Evidence for Polynuclear Aromatic Hydrocarbon Aggregation in Flames

Evidence for Polynuclear Aromatic Hydrocarbon Aggregation in Flames: Intermolecular Potential Calculations Jennifer D. Herdman and J. Houston Miller Department of Chemistry, The George Washington University Washington, DC 20052 2007 Fall Technical Meeting Eastern States Section of the Combustion Institute University of Virginia October 21 -24, 2007 Support from the National Science Foundation (NSF CTS- 0330230, Drs. Farley Fisher and Linda Blevins).
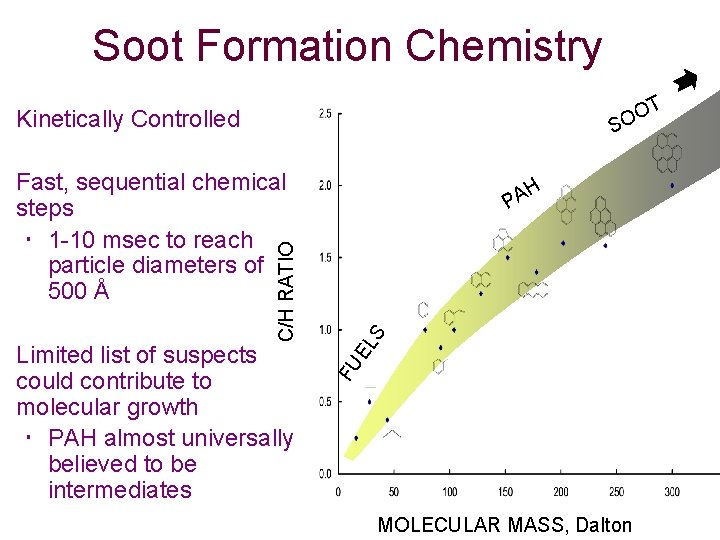
Soot Formation Chemistry OT O S Kinetically Controlled S EL Limited list of suspects could contribute to molecular growth PAH almost universally believed to be intermediates H A P FU C/H RATIO Fast, sequential chemical steps 1 -10 msec to reach particle diameters of 500 Å MOLECULAR MASS, Dalton
