Evolving Patterns Of Use Of Aldosterone Inhibition In

Evolving Patterns Of Use Of Aldosterone Inhibition In Chronic Heart Failure; A Report From Get With The Guidelines HF Nancy M. Albert, Clyde W. Yancy, Li Liang, Adrian Hernandez, Gregg C. Fonarow, and the Get with the Guidelines Steering Committee and Hospitals

Presenter Disclosure Information AHA Scientific Sessions Evolving Patterns Of Use Of Aldosterone Inhibition In Chronic Heart Failure; A Report From Get With The Guidelines HF n n I will not discuss off label or investigational use of drugs or devices in my presentation. I have financial relationships to disclose: n Consultant and Speakers Bureau: Glaxo. Smith. Kline n Consultant: Medtronic GWTG-HF was sponsored in part by funding from Glaxo. Smith. Kline to the American Heart Association

Background: Level B Evidence n Aldosterone inhibition recommendations: n Moderately severe-severe HF symptoms (i. e. hospitalized for HF) and n Reduced LVEF n Careful monitoring to preserve renal function and normal K+ n n Serum creatinine n ≤ 2. 5 mg/d. L- men n ≤ 2. 0 mg/d. L – women Serum potassium < 5. 0 m. Eq/L Hunt SA, et al. ACC/AHA 2005 Practice Guidelines. Available at http: //www. acc. org.
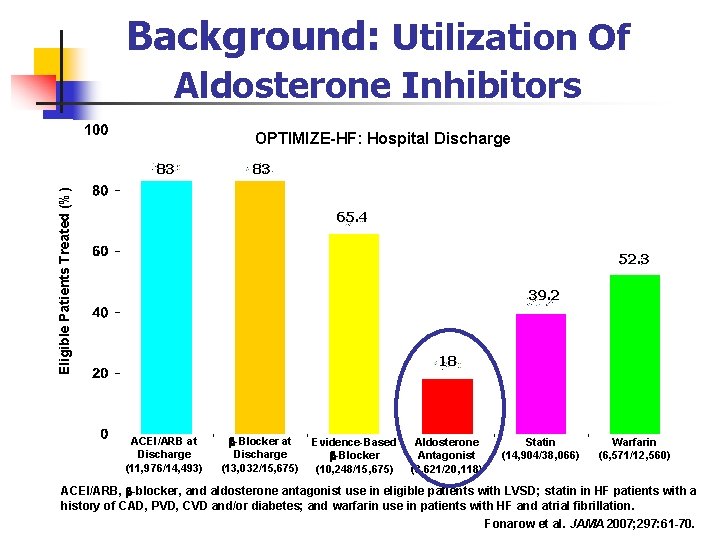
Background: Utilization Of Aldosterone Inhibitors OPTIMIZE-HF: Hospital Discharge 83 Eligible Patients Treated (%) 83 65. 4 52. 3 39. 2 18 ACEI/ARB at Discharge (11, 976/14, 493) -Blocker at Discharge (13, 032/15, 675) Evidence-Based -Blocker (10, 248/15, 675) Aldosterone Antagonist (3, 621/20, 118) Statin (14, 904/38, 066) Warfarin (6, 571/12, 560) ACEI/ARB, -blocker, and aldosterone antagonist use in eligible patients with LVSD; statin in HF patients with a history of CAD, PVD, CVD and/or diabetes; and warfarin use in patients with HF and atrial fibrillation. Fonarow et al. JAMA 2007; 297: 61 -70.
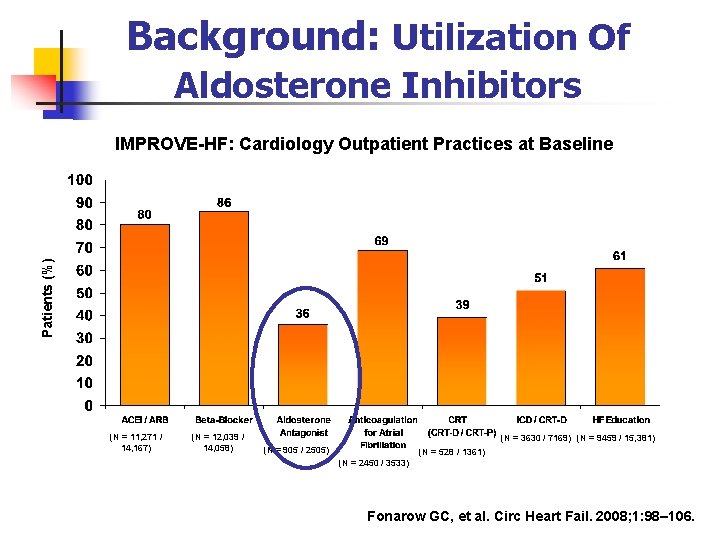
Background: Utilization Of Aldosterone Inhibitors Patients (%) IMPROVE-HF: Cardiology Outpatient Practices at Baseline (N = 11, 271 / 14, 167) (N = 12, 039 / 14, 058) (N = 3630 / 7169) (N = 9459 / 15, 381) (N = 905 / 2505) (N = 528 / 1361) (N = 2450 / 3533) Fonarow GC, et al. Circ Heart Fail. 2008; 1: 98– 106.

