EXAMPLE NaMe COO3 BH Other Ways to Prepare
EXAMPLE Na(Me. COO)3 BH

Other Ways to Prepare Primary amines REDUCTION OF NITROANILINES - as before REDUCTION OF NITRILES RCH 2 NH SN 2 conditions essentially replaces halogen with a CH 2 NH 2 group. Increases carbon chain length by one carbon
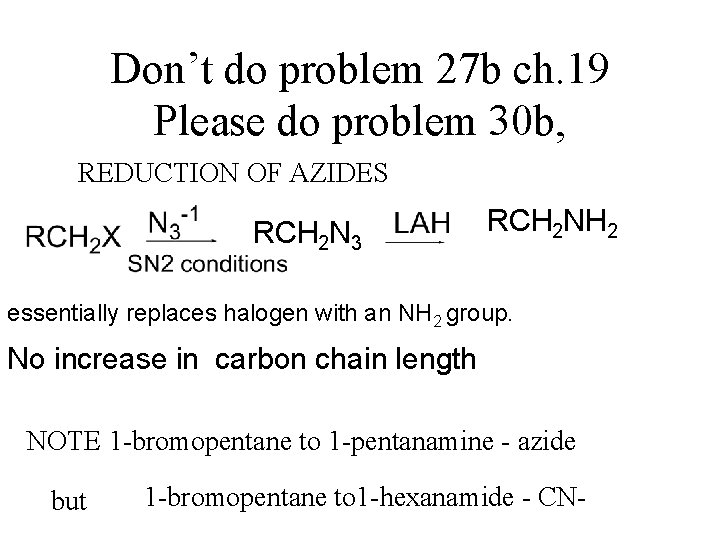
Don’t do problem 27 b ch. 19 Please do problem 30 b, REDUCTION OF AZIDES RCH 2 N 3 RCH 2 NH 2 essentially replaces halogen with an NH 2 group. No increase in carbon chain length NOTE 1 -bromopentane to 1 -pentanamine - azide but 1 -bromopentane to 1 -hexanamide - CN-
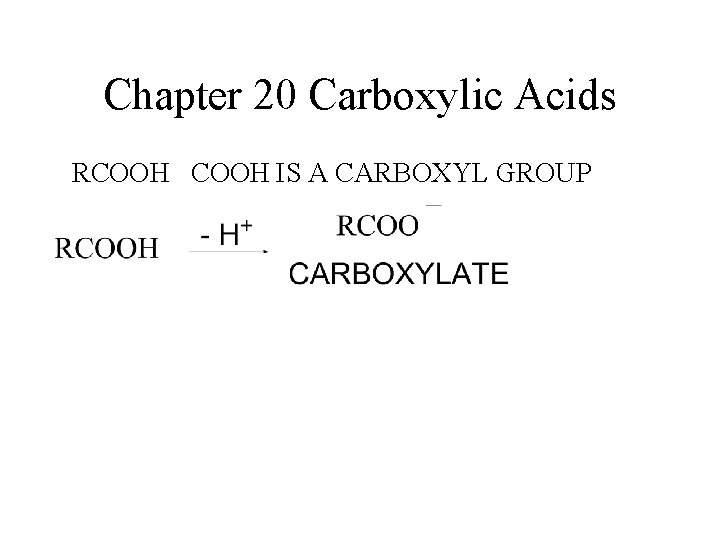
Chapter 20 Carboxylic Acids RCOOH IS A CARBOXYL GROUP
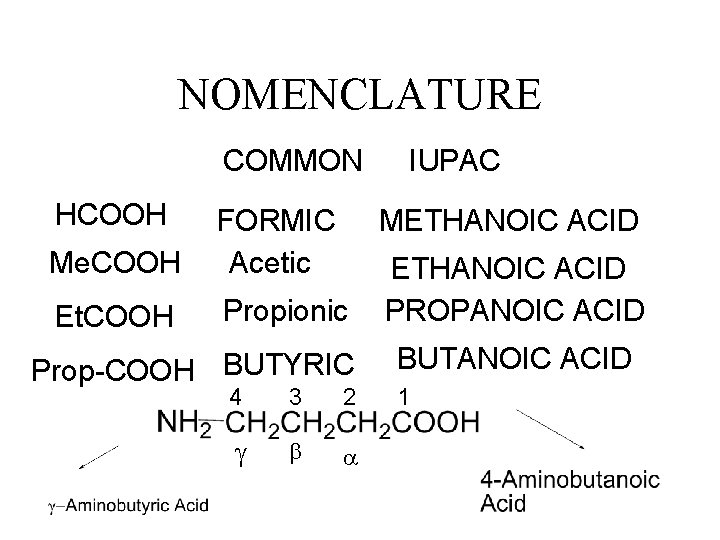
NOMENCLATURE COMMON HCOOH Me. COOH FORMIC Acetic Et. COOH Propionic IUPAC METHANOIC ACID Prop-COOH BUTYRIC 4 3 2 g b a ETHANOIC ACID PROPANOIC ACID BUTANOIC ACID 1

Nomenclature of Dibasic Acids HOOC-COOH HOOCCH 2 COOH Oxalic acid Malonic acid HOOC(CH 2)2 COOH Succinic acid HOOC(CH 2)3 COOH Glutaric acid HOOC(CH 2)4 COOH Adipic acid HOOC(CH 2)5 COOH Pimelic acid
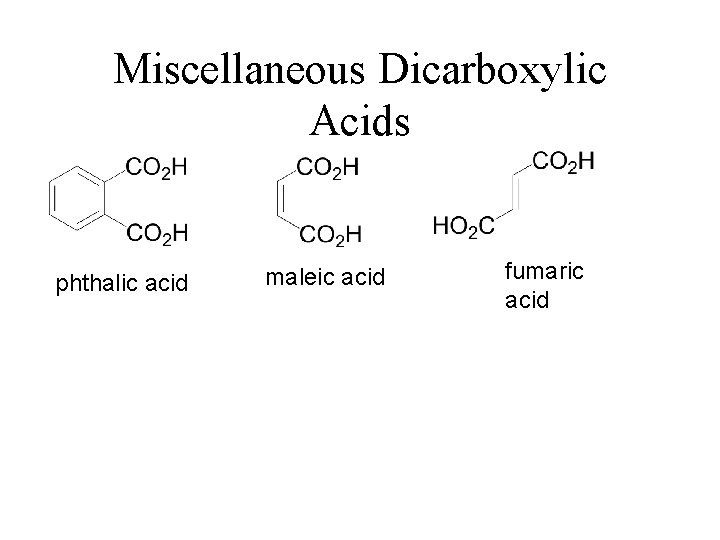
