EvidenceBased Medicine Use of current best evidence in



Evidence-Based Medicine ※ Use of current best evidence in making decisions about the care of individual patients. ※ EBM is the integration of best research evidence with clinical expertise and patient values. 以流行病學和統計學的方法,從龐大的 醫學資料庫中嚴格評讀、綜合分析並找 出值得信賴的部分,並將所能獲得的最 佳文獻證據,應用於臨床 作中,使病 人得到最佳的照顧。

Decision Making in Health Care • Searching bibliographic databases • Browse journals • Ask colleagues • Textbooks • Remember what you learned during your professional training • “Do no harm” EBM: Not only a skill but also an attitude change!


進階學習 目前國外推動實證醫學著名的單位 • 加拿大Mc. Master University的HIRU(Health Information Research Unit)是Cochrane Collaboration的重鎮 –http: //hiru. mcmaster. ca/ • 英國Oxford University的 Centre for Evidence-Based Medicine –http: //cemb. jr 2. ox. ac. uk • 美國American College of Physician(ACP), 在全球資訊網出版 ACP Journal Club Online –http: //www. acpjc. org

What Does EBM Achieve • Refinement and reduction – 化繁為簡 • Efficiency – 效率 • Generalizability and consistency – 可類推性及一致性 • Reliability and precision – 可信賴度 –提供較準確的預估 Systemic review meta-analysis


進行實證醫學的五個進行步驟 1. Formulate an answerable question. 由個案的臨床資料形成可回答的臨床問題 2. Track down the best evidence. 尋找最佳的實證� 各種文獻及資料庫,包括發表及未發表的資料 � 3. Critically appraise the evidence for validity, impact, and applicability. 評估各種醫學報告的可信度、臨床重要性,以及可應用性 4. Integrate with our clinical expertise and patient values. 整合並應用於實際患者的治療決策﹝臨床應用﹞ 5. Evaluate our effectiveness and efficacy. 效果評估

Asking Answerable Clinical Questions Well-built Clinical Question • “Background” question有關疾病一般知識性問題 – Ask general knowledge about a disorder – Have two essential components: • A question root (who, what, why, when…) with a verb • A disorder, or an aspect of a disorder • “Foreground” question處理病人所需特別知識的問題 – Ask for specific knowledge about managing patients with a disorder – Have four (or three) essential components (PICO): • • 1. Patient and/or problem 2. Intervention (treatment) 3. Comparison intervention 4. Outcomes

臨床問題類型: • 危害或致病因子探討(Risk factor) – Cohort study – Case-control study • 診斷 (Diagnosis) – Sensitivity, specificity – Predictive value (PPV, NPV) • 治療 (Therapy) – Clinical trial, field trial • 預後 (Prognosis) – Prediction model, simulation model


There are four elements of a wellformulated question (PICO ) ( Patient ~ Who is the patient or what is the problem being addressed? Intervention ~ What is the intervention? intervention Comparison ~ What are the alternatives? alternatives Outcome ~ What are the outcomes? outcomes

Searching The Best Evidence 尋找最佳實證資料 • 一是研究論文資料庫(primary journals or databases) ~ 如 Medline, NEJM, Lancet… • 或是直接使用實證醫學資料庫 (secondary journals or databases) ~ ACP journal club, Cochrane. • 盡量搜尋與病人問題相同且證據等級(level of evidence) 較高之文獻,再謹慎的評讀與評估其在此 問題的適用性。