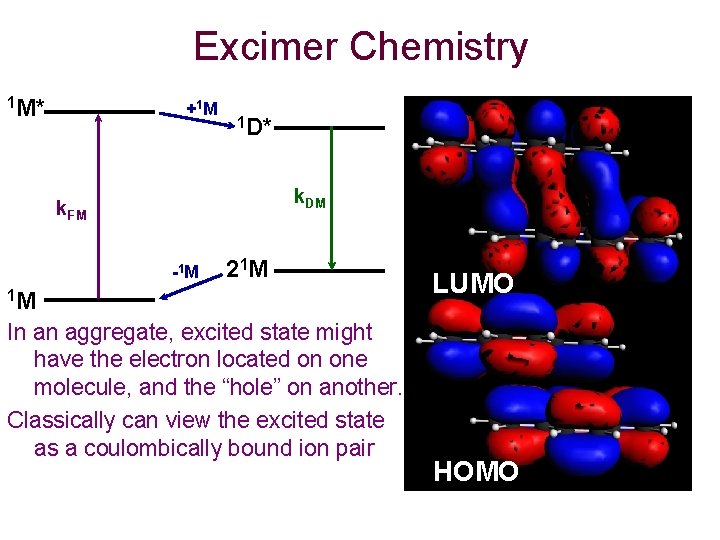
Excimer Chemistry 1 M* +1 M 1 D* k. DM k. FM -1 M 2 1 M 1 M In an aggregate, excited state might have the electron located on one molecule, and the “hole” on another. Classically can view the excited state as a coulombically bound ion pair LUMO HOMO
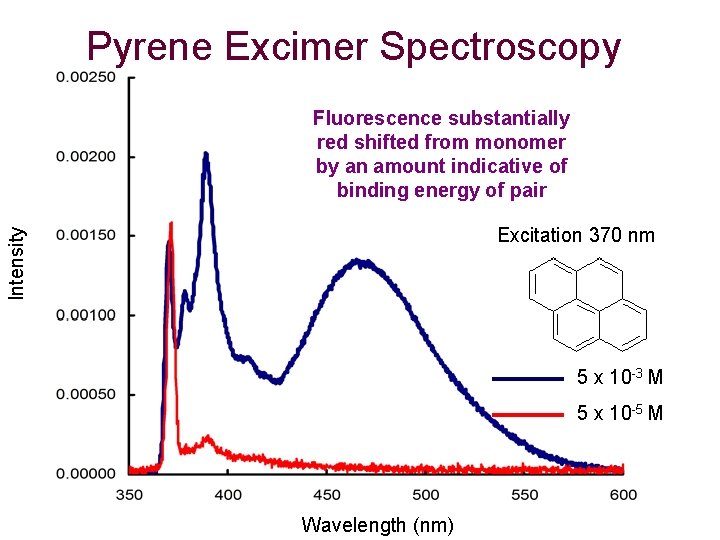
Pyrene Excimer Spectroscopy Fluorescence substantially red shifted from monomer by an amount indicative of binding energy of pair Intensity Excitation 370 nm 5 x 10 -3 M 5 x 10 -5 M Wavelength (nm)

PAH Agglomeration 20 th Symposium: (Miller et al. ) Equilibrium concentrations for PAH dimers in flames were estimated from estimated monomer concentrations Dimer concentrations < number densities of young soot particles 23 rd Symposium: (Miller) Calculated dimer lifetimes assuming collisional stabilization of colliding pair Lifetimes were long enough for large PAH ( 600 Dalton). 