Miscellaneous Dicarboxylic Acids phthalic acid maleic acid fumaric acid
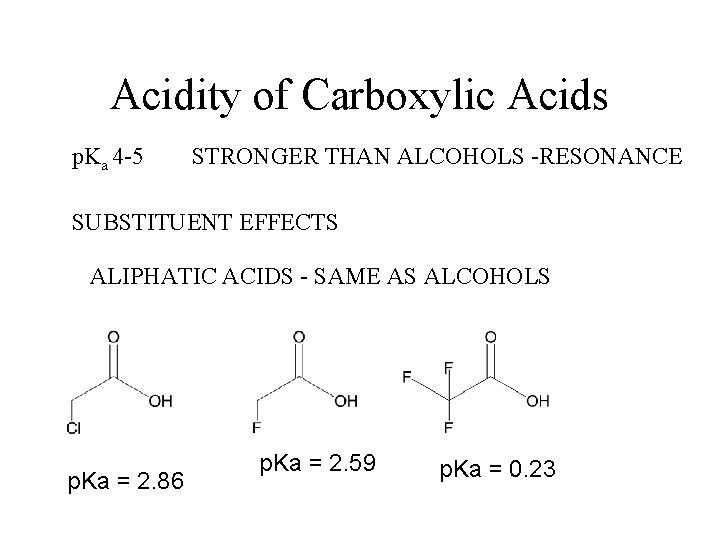
Acidity of Carboxylic Acids p. Ka 4 -5 STRONGER THAN ALCOHOLS -RESONANCE SUBSTITUENT EFFECTS ALIPHATIC ACIDS - SAME AS ALCOHOLS p. Ka = 2. 86 p. Ka = 2. 59 p. Ka = 0. 23
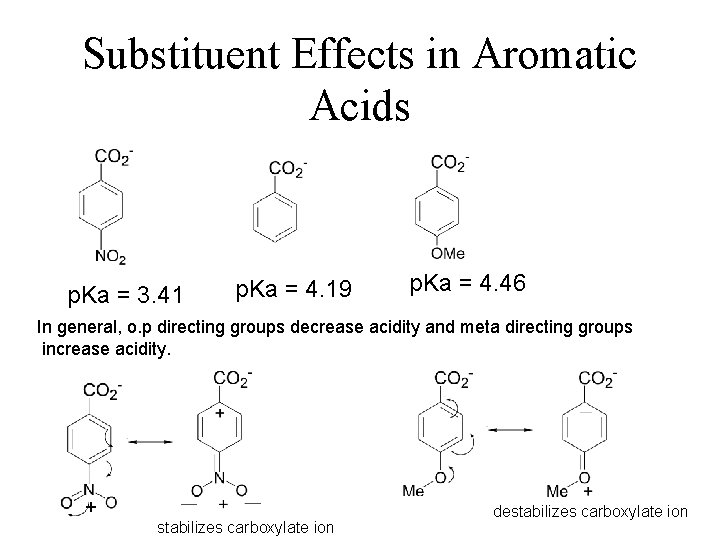
Substituent Effects in Aromatic Acids p. Ka = 3. 41 p. Ka = 4. 19 p. Ka = 4. 46 In general, o. p directing groups decrease acidity and meta directing groups increase acidity. + stabilizes carboxylate ion destabilizes carboxylate ion

Salts of Carboxylates FORMATION OF CARBOXYLATES RECALL - C 6 H 5 OH DOES NOT REACT WITH Na. HCO 3
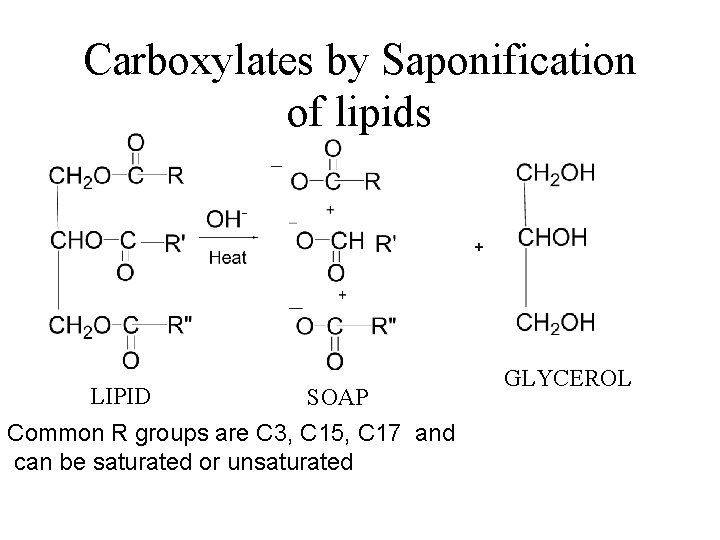
Carboxylates by Saponification of lipids LIPID SOAP Common R groups are C 3, C 15, C 17 and can be saturated or unsaturated GLYCEROL

HYDROGENATION OF OILS MARGARINE In clinical studies, trans fatty acids or hydrogenated fats tend to raise total blood cholesterol levels and LDL ("bad") cholesterol and lower HDL (“GOOD”) cholesterol when used instead of cis fatty acids or natural oils. These changes may increase the risk of heart disease.

