Example Exercise 9 1 Interpreting Chemical Equation Coefficients























- Slides: 32

Example Exercise 9. 1 Interpreting Chemical Equation Coefficients Given the chemical equation for the combustion of methane, CH 4, balance the equation and interpret the coefficients in terms of (a) moles and (b) liters: spark CH 4(g) + O 2(g) CO 2(g) + H 2 O(g) Solution The balanced chemical equation is CH 4(g) + 2 O 2(g) spark CO 2(g) + 2 H 2 O(g) a. The coefficients in the equation (1: 2: 1: 2) indicate the ratio of moles as well as molecules. 1 mol CH 4 + 2 mol O 2 1 mol CO 2 + 2 mol H 2 O b. The coefficients in the equation (1: 2: 1: 2) indicate the ratio of volumes of gases. If we express the volume in liters, we have 1 L CH 4 + 2 L O 2 1 L CO 2 + 2 L H 2 O Practice Exercise Given the chemical equation for the combustion of propane, C 3 H 8, balance the equation and interpret the coefficients in terms of (a) moles and (b) milliliters: C 3 H 8(g) + O 2(g) Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin spark CO 2(g) + H 2 O(g) © 2018 Pearson Education, Inc.

Example Exercise 9. 1 Interpreting Chemical Equation Coefficients Continued Answers: a. 1 mol C 3 H 8 + 5 mol O 2 b. 1 m. L C 3 H 8 + 5 m. L O 2 3 mol CO 2 + 4 mol H 2 O 3 m. L CO 2 + 4 m. L H 2 O Concept Exercise Which of the following is in the same ratio as the coefficients in a balanced equation: moles of gas, mass of gas, volume of gas? Answer: See Appendix G, 9. 1. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

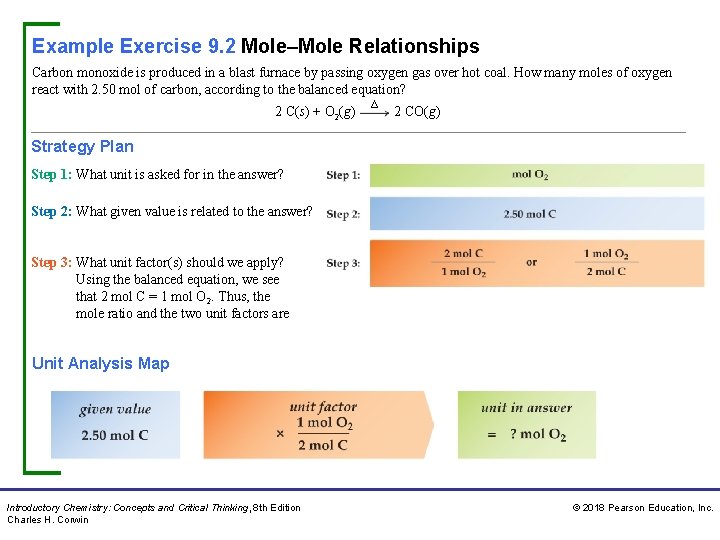
Example Exercise 9. 2 Mole–Mole Relationships Carbon monoxide is produced in a blast furnace by passing oxygen gas over hot coal. How many moles of oxygen react with 2. 50 mol of carbon, according to the balanced equation? 2 C(s) + O 2(g) ∆ 2 CO(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? Using the balanced equation, we see that 2 mol C = 1 mol O 2. Thus, the mole ratio and the two unit factors are Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 2 Mole–Mole Relationships Continued Solution We select the unit factor that cancels the unit in the given value Thus, Practice Exercise Iron is produced from iron ore in a blast furnace by passing carbon monoxide gas through molten iron(III) oxide. The balanced equation is Fe 2 O 3(l) + 3 CO(g) ∆ 2 Fe(l) + 3 CO 2(g) a. How many moles of carbon monoxide react with 2. 50 mol of Fe 2 O 3? b. How many moles of iron are produced from 2. 50 mol of Fe 2 O 3? Answers: a. 7. 50 mol CO b. 5. 00 mol Fe Concept Exercise How many unit factors are required to solve a mole–mole problem? Answer: See Appendix G, 9. 2. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 3 Classifying Stoichiometry Problems Classify the type of stoichiometry problem for each of the following: a. How many grams of Zn metal react with hydrochloric acid to give 0. 500 g of zinc chloride? b. How many liters of H 2 gas react with chlorine gas to yield 50. 0 cm 3 of hydrogen chloride gas? c. How many kilograms of Fe react with sulfuric acid to produce 50. 0 m. L of hydrogen gas? Solution After analyzing a problem for the unknown quantity and the relevant given value, we classify the type of problem. a. The problem asks for grams of Zn (mass) that react to give 0. 500 g of Zn. Cl 2 (mass). This a mass–mass type of problem. b. The problem asks for liters of H 2 gas (volume) that react to yield 50. 0 cm 3 of HCl gas (volume). This is a volume– volume type of problem. c. The problem asks for kilograms of Fe (mass) that react to produce 50. 0 m. L of H 2 gas (volume). This is a mass– volume type of problem. Practice Exercise Classify the type of stoichiometry problem for each of the following: a. How many grams of Hg. O decompose to give 0. 500 L of oxygen gas at STP? b. How many grams of Ag. Cl are produced from the reaction of 0. 500 g of solid sodium chloride with silver nitrate solution? c. How many milliliters of H 2 gas react with nitrogen gas to yield 1. 00 L of ammonia gas? Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 3 Classifying Stoichiometry Problems Continued Answers: a. mass–volume b. mass–mass c. volume–volume Concept Exercise What are three types of stoichiometry problems? Answers: See Appendix G, 9. 3. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

