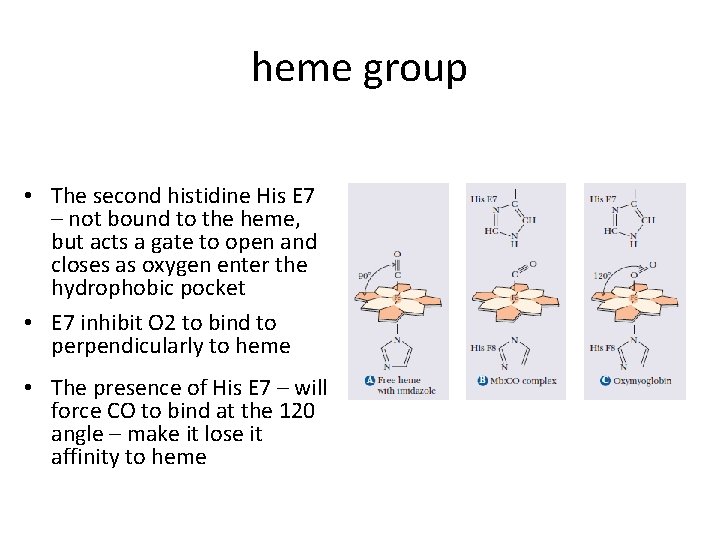
heme group • The second histidine His E 7 – not bound to the heme, but acts a gate to open and closes as oxygen enter the hydrophobic pocket • E 7 inhibit O 2 to bind to perpendicularly to heme • The presence of His E 7 – will force CO to bind at the 120 angle – make it lose it affinity to heme

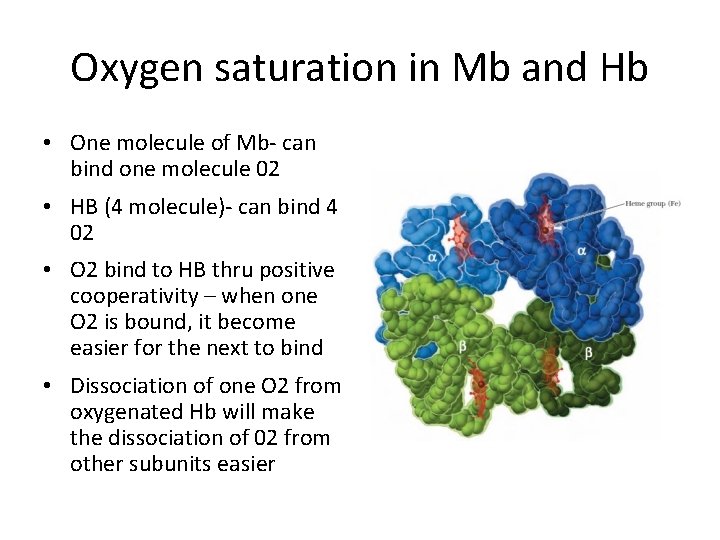
Oxygen saturation in Mb and Hb • One molecule of Mb- can bind one molecule 02 • HB (4 molecule)- can bind 4 02 • O 2 bind to HB thru positive cooperativity – when one O 2 is bound, it become easier for the next to bind • Dissociation of one O 2 from oxygenated Hb will make the dissociation of 02 from other subunits easier

Different form of HB • Hb is bound to 02 - oxyhemoglobin – relaxed (R state) • Without 02 – deoxyhb – tense (T) state • If Fe 2+ is oxidized to Fe 3+ - unable to bind 02 methemoglobin • C 0 and NO have higher affinity for heme FE 2+ than 02 - toxicity

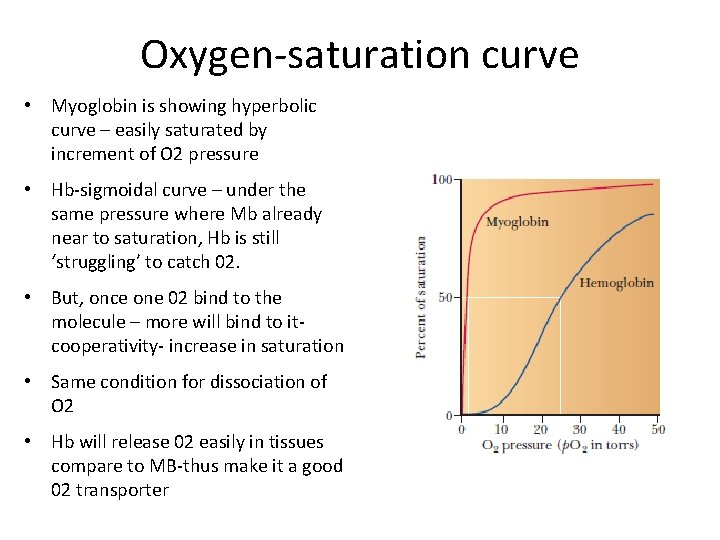
Oxygen-saturation curve • Myoglobin is showing hyperbolic curve – easily saturated by increment of O 2 pressure • Hb-sigmoidal curve – under the same pressure where Mb already near to saturation, Hb is still ‘struggling’ to catch 02. • But, once one 02 bind to the molecule – more will bind to itcooperativity- increase in saturation • Same condition for dissociation of O 2 • Hb will release 02 easily in tissues compare to MB-thus make it a good 02 transporter