Spectroscopy IR - Broad OH 3300 -2800 cm. MS Mc. Lafferty if delta hydrogen present 1 -H NMR delta around 12 -13 ppm 13 -H NMR delta around 200 ppm
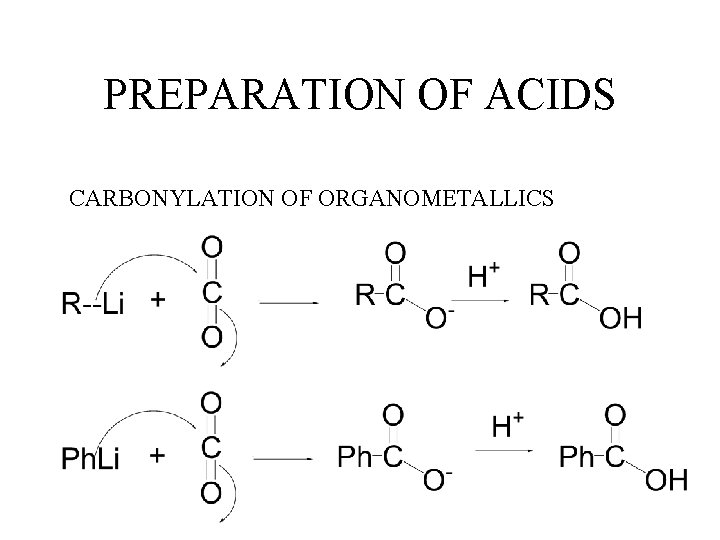
PREPARATION OF ACIDS CARBONYLATION OF ORGANOMETALLICS
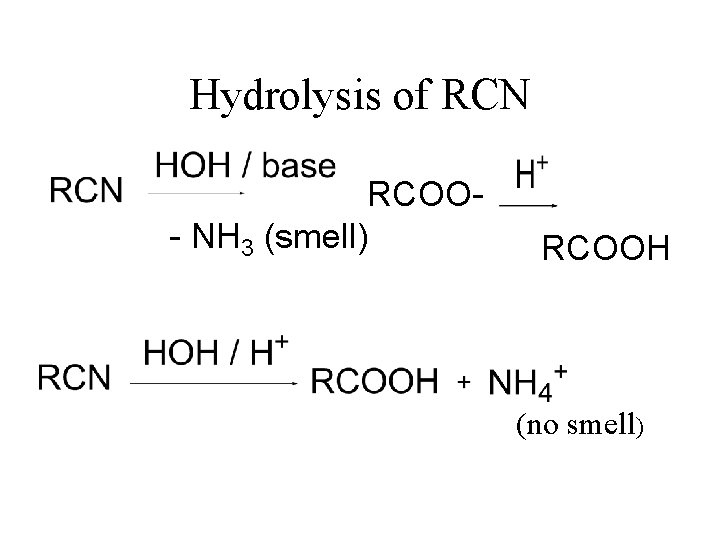
Hydrolysis of RCN RCOO- NH 3 (smell) RCOOH (no smell)


Basicity of amines The greater the availability of the lone pair electrons on nitrogen, the greater the base. In the old days, p. Kb was a measure of base strength. Kb = [RNH 3+] [OH-] / RNH 2 p. Kb = - log Kb The stronger the base the lower the p. Kb EFFECTS ON AMINE BASICITY 1. INDUCTIVE EFFECT - ALKYL SUBSTITUTION METHYL GROUP INCREASES ELECTRON DENSITY ON N 3. 36 3. 28 2 METHYLS ARE BETTER THAN ONE WATCH OUT THREE METHYL GROUPS DECREASES BASICITY p. Kb = 4. 26 - Steric inhibition of solvation of HOH with the NH+ of the R 3 NH+ cation.