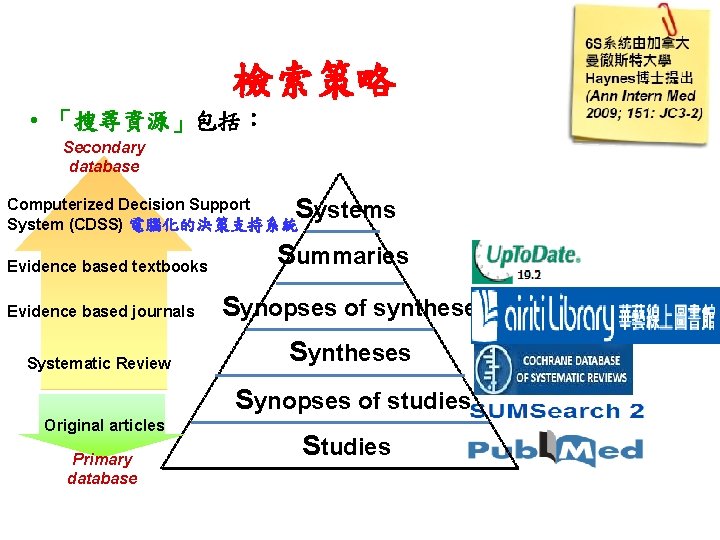
檢索策略 • 「搜尋資源」包括: Secondary database Systems Computerized Decision Support System (CDSS) 電腦化的決策支持系統 Evidence based textbooks Evidence based journals Systematic Review Summaries Synopses of syntheses Synopses of studies Original articles Primary database Studies


統計數字會說話 評估時以具體的數字呈現結果 • 敏感度(sensitivity)、特異度(specificity)、概似比(likelihood ratio)、檢測前機率(pre-test probability)、檢測後機率(post-test probability) • ARR (Absolute risk reduction) = EER (Experimental Event Rate) - CER (Control Event Rate)、 Number needed to treat, NNT=1/ARR (要比對照組 多一位存活或少個併發症之實驗組樣本數目, NNT越小 治療效果越好或併發症 越少)、相對危險度減少百分比(relative risk reduction,RRR) • 絕對危險度增加百分比(absolute risk increase,ARI)= EER (Experimental Event Rate) - CER (Control Event Rate)、 Number needed to harm, NNH=1/ARI(要比對照組多一位死亡或多個併發症之 實驗組樣本數目, NNH越小, 治療效果越差或併發症越多) • 相對危險 (Relative risk)、勝算 (Odds)、勝算比 (Odds ratio) 、信賴區間 (confidence interval)

• Evidence is never enough ! • Evidences are inconsistent!

Interpretation of evidence is subjective: it dependents on how you view it.

Asking Answerable Clinical Question Patient/Problem Insulin-dependent diabetics Intervention Intensive insulin regimen Comparison Regular insulin regimen Outcomes Retinopathy Symptomatic hypoglycemia

Treatment Effects • Occurrence of diabetic retinopathy at 5 years among insulin-dependent diabetic in the DCCT trial • Usual insulin regimen (CER: control event rate): 38% • Intensive insulin regimen (EER: experimental event rate): 13% Risk Reduction (calculation): NNT • Absolute risk reduction (ARR) = │CER-EER│ = 38%-13% = 25% • Relative risk reduction (RRR) = │CER-EER│/CER = 25%/38% =66% • Number needed to treat (NNT) = 1/ARR = 1/25% = 4 patients – NNT: The number of patients that need to be treated to prevent one bad outcome or get one good outcome.