Background: Potential for Harm n n n Usage since RALES 1 1 n > 7 fold : 3% to 21. 3% n > 4 fold : 34/1000 pts. (94) to 149/1000 pts (01) 30. 9% did not meet enrollment criteria 1 n 22. 8%, hyperkalemia; 14. 1% Sr. Creatinine ≥ 2. 5 mg/d. L; 17. 3%, e. GFR < 30 ml/min Discontinued in 7. 2% (67/926 cases)3 n 49%, hyperkalemia; 51%, renal failure Hospitalization for hyperkalemia 2 n 2. 4/1000 (1994) to 11. 0/1000 (2001) Mortality for hyperkalemia 2 n 0. 3/1000 (1994) to 2. 0/1000 (2001) FA, et al. Circulation 2005; 112: 39 -47. 2 Juurlink DN, et al. NEJM 2004; 351: 543 -551. 3 Tamirisa KP et al. Am Heart J 2004; 148: 971 -978. 1 Masoudi

PURPOSE n n Problem: n It is unknown if HF patients in a quality of care hospital program receive aldosterone inhibitors more often and receive this therapy per recommendations Research Question: n Has the appropriateness of aldosterone inhibitor usage among patients hospitalized for heart failure improved since 2005?

METHODS: Sample n n Get With the Guidelines-Heart Failure (GWTG-HF) n National initiative of the AHA to improve guidelines adherence in patients hospitalized with HF Study Cohort n 242 participating hospitals n 45, 322 patients hospitalized for HF n n n Discharged home Without contraindications to aldosterone inh. January 1, 2005 – December 26, 2007

Patient Management Tool n Data was recorded using the Patient Management Tool™ (Outcome, Cambridge, MA), a Web-based interactive assessment and reporting system that tracks treatment and facilitates evidence- based medicine