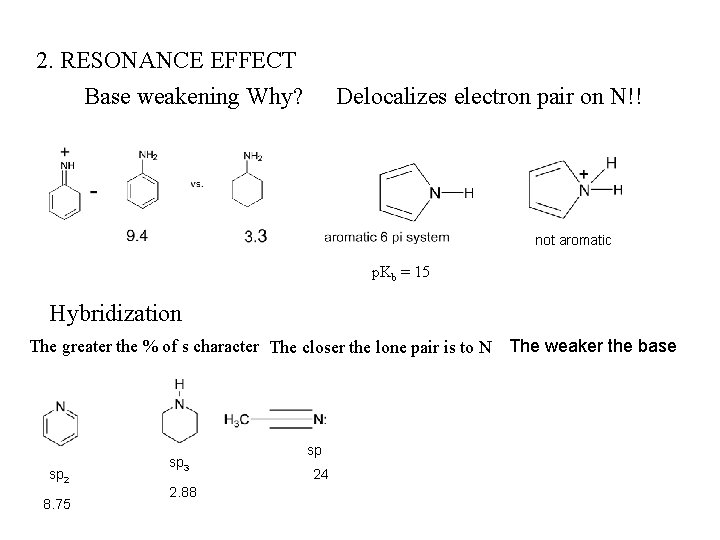
2. RESONANCE EFFECT Base weakening Why? Delocalizes electron pair on N!! not aromatic p. Kb = 15 Hybridization The greater the % of s character The closer the lone pair is to N The weaker the base sp 2 8. 75 sp 3 2. 88 sp 24

REACTIONS OF AMINES Alkylation of amines by alkyl halides - Only two situations of importance Excess ammonia - stops are monoalkylation stage Ph. CH 2 Br + excess NH 3 Ph. CH 2 NH 2 Excess Methyl iodide - all the way to quaternary salt Propyl-NH 2 + ex Me. I Propyl-N(Me)3+
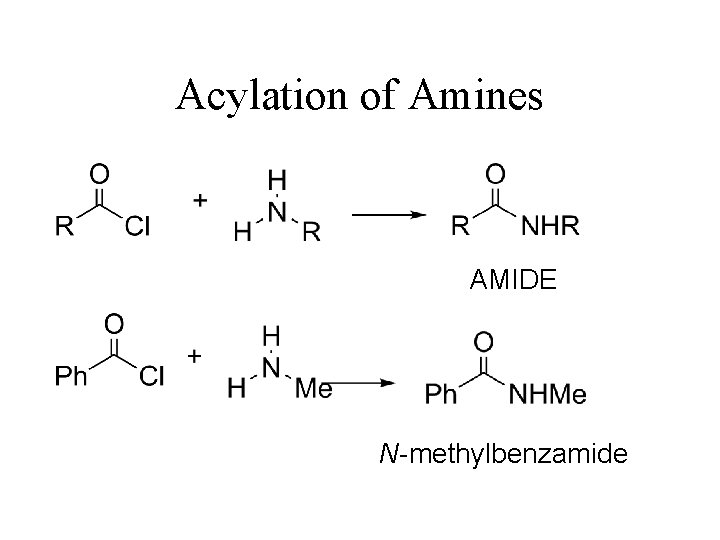
Acylation of Amines AMIDE N-methylbenzamide