Harm • The proportion of patients with at least one episode of symptomatic hypoglycemia • Usual insulin regimen (CER: control event rate): 23% • Intensive insulin regimen (EER: experimental event rate): 57% Risk Increase (calculation): NNH • Absolute risk increase (ARI) = EER - CER = 57% - 23% = 34% • Relative risk increase (RRI) = EER-CER/CER = 57% - 23%/23% = 148% • Number needed to harm (NNH) = 1/ARI = 1/0. 34 = 3 patients (取整數) – NNH: The number of patients that need to be treated to cause one bad outcome (being harmed).
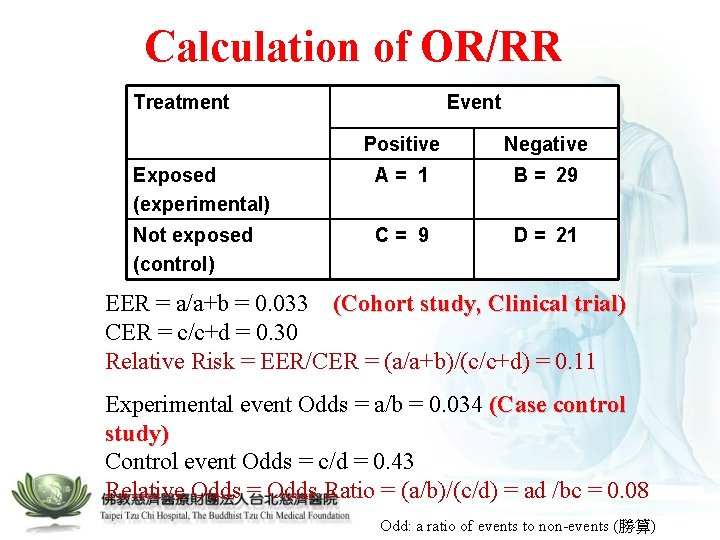
Calculation of OR/RR Treatment Event Positive Negative Exposed (experimental) A= 1 B = 29 Not exposed (control) C= 9 D = 21 EER = a/a+b = 0. 033 (Cohort study, Clinical trial) CER = c/c+d = 0. 30 Relative Risk = EER/CER = (a/a+b)/(c/c+d) = 0. 11 Experimental event Odds = a/b = 0. 034 (Case control study) Control event Odds = c/d = 0. 43 Relative Odds = Odds Ratio = (a/b)/(c/d) = ad /bc = 0. 08 Odd: a ratio of events to non-events (勝算)


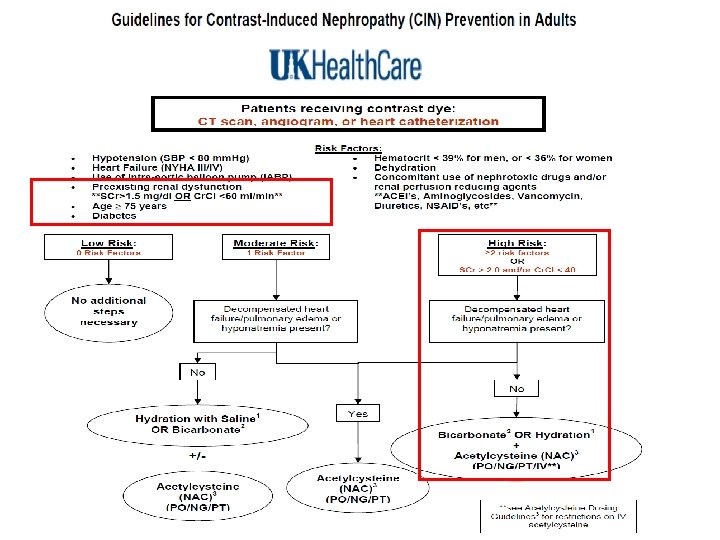

Prevention of contrast-induced nephropathy • We recommend the following preventive measures for patients at increased risk of contrast nephropathy, which is defined as a serum creatinine ≥ 1. 5 mg/d. L (132 micromol/L) or an estimated glomerular filtration rate <60 ml/1. 73 m 2, particularly in those with diabetes.

Renal replacement therapy • Among patients with stage 3 and 4 CKD, we recommend NOT performing prophylactic hemofiltration or hemodialysis after contrast exposure (Grade 1 B). • Among patients with stage 5 CKD, we suggest prophylactic hemodialysis after contrast exposure if there is already a functioning hemodialysis access (Grade 2 C).