METHODS: Definitions & Analysis n n n n Definitions LV systolic dysfunction: EF ≤ 35% Normal K+ level: ≤ 5. 5 mmol/L Normal serum creatinine: < 2. 5 mg/d. L Opt Medical Tx: BB, ACEi/ARB or diuretic if indicated Analysis Cochran-Mantel Haenzel general association statistics: Aldosterone in patient groups Cochran-Mantel Haenzel- Row Mean scores: Aldosterone and time n Within hospital clustering was considered Multivariable logistic regression analysis using Generalized Estimating Equations to account for pt & hosp characteristics and clustering within hospitals
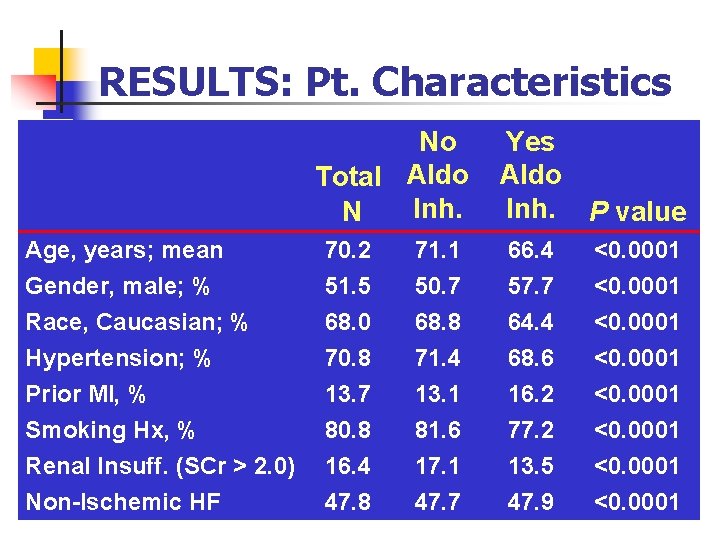
RESULTS: Pt. Characteristics No Total Aldo Inh. N Yes Aldo Inh. P value Age, years; mean Gender, male; % Race, Caucasian; % Hypertension; % Prior MI, % 70. 2 51. 5 68. 0 70. 8 13. 7 71. 1 50. 7 68. 8 71. 4 13. 1 66. 4 57. 7 64. 4 68. 6 16. 2 <0. 0001 Smoking Hx, % Renal Insuff. (SCr > 2. 0) Non-Ischemic HF 80. 8 16. 4 47. 8 81. 6 17. 1 47. 7 77. 2 13. 5 47. 9 <0. 0001
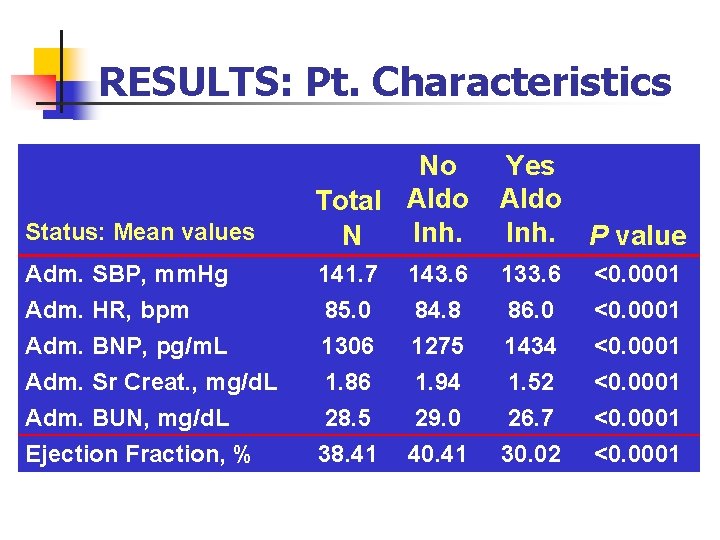
RESULTS: Pt. Characteristics Status: Mean values No Total Aldo Inh. N Yes Aldo Inh. P value Adm. SBP, mm. Hg Adm. HR, bpm Adm. BNP, pg/m. L Adm. Sr Creat. , mg/d. L Adm. BUN, mg/d. L 141. 7 85. 0 1306 1. 86 28. 5 143. 6 84. 8 1275 1. 94 29. 0 133. 6 86. 0 1434 1. 52 26. 7 <0. 0001 Ejection Fraction, % 38. 41 40. 41 30. 02 <0. 0001
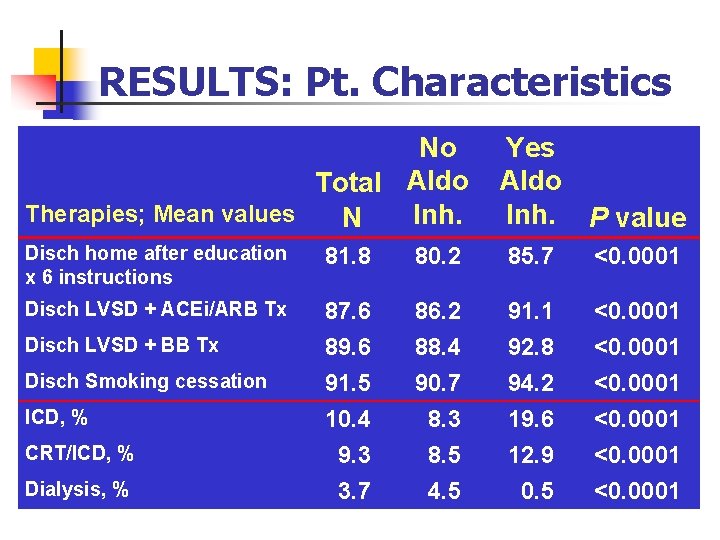
RESULTS: Pt. Characteristics No Total Aldo Therapies; Mean values Inh. N Yes Aldo Inh. P value Disch home after education x 6 instructions 81. 8 80. 2 85. 7 <0. 0001 Disch LVSD + ACEi/ARB Tx 87. 6 89. 6 91. 5 10. 4 9. 3 3. 7 86. 2 88. 4 90. 7 8. 3 8. 5 4. 5 91. 1 92. 8 94. 2 19. 6 12. 9 0. 5 <0. 0001 Disch LVSD + BB Tx Disch Smoking cessation ICD, % CRT/ICD, % Dialysis, %