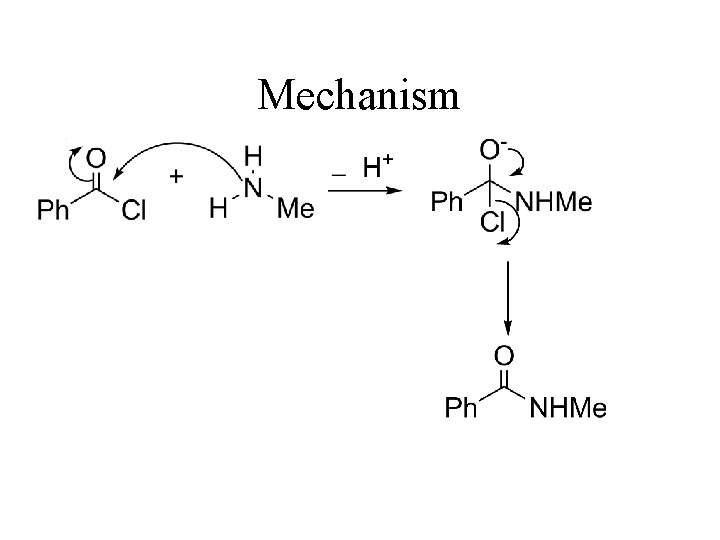
Mechanism

Amines as Leaving Groups Hofmann Elimination HEAT ANTI ELIMINATION - Less stable (less substituted) alkene
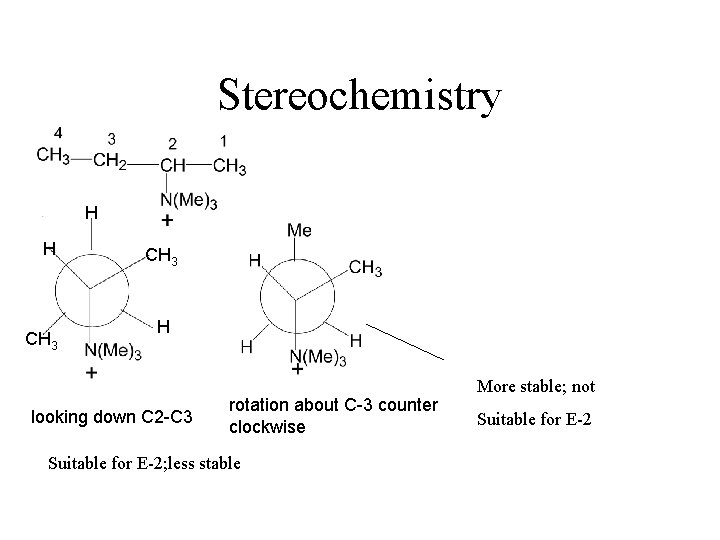
Stereochemistry H H CH 3 H looking down C 2 -C 3 rotation about C-3 counter clockwise Suitable for E-2; less stable More stable; not Suitable for E-2
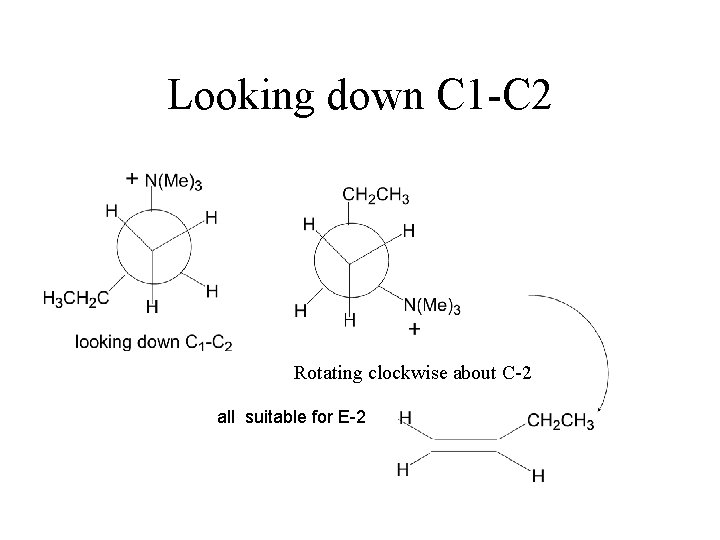
Looking down C 1 -C 2 Rotating clockwise about C-2 all suitable for E-2
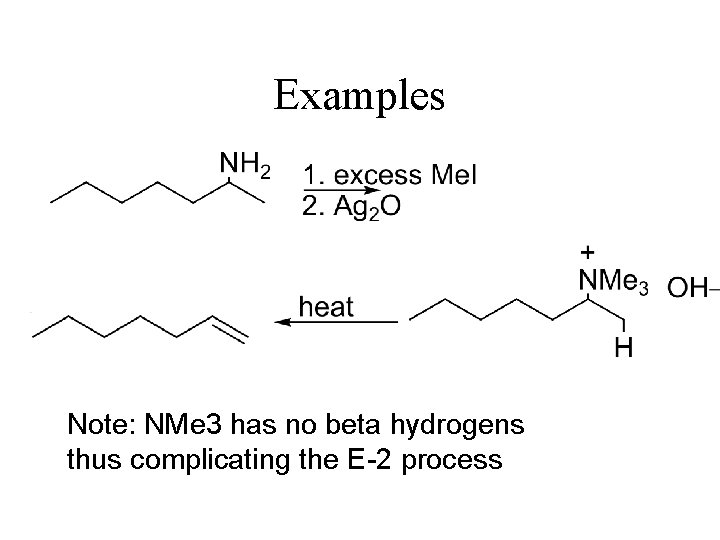
Examples Note: NMe 3 has no beta hydrogens thus complicating the E-2 process
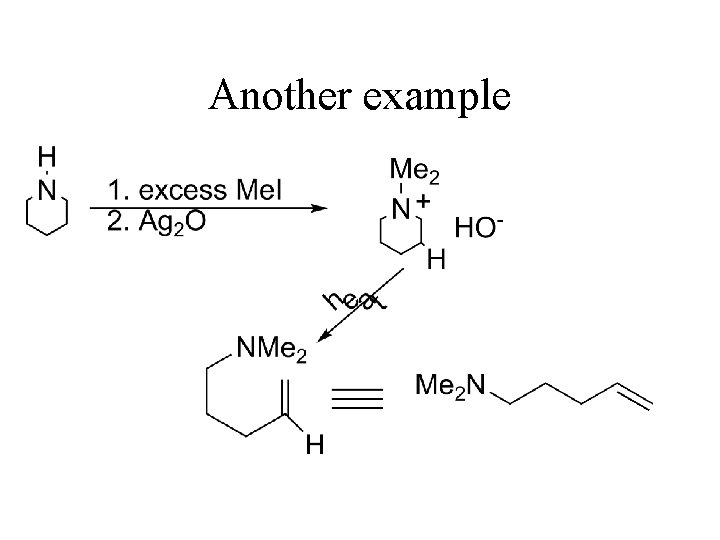