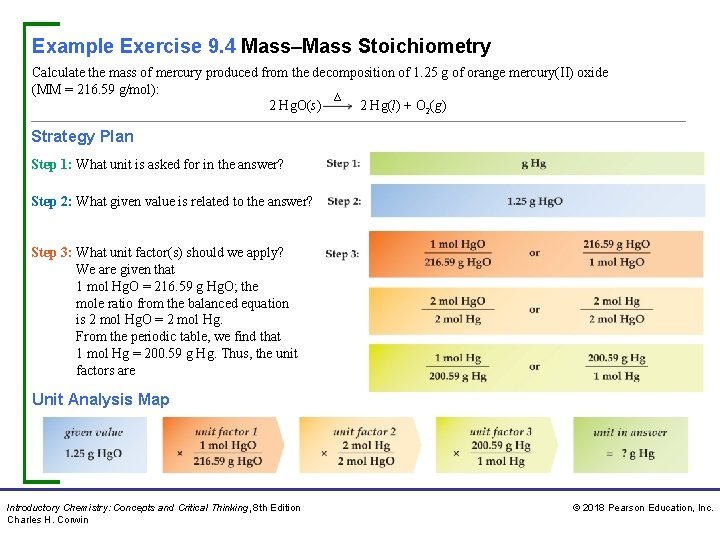
Example Exercise 9. 4 Mass–Mass Stoichiometry Calculate the mass of mercury produced from the decomposition of 1. 25 g of orange mercury(II) oxide (MM = 216. 59 g/mol): ∆ 2 Hg. O(s) 2 Hg(l) + O 2(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? We are given that 1 mol Hg. O = 216. 59 g Hg. O; the mole ratio from the balanced equation is 2 mol Hg. O = 2 mol Hg. From the periodic table, we find that 1 mol Hg = 200. 59 g Hg. Thus, the unit factors are Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 4 Mass–Mass Stoichiometry Continued Solution We select from each of the three pairs of ratios a unit factor that cancels the previous units. Thus, Practice Exercise Calculate the mass of carbon dioxide released from 10. 0 g of cobalt(III) carbonate given the unbalanced equation for the reaction: Co 2(CO 3) 3(s) ∆ Co 2 O 3(s) + CO 2(g) Answer: 4. 43 g CO 2 Concept Exercise What are three steps in the unit analysis method of problem solving? Answer: See Appendix G, 9. 4. Precipitation of Hgl 2 Potassium iodide and mercury(II) nitrate react to give an orange precipitate of Hgl 2. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