Appraisal and Apply • Valid: systemic review worksheet • Importance: what were the results? • Applicability: population and feasibility Is the SR valid Are the result Important? Can the results help me? 33


What question did the systematic review addressed (PICO)想要回答什麼問題? This paper: Yes ■ No � Unclear �

Is it unlikely that important, relevant studies were missed 有沒有遺漏重要的文獻? This paper: Yes� No ■ Unclear �

Were the included studies sufficiently valid for the type of question asked 選擇的文獻有效回答提出的問題? • This paper: Yes ■ No � Unclear �

Were the criteria used to select articles for inclusion appropriate 選擇文獻的準則適當? • This paper: Yes ■ No � Unclear �

Were the criteria used to select articles for inclusion appropriate 選擇文獻的準則適當? This paper: Yes ■ No � Unclear �

Were the results similar from study to study 各研究的 結果相似? This paper: Yes ■ No � Unclear �
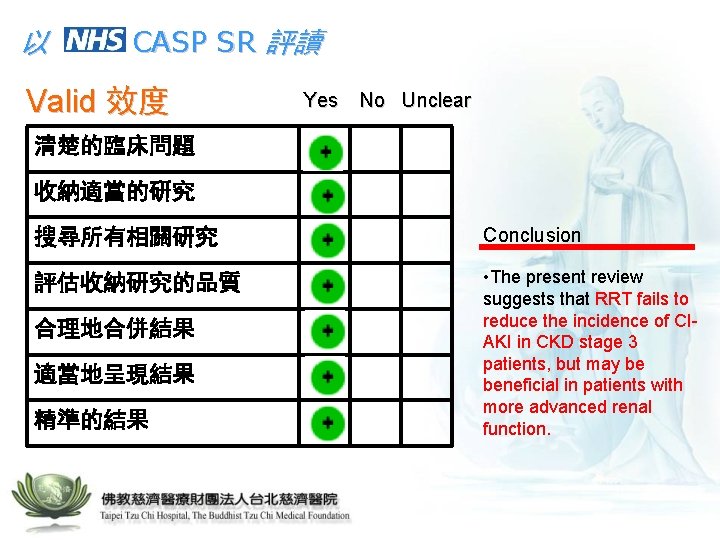
以 CASP SR 評讀 Valid 效度 Yes No Unclear 清楚的臨床問題 收納適當的研究 搜尋所有相關研究 Conclusion 評估收納研究的品質 • The present review suggests that RRT fails to reduce the incidence of CIAKI in CKD stage 3 patients, but may be beneficial in patients with more advanced renal function. 合理地合併結果 適當地呈現結果 精準的結果
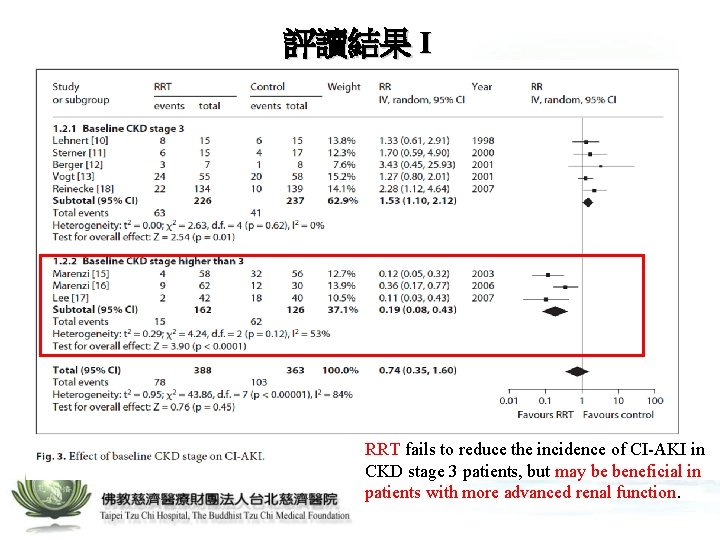
評讀結果 I RRT fails to reduce the incidence of CI-AKI in CKD stage 3 patients, but may be beneficial in patients with more advanced renal function.
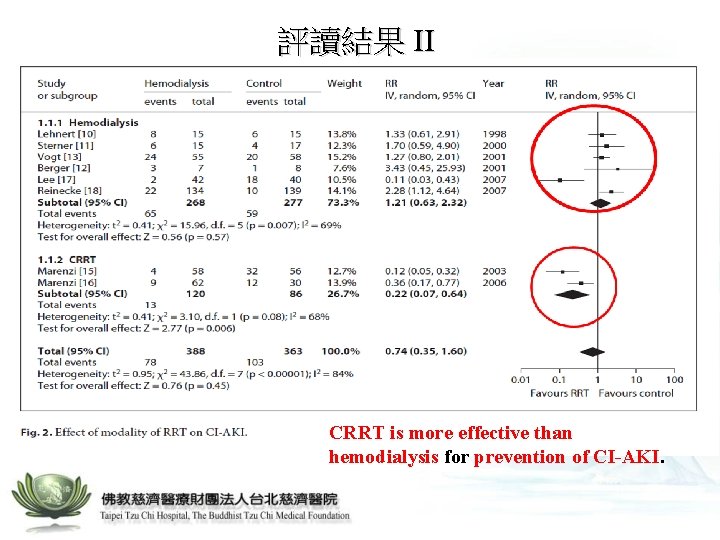
評讀結果 II CRRT is more effective than hemodialysis for prevention of CI-AKI.