RESULTS: Aldosterone Inh. Use of Aldosterone Inhibitor n. Overall n. EF ≤ 35% n. LVSD + Sr Creat < 2. 5 mg/d. L n. LVSD + Cr Clearance > 30 ml min n. LVSD + Sr K+ ≤ 5. 5 mmol/L n. LVSD + optimal medical Tx when n not contraindicated n. ACC/AHA guidelines (EF ≤ 35%) % 18. 9 28. 9 31. 8 32. 6 30. 5 29. 4 33. 8
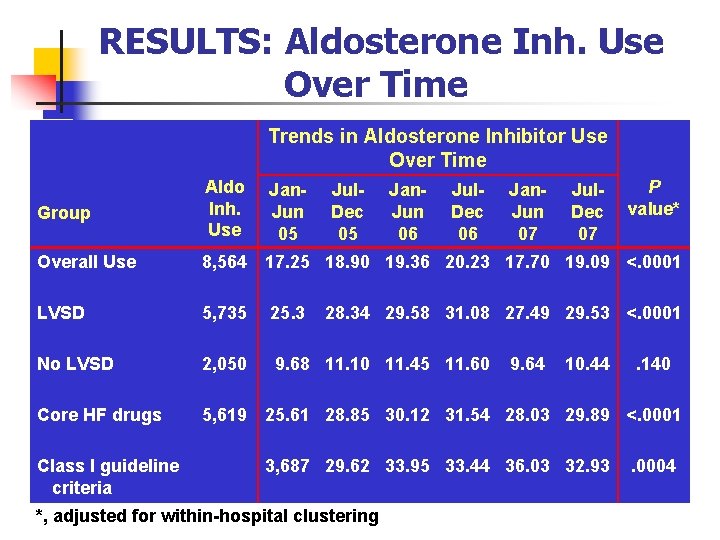
RESULTS: Aldosterone Inh. Use Over Time Trends in Aldosterone Inhibitor Use Over Time Group Aldo Inh. Use Overall Use 8, 564 17. 25 18. 90 19. 36 20. 23 17. 70 19. 09 <. 0001 LVSD 5, 735 25. 3 No LVSD 2, 050 9. 68 11. 10 11. 45 11. 60 Core HF drugs 5, 619 25. 61 28. 85 30. 12 31. 54 28. 03 29. 89 <. 0001 Class I guideline criteria Jan. Jun 05 Jul. Dec 05 Jan. Jun 06 Jul. Dec 06 Jan. Jun 07 Jul. Dec 07 P value* 28. 34 29. 58 31. 08 27. 49 29. 53 <. 0001 9. 64 10. 44 3, 687 29. 62 33. 95 33. 44 36. 03 32. 93 *, adjusted for within-hospital clustering . 140 . 0004
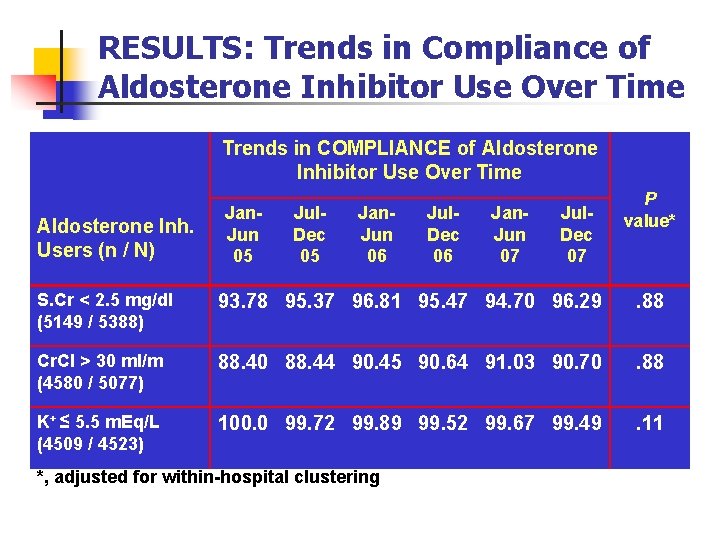
RESULTS: Trends in Compliance of Aldosterone Inhibitor Use Over Time Trends in COMPLIANCE of Aldosterone Inhibitor Use Over Time Aldosterone Inh. Users (n / N) Jan. Jun 05 Jul. Dec 05 Jan. Jun 06 Jul. Dec 06 Jan. Jun 07 Jul. Dec 07 P value* S. Cr < 2. 5 mg/dl (5149 / 5388) 93. 78 95. 37 96. 81 95. 47 94. 70 96. 29 . 88 Cr. Cl > 30 ml/m (4580 / 5077) 88. 40 88. 44 90. 45 90. 64 91. 03 90. 70 . 88 K+ ≤ 5. 5 m. Eq/L (4509 / 4523) 100. 0 99. 72 99. 89 99. 52 99. 67 99. 49 . 11 *, adjusted for within-hospital clustering