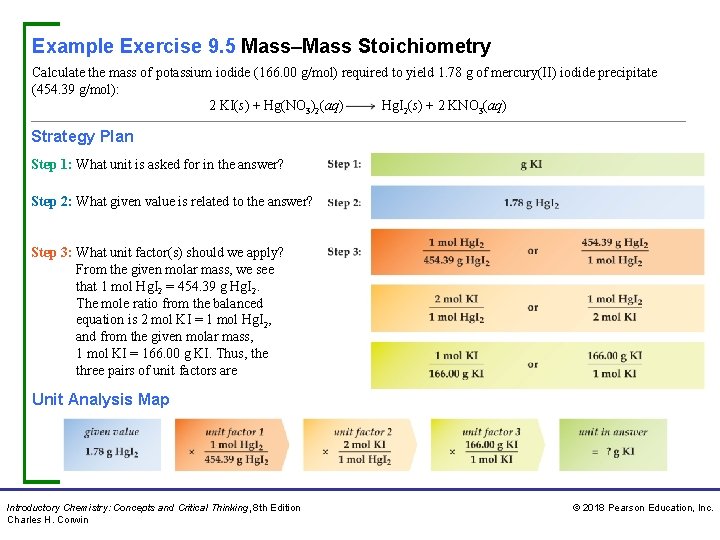
Example Exercise 9. 5 Mass–Mass Stoichiometry Calculate the mass of potassium iodide (166. 00 g/mol) required to yield 1. 78 g of mercury(II) iodide precipitate (454. 39 g/mol): 2 KI(s) + Hg(NO 3)2(aq) Hg. I 2(s) + 2 KNO 3(aq) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? From the given molar mass, we see that 1 mol Hg. I 2 = 454. 39 g Hg. I 2. The mole ratio from the balanced equation is 2 mol KI = 1 mol Hg. I 2, and from the given molar mass, 1 mol KI = 166. 00 g KI. Thus, the three pairs of unit factors are Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 5 Mass–Mass Stoichiometry Continued Solution We select from each of the three pairs of ratios a unit factor that cancels the previous units. Thus, Practice Exercise Calculate the mass of iron filings required to produce 0. 455 g of silver metal given the unbalanced equation for the reaction: Fe(s) + Ag. NO 3(aq) Fe(NO 3)3(aq) + Ag(s) Answer: 0. 0785 g Fe Concept Exercise In general, how many unit factors are required to solve a mass–mass stoichiometry problem? Answer: See Appendix G, 9. 5. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