Number need to treat I: CRRT II: RRT in >CKD stage 3

Can you apply this valid, important evidence from a systematic review in caring for your patient? 1. Is your patient so different from those in the study that its results cannot apply? 性別 共病 種族 年齡 疾病特徵 2. Treatment feasible in your patient in your setting? Yes 3. What are our patient’ potential benefits and harm from therapy? (NNT, NNH) Yes



What question did the study ask? P Patient/problem I Pts (>65 years) with CKD (>stage 3) receiving CAG, CT, etc. RRT (HD, HF&HDF) Intervention C Comparison O SMT (hydration, hydration+ acetylcysteine) New ESRD, chronic RRT, or Mortality Outcome S Study design 9 randomized controlled trials, and 2 observational studies

Is it unlikely that important, relevant studies were missed? (有無遺漏重要文獻? ) □是 ■否 □不清楚 1. Studies must have contained 10 or more human subjects. 2. We searched the MEDLINE, (Pub. Med interface) and EMBASE up to March 31, 2011, using Boolean search strategies, without language restriction. 3. Two authors (CYG, DNC) independently assessed studies for inclusion and extracted data of interest. 4. 9 randomized controlled trials, and 2 observational studies. 49

Were the criteria used to select articles for inclusion appropriate? (收入條件適當? ) ■是 □否 □不清楚 1. Eleven studies fulfilled the selection criteria and were included in the quantitative analysis. 2. Pts with significant CKD (>stage 3) 3. Receiving diagnostic and therapeutic angiographic studies, computed tomography, or other radiographic investigations. 4. A total of 1010 pts treated in 5 countries; 445 pts in the RRT group and 565 in SMT group. 50