29 th Symposium: (Schuetz and Frenklach) Semi-empirical molecular dynamics used to calculate dimer lifetimes for pyrene. Deposition of energy into internal rotations extended lifetimes.

Computations Dispersive: van de Waal 1983 Miller et al. 1984 Electrostatic: van de Waal 1983 Intermolecular Potential = Dispersive + Electrostatic
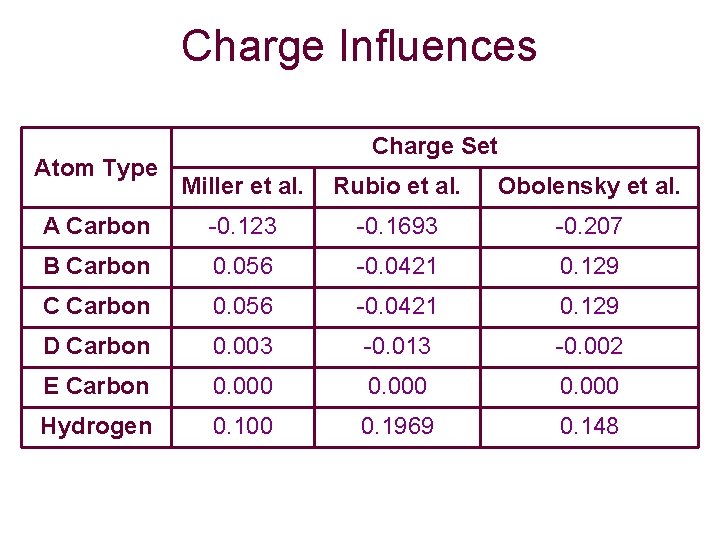
Charge Influences Atom Type Charge Set Miller et al. Rubio et al. Obolensky et al. A Carbon -0. 123 -0. 1693 -0. 207 B Carbon 0. 056 -0. 0421 0. 129 C Carbon 0. 056 -0. 0421 0. 129 D Carbon 0. 003 -0. 013 -0. 002 E Carbon 0. 000 Hydrogen 0. 100 0. 1969 0. 148
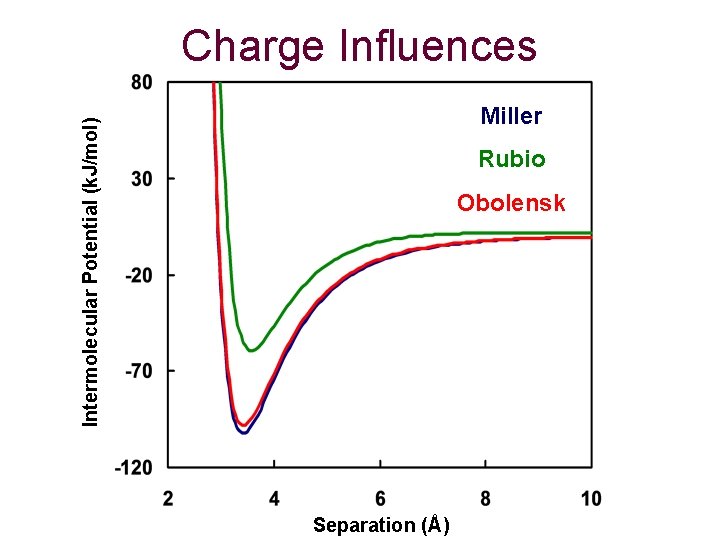
Charge Influences Intermolecular Potential (k. J/mol) Miller Rubio Obolensk Separation (Å)
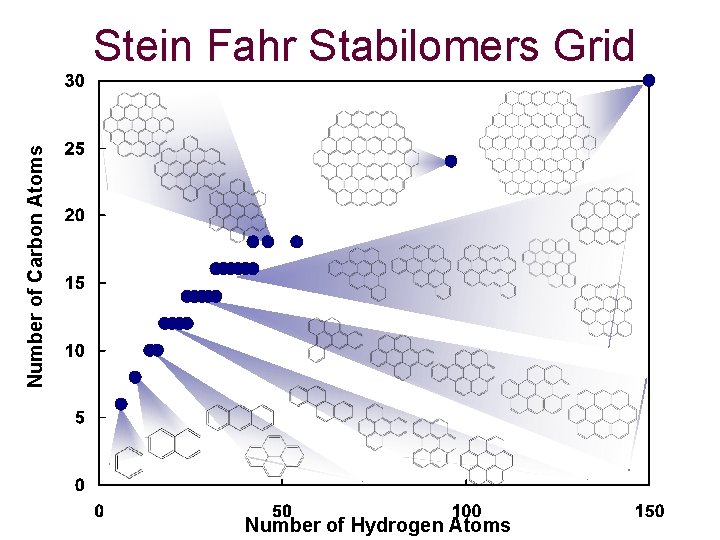