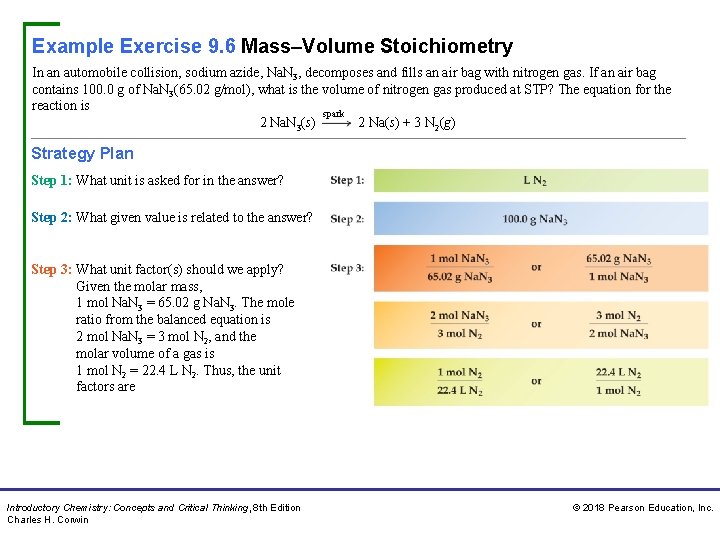
Example Exercise 9. 6 Mass–Volume Stoichiometry In an automobile collision, sodium azide, Na. N 3, decomposes and fills an air bag with nitrogen gas. If an air bag contains 100. 0 g of Na. N 3(65. 02 g/mol), what is the volume of nitrogen gas produced at STP? The equation for the reaction is spark 2 Na. N 3(s) 2 Na(s) + 3 N 2(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? Given the molar mass, 1 mol Na. N 3 = 65. 02 g Na. N 3. The mole ratio from the balanced equation is 2 mol Na. N 3 = 3 mol N 2, and the molar volume of a gas is 1 mol N 2 = 22. 4 L N 2. Thus, the unit factors are Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 6 Mass–Volume Stoichiometry Continued Unit Analysis Map Solution We select from each of the three pairs of ratios a unit factor that cancels the previous units. Thus, Practice Exercise Calculate the volume of hydrogen gas produced at STP from 1. 55 g of sodium metal in water, given the unbalanced equation for the reaction: Na(s) + H 2 O(l) Na. OH(aq) + H 2(g) Answer: 0. 755 L H 2 Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 6 Mass–Volume Stoichiometry Continued Concept Exercise What are three steps in the unit analysis method of problem solving? Answer: See Appendix G, 9. 6. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

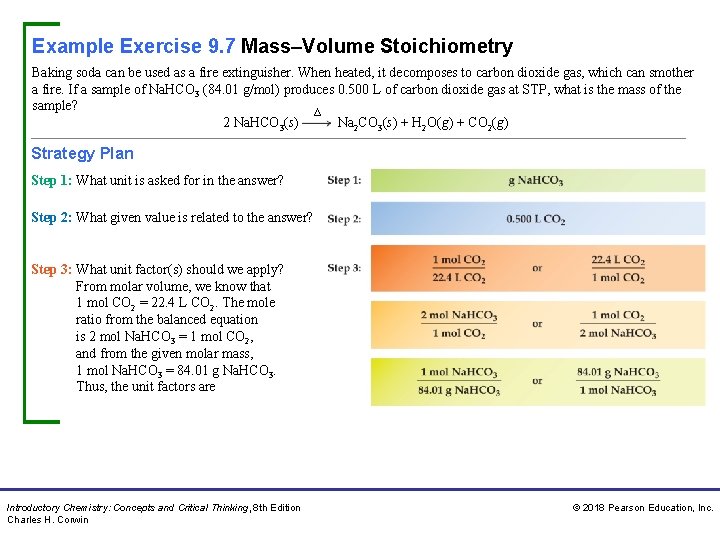
Example Exercise 9. 7 Mass–Volume Stoichiometry Baking soda can be used as a fire extinguisher. When heated, it decomposes to carbon dioxide gas, which can smother a fire. If a sample of Na. HCO 3 (84. 01 g/mol) produces 0. 500 L of carbon dioxide gas at STP, what is the mass of the sample? ∆ 2 Na. HCO 3(s) Na 2 CO 3(s) + H 2 O(g) + CO 2(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? From molar volume, we know that 1 mol CO 2 = 22. 4 L CO 2. The mole ratio from the balanced equation is 2 mol Na. HCO 3 = 1 mol CO 2, and from the given molar mass, 1 mol Na. HCO 3 = 84. 01 g Na. HCO 3. Thus, the unit factors are Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 7 Mass–Volume Stoichiometry Continued Unit Analysis Map Solution We select from each of the three pairs of ratios a unit factor that cancels the previous units. Thus, Practice Exercise Calculate the mass of aluminum metal required to release 2160 m. L of hydrogen gas at STP from sulfuric acid given the unbalanced equation for the reaction: Al(s) + H 2 SO 4(aq) Al 2(SO 4)3(aq) + H 2(g) Answer: 1. 73 g Al Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 7 Mass–Volume Stoichiometry Continued Concept Exercise In general, how many unit factors are required to solve a mass–volume stoichiometry problem? Answer: See Appendix G, 9. 7. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