Were the included studies sufficiently valid for the type of question asked? (品質評估? ) ■是 □否 □不清楚 51
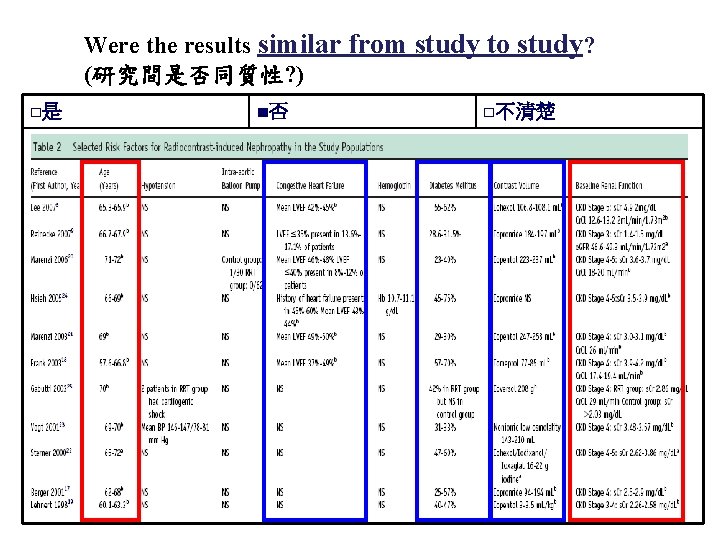
Were the results similar from study to study? (研究間是否同質性? ) □是 ■否 □不清楚 52
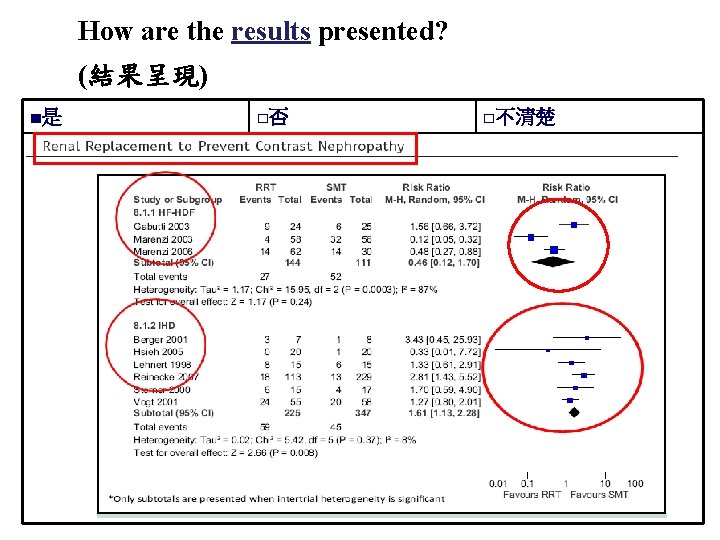
How are the results presented? (結果呈現) ■是 □否 □不清楚 53
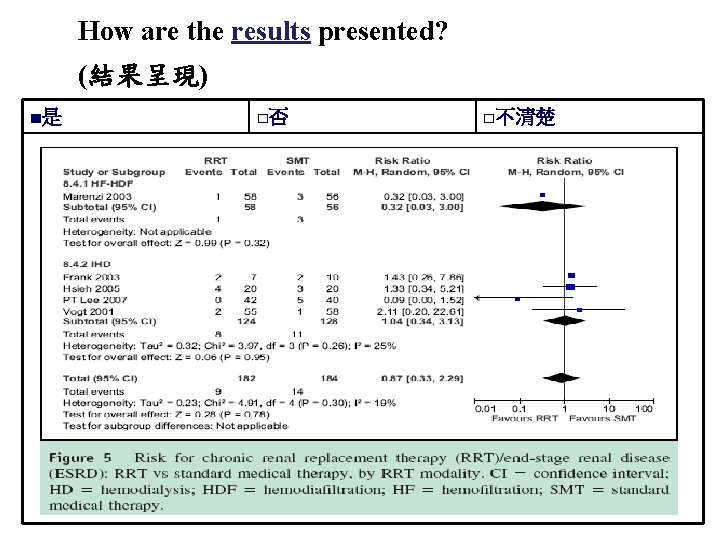
How are the results presented? (結果呈現) ■是 □否 □不清楚 54

結論 • Decrease : incidence of RCIN: No significant difference (RR: 0. 81, 95%CI: 0. 37 -1. 86). • Need for chronic RRT: No significant difference (RRT: SMT=4. 9%: 7. 6%, RR: 0. 87, 95%CI: 0. 33 -2. 29). • Mortality: No significant difference (RRT: SMT=2. 6%: 3. 7%, RR: 0. 65, 95%CI: 0. 17 -2. 49). HD with increased risk of RCIN: RRT-related toxicity(RR: 1. 61) HF with decreased risk of RCIN: unclear(RR: 0. 46, 95%CI: 0. 121. 7)



Interpretation of evidence is subjective: it dependents on how you view it!!

感恩聆聽! 2016 -5 -24 福山植物園, photo by C-H Chen
- Slides: 59