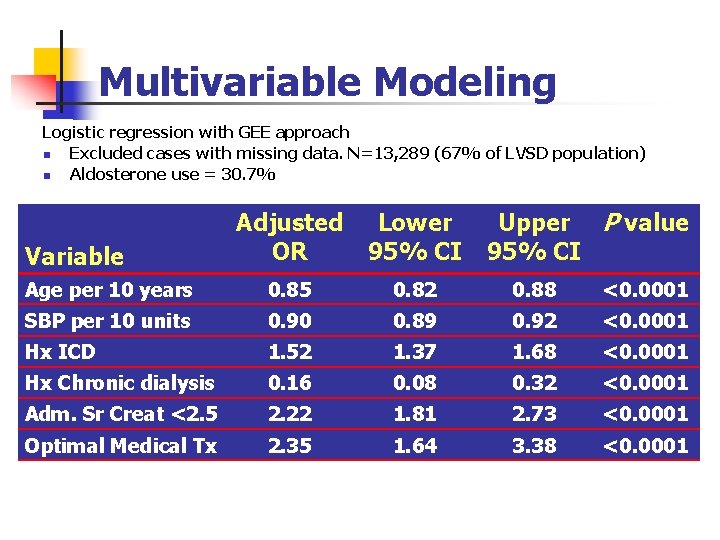
Multivariable Modeling Logistic regression with GEE approach n Excluded cases with missing data. N=13, 289 (67% of LVSD population) n Aldosterone use = 30. 7% Upper P value 95% CI Adjusted OR Lower 95% CI Age per 10 years 0. 85 0. 82 0. 88 <0. 0001 SBP per 10 units 0. 90 0. 89 0. 92 <0. 0001 Hx ICD 1. 52 1. 37 1. 68 <0. 0001 Hx Chronic dialysis 0. 16 0. 08 0. 32 <0. 0001 Adm. Sr Creat <2. 5 2. 22 1. 81 2. 73 <0. 0001 Optimal Medical Tx 2. 35 1. 64 3. 38 <0. 0001 Variable

LIMITATIONS n n Data presented are dependent upon the accuracy and completeness of data abstraction from medical chart review GWTG-HF hospitals are self selected Rationale for decisions regarding therapy utilization may not be captured These findings may not apply to practices that differ in patient characteristics or care patterns from GWTG-HF hospitals

CONCLUSIONS n n n These data are among the first to assess aldosterone inhibitor use in hospitalized patients and appropriateness since ~ 2005. Within pts enrolled in GWTG HF, they demonstrate: n Appropriate use of aldosterone inhibitors increased modestly from 2005 -2007 n Non-indicated use was low n Overall use of aldosterone inhibitors remains lower than expected n Users are more likely to have higher compliance on other performance and quality measures Additional research is required to identify ongoing impediments to aldosterone inhibitors use.
- Slides: 19