Number of Carbon Atoms Stein Fahr Stabilomers Grid Number of Hydrogen Atoms
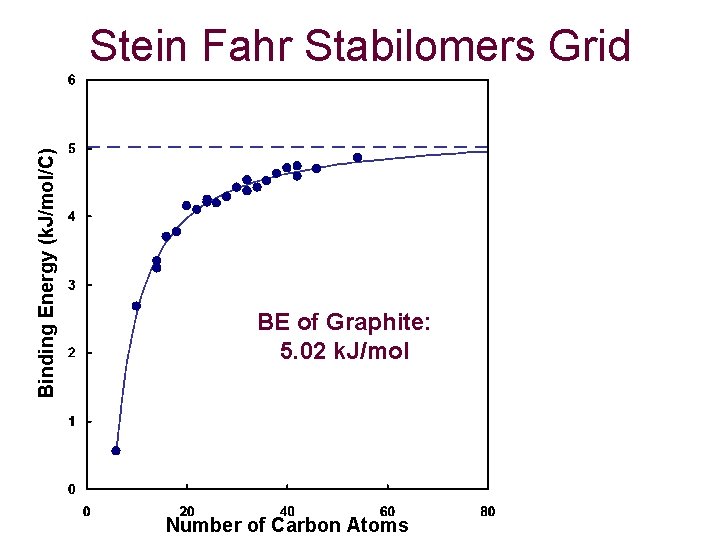
Binding Energy (k. J/mol/C) Stein Fahr Stabilomers Grid BE of Graphite: 5. 02 k. J/mol Number of Carbon Atoms
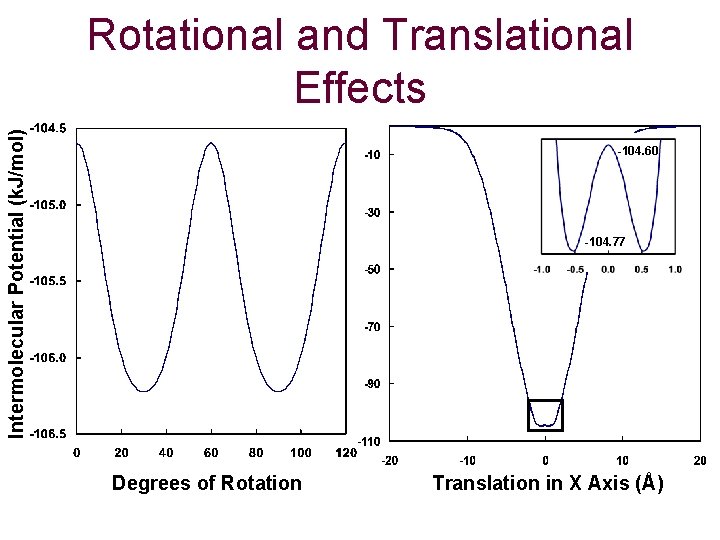
Intermolecular Potential (k. J/mol) Rotational and Translational Effects -104. 60 -104. 77 Degrees of Rotation Translation in X Axis (Å)
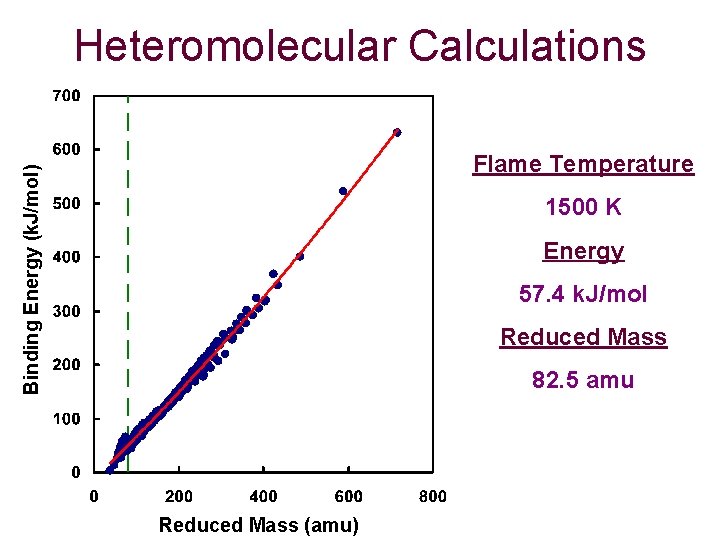
Heteromolecular Calculations Binding Energy (k. J/mol) Flame Temperature 1500 K Energy 57. 4 k. J/mol Reduced Mass 82. 5 amu Reduced Mass (amu)
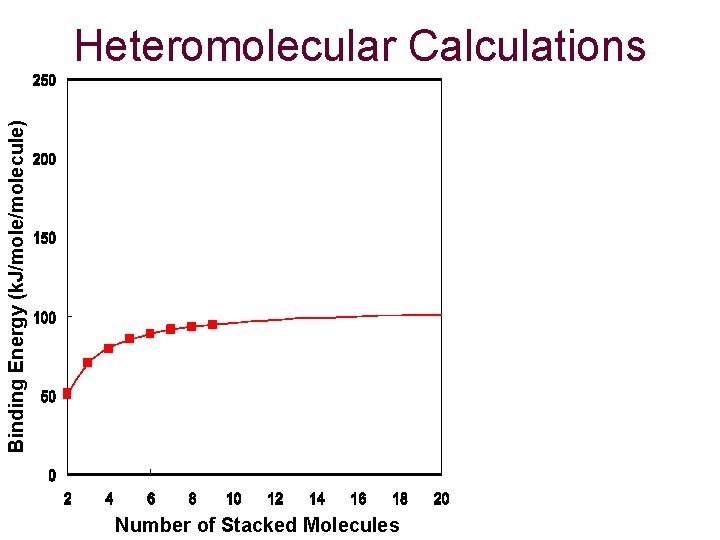
Binding Energy (k. J/molecule) Heteromolecular Calculations Number of Stacked Molecules
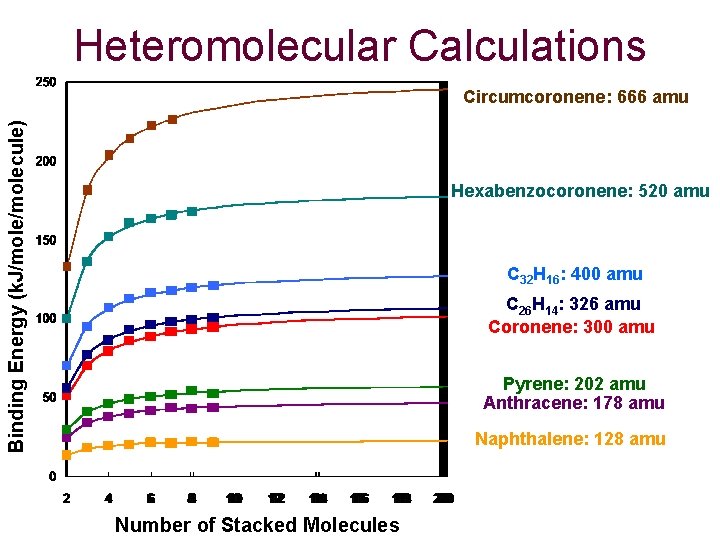