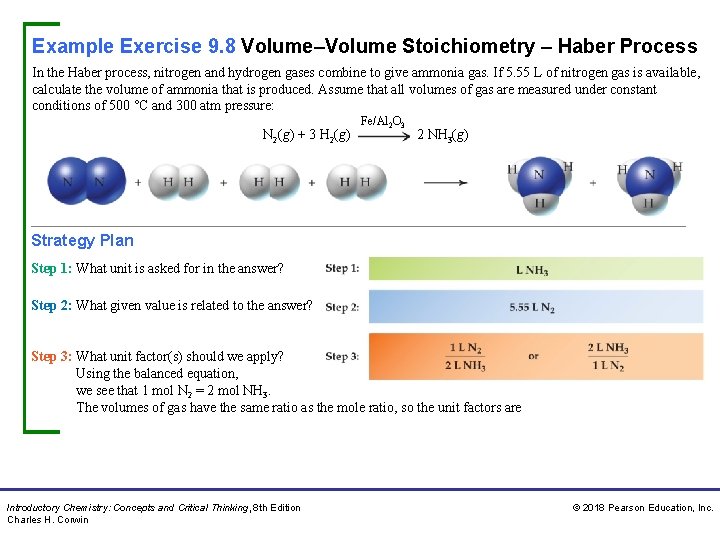
Example Exercise 9. 8 Volume–Volume Stoichiometry – Haber Process In the Haber process, nitrogen and hydrogen gases combine to give ammonia gas. If 5. 55 L of nitrogen gas is available, calculate the volume of ammonia that is produced. Assume that all volumes of gas are measured under constant conditions of 500 °C and 300 atm pressure: N 2(g) + 3 H 2(g) Fe/Al 2 O 3 2 NH 3(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? Using the balanced equation, we see that 1 mol N 2 = 2 mol NH 3. The volumes of gas have the same ratio as the mole ratio, so the unit factors are Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 8 Volume–Volume Stoichiometry – Haber Process Continued Unit Analysis Map Solution We select the unit factor that cancels the unit in the given value (L N 2). Thus, Practice Exercise Calculate the volumes of (a) hydrogen chloride gas and (b) oxygen gas that react to yield 40. 00 m. L of chlorine gas given the following unbalanced equation. Assume that all gases are at the same temperature and pressure. HCl(g) + O 2(g) Answer: a. 80. 0 m. L HCl b. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin Cl 2(g) + H 2 O(g) 20. 0 m. L O 2 © 2018 Pearson Education, Inc.

Example Exercise 9. 8 Volume–Volume Stoichiometry – Haber Process Continued Concept Exercise In general, how many unit factors are required to solve a volume–volume stoichiometry problem? Answer: See Appendix G, 9. 8. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