Another example
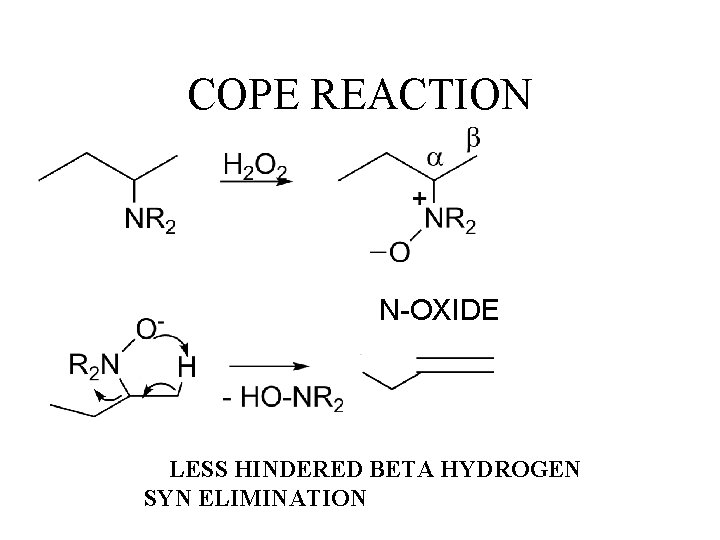
COPE REACTION N-OXIDE LESS HINDERED BETA HYDROGEN SYN ELIMINATION
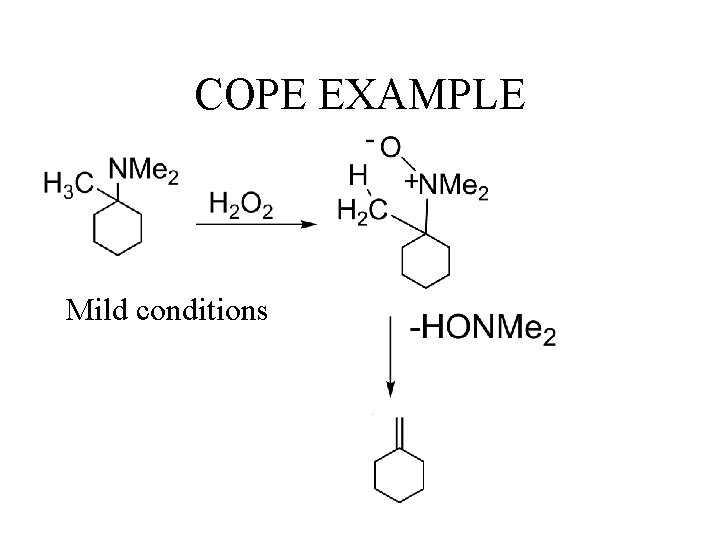
COPE EXAMPLE Mild conditions

Reaction of Primary Amines with HNO 2 Preparation of HNO 2 Na. NO 2 + HCl HNO 2 + Na. Cl
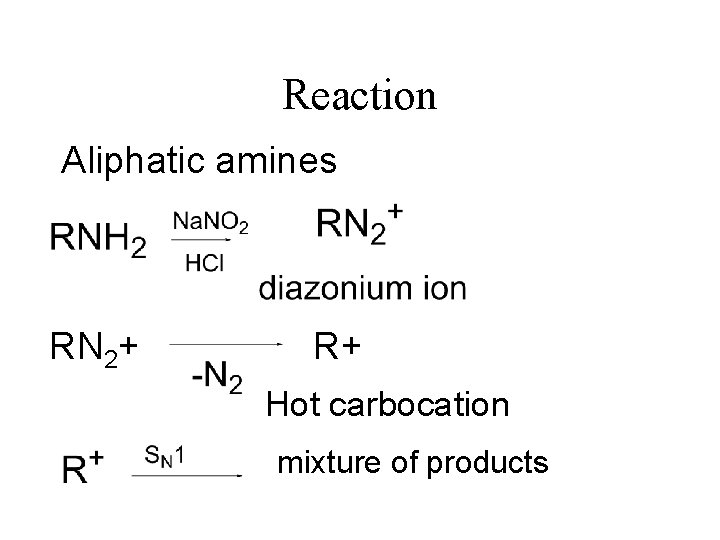
Reaction Aliphatic amines RN 2+ R+ Hot carbocation mixture of products

Aromatic Primary Amines Ar. N 2+ 0 - 5 o. C stable undergoes SN 1 with nucleophiles
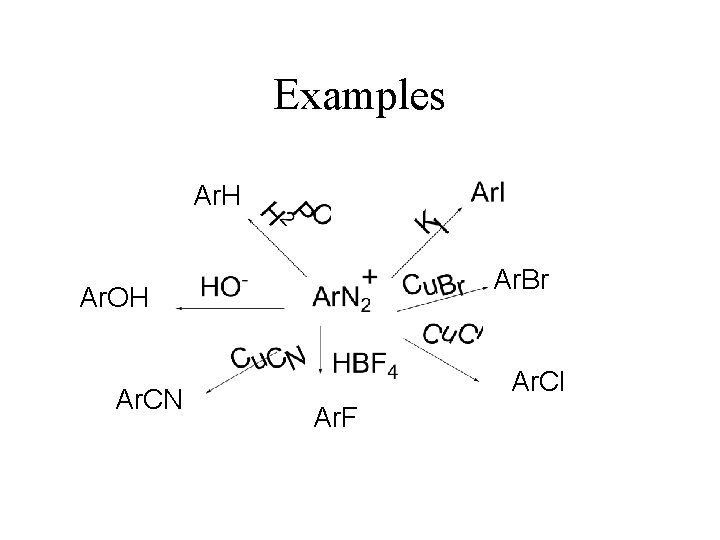
Examples Ar. H Ar. Br Ar. OH Ar. CN Ar. Cl Ar. F
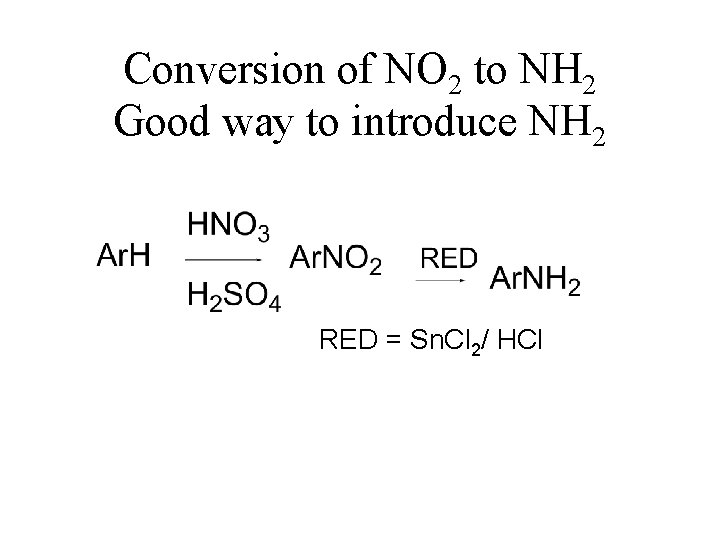