Heteromolecular Calculations Binding Energy (k. J/molecule) Circumcoronene: 666 amu Hexabenzocoronene: 520 amu C 32 H 16: 400 amu C 26 H 14: 326 amu Coronene: 300 amu Pyrene: 202 amu Anthracene: 178 amu Naphthalene: 128 amu Number of Stacked Molecules
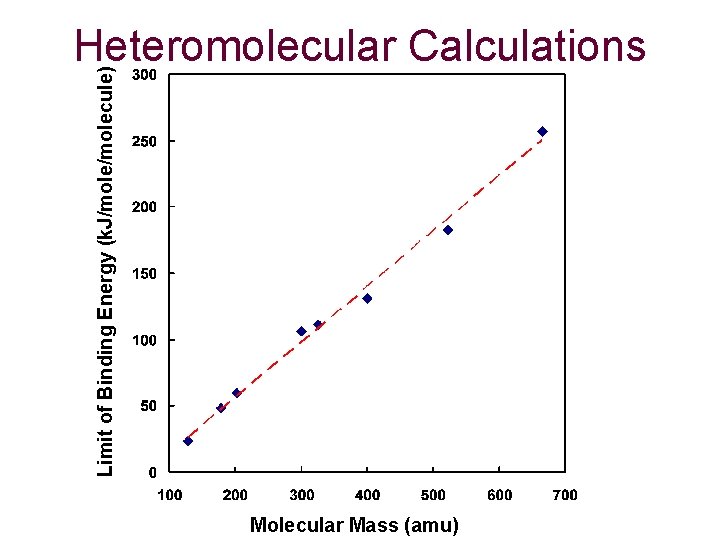
Limit of Binding Energy (k. J/molecule) Heteromolecular Calculations Molecular Mass (amu)

Conclusions and Future Atom Pair Calculations from 1984 have proven to be effective if an accurate charge set is chosen. Calculations of homomolecular dimers proves to asymptotically approach that of the binding energy of graphite (5. 02 k. J/molecule). Slightly parallel-displaced from eclipsed is generally the most favorable dimer structure. Stacks of molecules approach a limit, which increases linearly based on size. Future: Calculations of heteromolecular stacks, 3 -D clusters and applying these calculations to curved, fullerene structures.
- Slides: 16