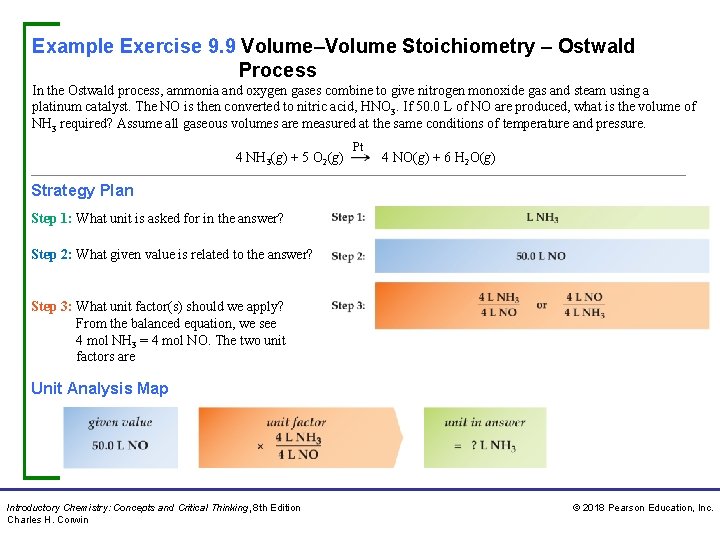
Example Exercise 9. 9 Volume–Volume Stoichiometry – Ostwald Process In the Ostwald process, ammonia and oxygen gases combine to give nitrogen monoxide gas and steam using a platinum catalyst. The NO is then converted to nitric acid, HNO 3. If 50. 0 L of NO are produced, what is the volume of NH 3 required? Assume all gaseous volumes are measured at the same conditions of temperature and pressure. 4 NH 3(g) + 5 O 2(g) Pt 4 NO(g) + 6 H 2 O(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? From the balanced equation, we see 4 mol NH 3 = 4 mol NO. The two unit factors are Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 9 Volume–Volume Stoichiometry – Ostwald Process Continued Solution We select the unit factor that cancels the unit in the given value . Thus, Practice Exercise Calculate the volumes of (a) nitrogen monoxide gas and (b) oxygen gas that react to yield 50. 0 m. L of nitrogen dioxide gas given the following unbalanced equation. Assume all gases are at the same temperature and pressure. NO(g) + O 2(g) Answers: a. 50. 0 m. L NO; NO 2(g) b. 25. 0 m. L O 2 Concept Exercise Why is only one unit factor usually required to solve a volume–volume stoichiometry problem? Answer: See Appendix G, 9. 9. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 10 Limiting Reactant Concept A 1. 00 mol sample of iron(II) oxide is heated with 1. 00 mol of aluminum metal and converted to molten iron. Identify the limiting reactant, and calculate the moles of iron produced in the so-called Thermite reaction: 3 Fe. O(l) + 2 Al(l) ∆ 3 Fe(l) + Al 2 O 3(s) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? From the balanced equation, we have the mole ratio 3 mol Fe. O/3 mol Fe, and 2 mol Al/3 mol Fe. Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 10 Limiting Reactant Concept Continued Solution We select the unit factor that cancels the unit in the given value . Thus, Second, we select the unit factor that cancels the unit in the given value Thus, Notice that Fe. O produces 1. 00 mol Fe, whereas Al produces 1. 50 mol Fe. Thus, Fe. O is the limiting reactant and the amount of product is 1. 00 mol Fe. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin . Thermite Reaction The thermite reaction of iron oxide and aluminum metal produces molten iron and a shower of sparks. © 2018 Pearson Education, Inc.