Conversion of NO 2 to NH 2 Good way to introduce NH 2 RED = Sn. Cl 2/ HCl
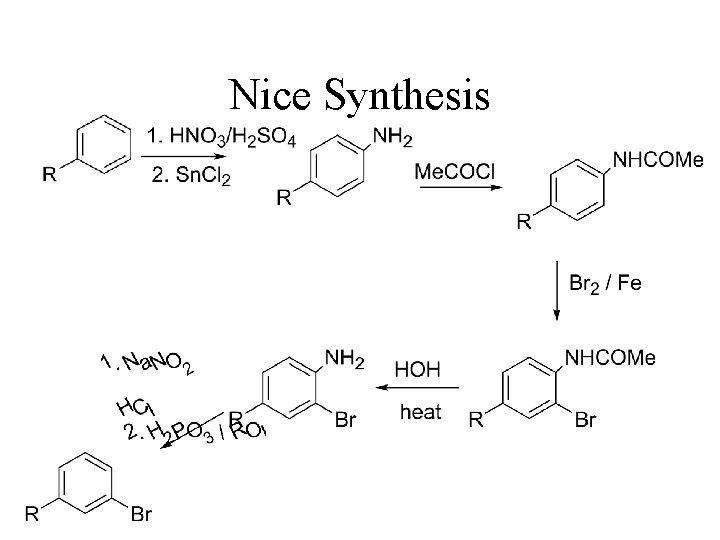
Nice Synthesis
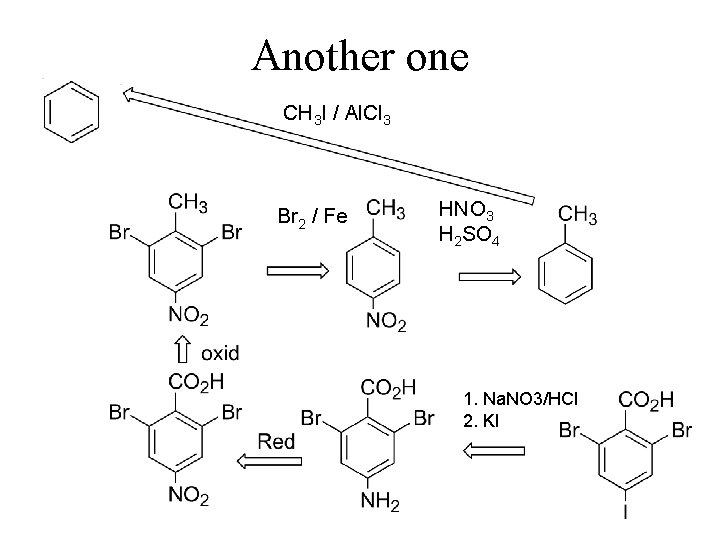
Another one CH 3 I / Al. Cl 3 Br 2 / Fe HNO 3 H 2 SO 4 1. Na. NO 3/HCl 2. KI
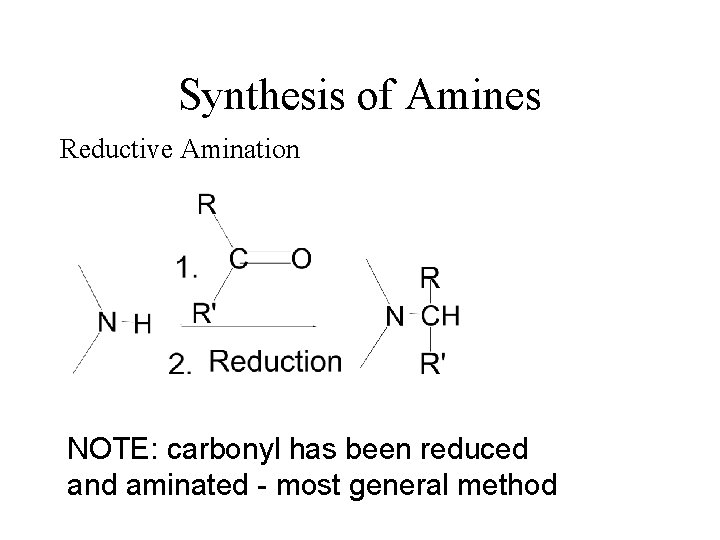
Synthesis of Amines Reductive Amination NOTE: carbonyl has been reduced and aminated - most general method
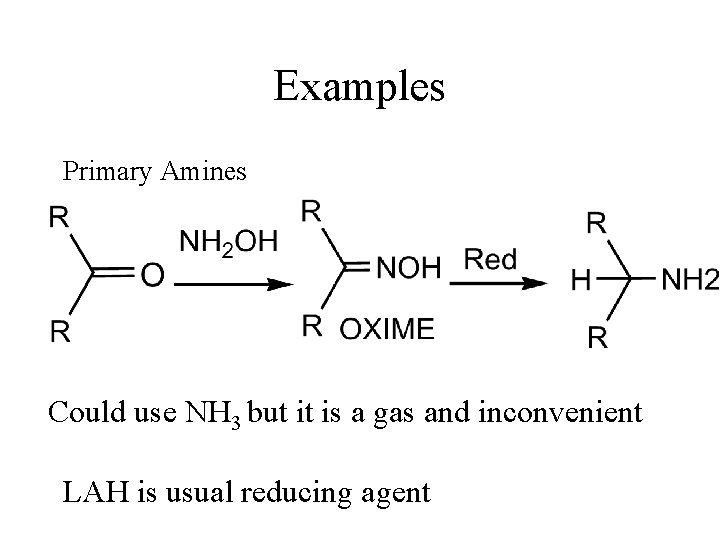
Examples Primary Amines Could use NH 3 but it is a gas and inconvenient LAH is usual reducing agent
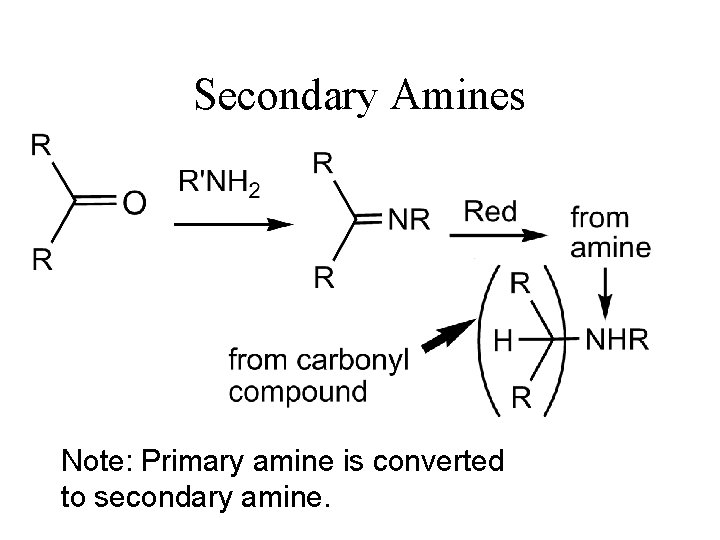
Secondary Amines Note: Primary amine is converted to secondary amine.