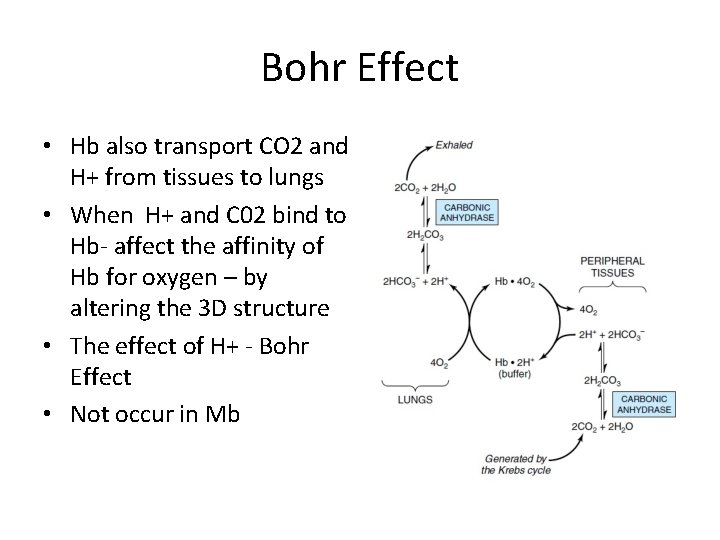
Bohr Effect • Hb also transport CO 2 and H+ from tissues to lungs • When H+ and C 02 bind to Hb- affect the affinity of Hb for oxygen – by altering the 3 D structure • The effect of H+ - Bohr Effect • Not occur in Mb
![Bohr effect H protonation of N terminal in Hb Create a Bohr effect • ↑[H+] – protonation of N terminal in Hb • Create a](https://slidetodoc.com/presentation_image_h/d02e4a82f44065503a4df69897dee593/image-12.jpg)
Bohr effect • ↑[H+] – protonation of N terminal in Hb • Create a salt bridge • Low affinity of Hb to O 2 • Metabolically active tissues need more 02 - they generate more C 02 and H+ which causes hemoglobin to release its 02 • C 02 produced in metabolism are in the form of H 2 CO 3→ HCO 3 - and H+ • HC 03 - is transported to lungs and combined with H+→ C 02 – exhaled • This process allow fine tuning Ph and level of C 02 and 02

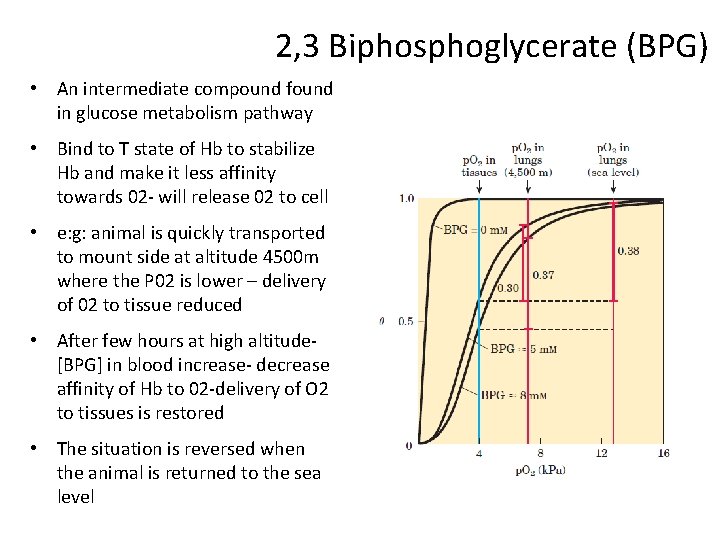
2, 3 Biphosphoglycerate (BPG) • An intermediate compound found in glucose metabolism pathway • Bind to T state of Hb to stabilize Hb and make it less affinity towards 02 - will release 02 to cell • e: g: animal is quickly transported to mount side at altitude 4500 m where the P 02 is lower – delivery of 02 to tissue reduced • After few hours at high altitude[BPG] in blood increase- decrease affinity of Hb to 02 -delivery of O 2 to tissues is restored • The situation is reversed when the animal is returned to the sea level

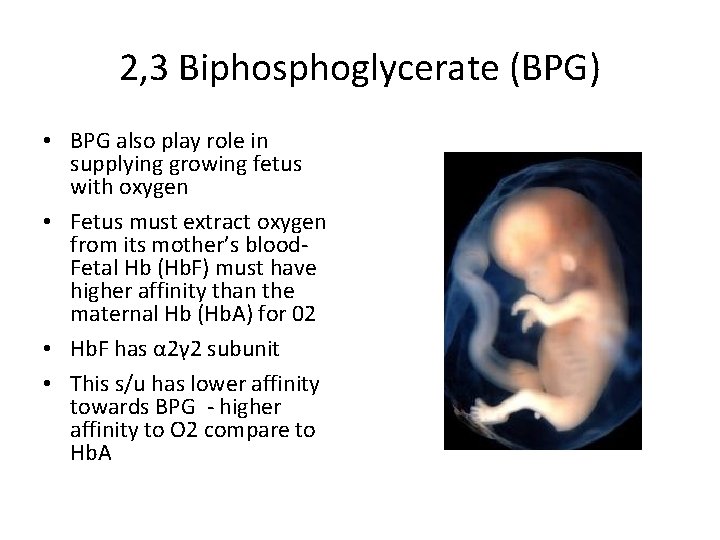
2, 3 Biphosphoglycerate (BPG) • BPG also play role in supplying growing fetus with oxygen • Fetus must extract oxygen from its mother’s blood. Fetal Hb (Hb. F) must have higher affinity than the maternal Hb (Hb. A) for 02 • Hb. F has α 2γ 2 subunit • This s/u has lower affinity towards BPG - higher affinity to O 2 compare to Hb. A