Example Exercise 9. 10 Limiting Reactant Concept Continued Practice Exercise A 5. 00 mol sample of iron(III) oxide is heated with 5. 00 mol aluminum metal and converted to molten iron. Identify the limiting reactant, and calculate the moles of iron produced given the unbalanced equation for the reaction: Fe 2 O 3(l) + Al(l) ∆ Fe(l) + Al 2 O 3(s) Answer: The limiting reactant is Al, which produces 5. 00 mol of Fe. Concept Exercise A Tour de France cyclist has 10 tires and 3 frames. How many complete bicycles can be assembled? Answer: See Appendix G, 9. 10. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 11 Mass–Mass Limiting Reactant In a reaction, 50. 0 g of manganese(IV) oxide reacts with 25. 0 g of aluminum. Identify the limiting reactant and calculate the mass of manganese metal produced. The equation for the reaction is 3 Mn. O 2(l) + 4 Al(l) ∆ 3 Mn(l) + 2 Al 2 O 3(s) Solution Step 1: We calculate the mass of Mn produced from 50. 0 g of Mn. O 2. We can outline the solution as follows: g Mn. O 2 mol Mn g Mn From the periodic table, we find the molar mass of Mn. O 2 is 86. 94 g/mol and Mn is 54. 94 g/mol. The unit analysis solution to the problem is Step 2: We calculate the mass of Mn produced from 25. 0 g of Al. We can outline the solution as follows: g Al mol Mn g Mn From the periodic table, we find that the molar mass of Al is 26. 98 g/mol. Starting with 25. 0 g of Al, the unit analysis solution is Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 11 Mass–Mass Limiting Reactant Continued Step 3: We compare the mass of product obtained from each of the reactants: 50. 0 g Mn. O 2 25. 0 g Al 31. 6 g Mn 38. 2 g Mn In this example, Mn. O 2 is the limiting reactant because it yields less product. Al is the excess reactant and is not completely consumed in the reaction. Thus, the maximum amount of product from the reaction is 31. 6 g of Mn. Practice Exercise In a reaction, 75. 0 g of manganese(IV) oxide react with 30. 0 g of aluminum. Identify the limiting reactant and calculate the mass of aluminum oxide produced: 3 Mn. O 2(l) + 4 Al(l) ∆ 3 Mn(l) + 2 Al 2 O 3(s) Answer: The limiting reactant is Al, which gives 56. 7 g of Al 2 O 3. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

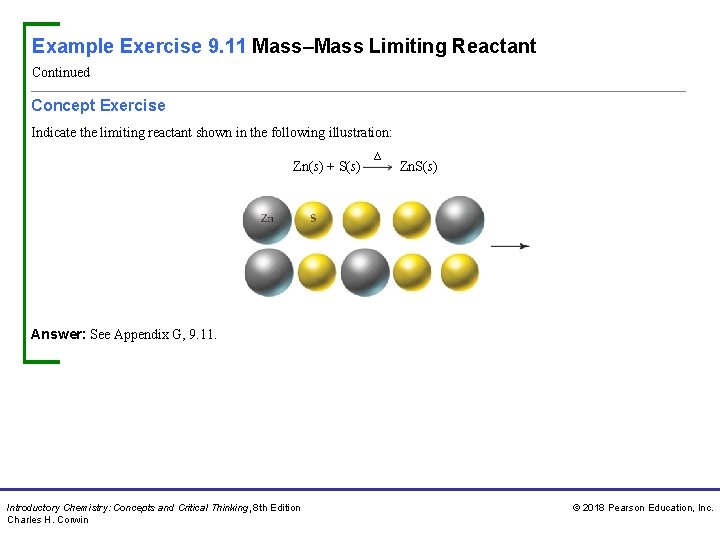
Example Exercise 9. 11 Mass–Mass Limiting Reactant Continued Concept Exercise Indicate the limiting reactant shown in the following illustration: Zn(s) + S(s) ∆ Zn. S(s) Answer: See Appendix G, 9. 11. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