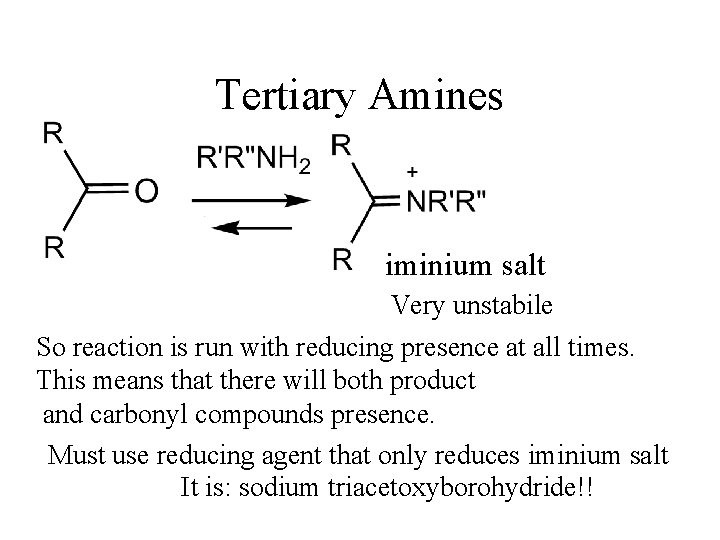
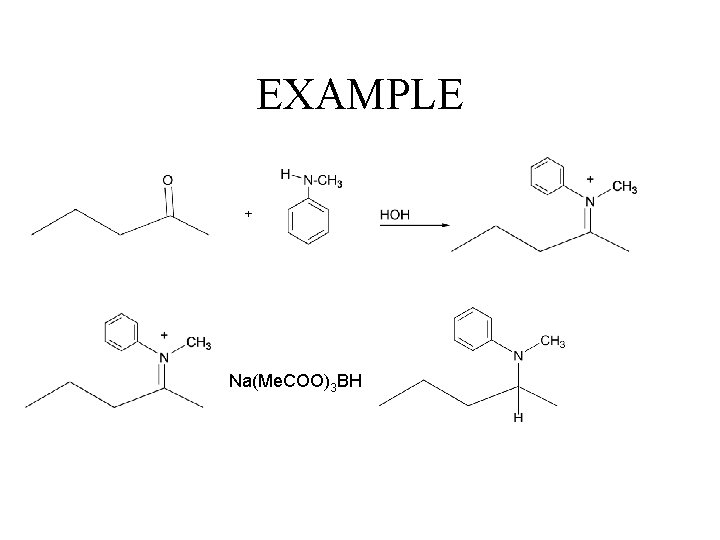
Tertiary Amines iminium salt Very unstabile So reaction is run with reducing presence at all times. This means that there will both product and carbonyl compounds presence. Must use reducing agent that only reduces iminium salt It is: sodium triacetoxyborohydride!!
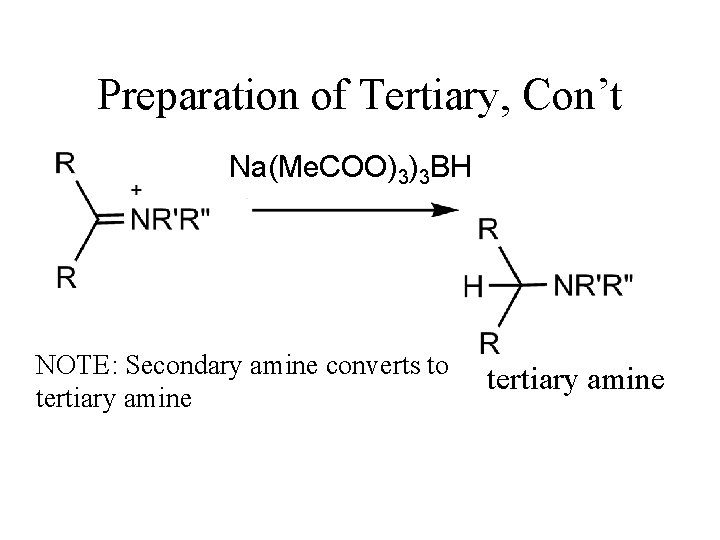
Preparation of Tertiary, Con’t Na(Me. COO)3)3 BH NOTE: Secondary amine converts to tertiary amine
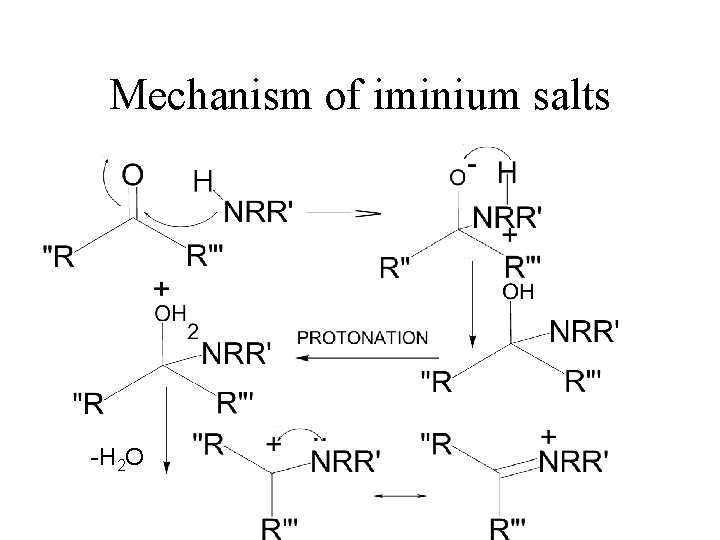
Mechanism of iminium salts -H 2 O
- Slides: 41