Evolving Importance of Post Approval Studies Danica MarinacDabic

Evolving Importance of Post. Approval Studies Danica Marinac-Dabic, MD, Ph. D Director, Division of Epidemiology Food and Drug Administrati

Summary ü Public health context/need ü Recent CDRH Initiatives ü Focus on innovative methods ü Focus on national/international infrastructure ü Focus on collaboration/partnership ü Vision for the future Division of Epidemiology Food and Drug Administrati

TPLC Context § TPLC begins with “on market” knowledge ü Utilization/diffusion into clinical practice ü Patterns of use in different populations ü Long-term safety and effectiveness ü Benefit/risk profile in a “real world setting” üLearning curve issues üHeterogeneity of treatment effects üComparative safety and effectiveness ü Public health impact/burden Division of Epidemiology Food and Drug Administrati
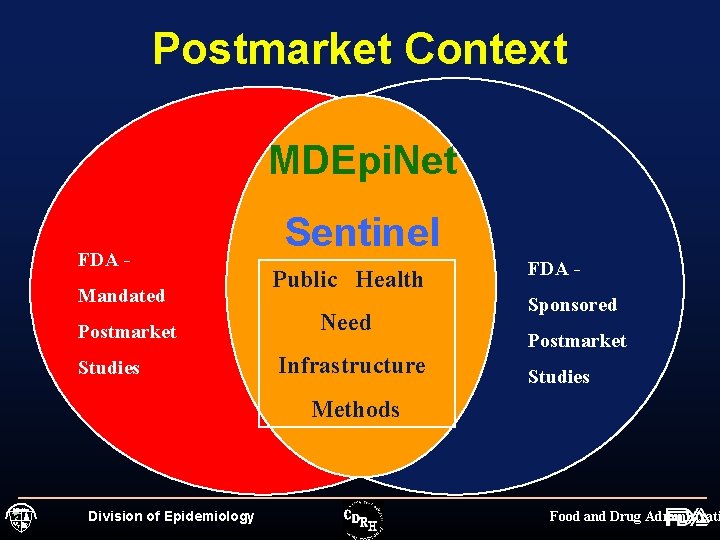
Postmarket Context MDEpi. Net FDA - Sentinel Mandated Public Health P Postmarket Need Studies Infrastructure FDA Sponsored Postmarket Studies Methods Division of Epidemiology Food and Drug Administrati

Focus on Infrastructure: Registries Initiative ICOR Initiative Division of Epidemiology Food and Drug Administrati

Ongoing Registry Efforts § Use existing registries for PAS studies and surveillance ü INTERMACS (NIH, CMS, FDA) ü Total Joint Replacement Registry (Kaiser) ü Australian National Joint Replacement Registry § Facilitate new registry development ü Transcatheter Aortic Valve Replacement Registry (ACC/STS) ü Fibrillation Registry (ACC, HRS, STS) ü American Joint Replacement Registry (AAOS) ü Diagnostic and Therapeutic Bronhoscopy Registry (ACCP) ü Uro-Gynecological Mesh Registry ü IMPACT Registry (ACC) Division of Epidemiology Food and Drug Administrati

Ongoing Registry Efforts (cont) § Use existing registries for discretionary studies ü ICD Registry (ACC-NCDR) ü Adult Cardio-Thoracic Database (STS) ü Total Joint Replacement Registry (Kaiser) ü OUS Orthopedic Registries (Australia, Denmark) § Explore registry capabilities ü Active surveillance: short-term and longitudinal (DELTA) ü Linkages studies with Medicare claims data § Advocate for registries (AHRQ’s Guide on Registries) § Build methodological infrastructure for registries (ICOR) Division of Epidemiology Food and Drug Administrati

Focus on Methods: Evidence Synthesis Initiative Division of Epidemiology Food and Drug Administrati

Innovative Methods Make better use of existing pre & post approval data Integrate/combine when appropriate! Methods of Data Integration Use Simultaneous: ü Meta-analysis ü Network meta-analysis ü Cross-design synthesis Bayes Division factors of Epidemiology Direct measure of evidence Food and Drug Administrati

Focus on Strategic Partnerships: MDEpi. Net Initiative Division of Epidemiology Food and Drug Administrati

MDEpi. Net Initiative MISSION üTo develop infrastructure and innovative methodological approaches for conducting robust studies to improve medical device safety and effectiveness understanding throughout the device life cycle through Public Private Partnership with academia and other stakeholders. Division of Epidemiology Food and Drug Administrati

MDEpi. Net Academic Institutions § Harvard University (Methodology Center) § Cornell University (Science and Infrastructure Center) § § § § University of North Carolina Duke University of Alabama University of Washington Vanderbilt University of Southern California University of Oxford Division of Epidemiology Food and Drug Administrati

Impact - Vision for the Future § Improved knowledge of extent of observational clinical data available for regulatory decision making ü inventory and evaluate data sources, to leverage existing clinical data for premarket submissions ü use data sources and infrastructure to evaluate signals that are raised, thus decreasing the need for additional de novo studies in the postmarket setting and providing an evidentiary basis for next generation device development ü identify additional needs for infrastructure and methodologies for device performance evaluation Division of Epidemiology Food and Drug Administrati

Impact – Vision for the Future (cont) § Reduced burden on industry ü reduce burden on each sponsor to develop own infrastructure for premarket clinical studies and mandated postmarket studies and surveillance ü eliminate the need for multiple costly studies being independently developed by several sponsors for similar devices ü with knowledge of all worldwide data/infrastructure, all manufacturers will benefit because evidence can be used to inform next generation of medical devices § Pan –stakeholder input through Public Private Partnership ü all medical device stakeholders represented for decision-making process Division of Epidemiology Food and Drug Administrati
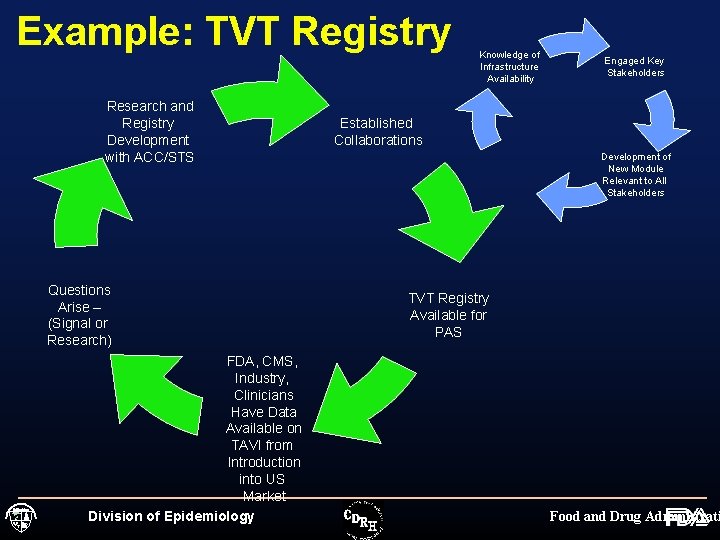
Example: TVT Registry Research and Registry Development with ACC/STS Questions Arise – (Signal or Research) FDA, CMS, Industry, Clinicians Have Data Available on TAVI from Introduction into US Market Division of Epidemiology Knowledge of Infrastructure Availability Engaged Key Stakeholders Established Collaborations Development of New Module Relevant to All Stakeholders TVT Registry Available for PAS Food and Drug Administrati

Thank you! danica. marinac-dabic@fda. hhs. gov (301) 796 -6689 Division of Epidemiology Food and Drug Administrati
- Slides: 16