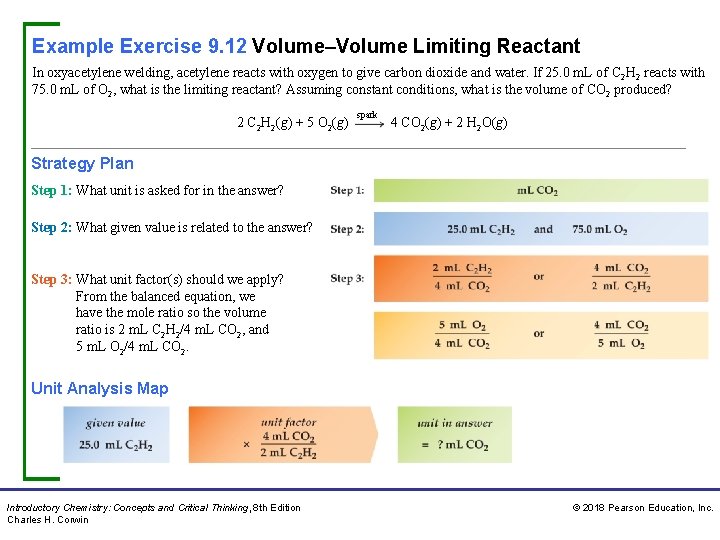
Example Exercise 9. 12 Volume–Volume Limiting Reactant In oxyacetylene welding, acetylene reacts with oxygen to give carbon dioxide and water. If 25. 0 m. L of C 2 H 2 reacts with 75. 0 m. L of O 2, what is the limiting reactant? Assuming constant conditions, what is the volume of CO 2 produced? 2 C 2 H 2(g) + 5 O 2(g) spark 4 CO 2(g) + 2 H 2 O(g) Strategy Plan Step 1: What unit is asked for in the answer? Step 2: What given value is related to the answer? Step 3: What unit factor(s) should we apply? From the balanced equation, we have the mole ratio so the volume ratio is 2 m. L C 2 H 2/4 m. L CO 2, and 5 m. L O 2/4 m. L CO 2. Unit Analysis Map Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 12 Volume–Volume Limiting Reactant Continued Solution First, we find the amount of product that can be produced from the first reactant (m. L C 2 H 2). Thus, Second, we find the amount of product that can be produced from the second reactant (m. L O 2): Third, we compare the amount of product from each reactant and see that C 2 H 2 produces 50. 0 m. L CO 2, whereas O 2 produces 60. 0 m. L CO 2. Thus, C 2 H 2 is the limiting reactant and the amount of product is limited to 50. 0 m. L CO 2. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 12 Volume–Volume Limiting Reactant Continued Practice Exercise Ethane undergoes combustion to give carbon dioxide and water. If 10. 0 L of C 2 H 6 reacts with 25. 0 L of O 2, what is the limiting reactant? Assuming constant conditions, what is the volume of CO 2 produced? The equation is 2 C 2 H 6(g) + 7 O 2(g) spark 4 CO 2(g) + 6 H 2 O(g) Answers: The limiting reactant is O 2, which gives 14. 3 L of CO 2. Concept Exercise Indicate the limiting reactant shown in the following illustration: Zn(s) + S(s) ∆ Zn. S(s) Answer: See Appendix G, 9. 12. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 13 Percent Yield A student dissolves 1. 50 g of copper(II) nitrate in water. After adding aqueous sodium carbonate solution, the student obtains 0. 875 g of Cu. CO 3 precipitate. If theoretical yield is 0. 988 g, what is the percent yield? Cu(NO 3) 2(aq) + Na 2 CO 3(aq) Cu. CO 3(s) + 2 Na. NO 3(aq) Solution The percent yield is the ratio of the actual yield compared to theoretical yield. In this experiment, the actual yield is 0. 875 g and theoretical yield is 0. 988 g. The percent yield, therefore, is The percent yield obtained by the student is 88. 6%. Practice Exercise Ammonium nitrate is used in explosives and is produced by the reaction of ammonia, NH 3, and nitric acid. The equation for the reaction is NH 3(g) + HNO 3(aq) NH 4 NO 3(s) If 15. 0 kg of ammonia gives an actual yield of 65. 3 kg of ammonium nitrate, what is the percent yield? The calculated yield of ammonium nitrate for the experiment is 70. 5 kg. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.

Example Exercise 9. 13 Percent Yield Continued Answer: 92. 6% Concept Exercise Is it possible to have a percent yield greater than 100%? Answer: See Appendix G, 9. 13. Introductory Chemistry: Concepts and Critical Thinking, 8 th Edition Charles H. Corwin © 2018 Pearson Education, Inc.