Evidence Based Use of Sugammadex in Children Lisa

Evidence Based Use of Sugammadex in Children Lisa Herbinger, DNP, CRNA Copyright 2019. All Rights Reserved.

Conflict of Interest Disclosure Statement AANA Annual Congress 2019 Lisa Herbinger, DNP, CRNA § I have no financial relationships with any commercial interest related to the content of this activity. § I will discuss the following off-label use during my presentation: – Sugammadex for children

• Recall the pharmacokinetics of steroidal neuromuscular blocking (NMB) agents and NMB antagonists in children. Learner Outcomes • Describe the risks, benefits and contraindications of sugammadex specifically related to children and special pediatric population groups. • Discuss the available evidence and current studies being done related to the use of sugammadex in children.

Get out those cell phones and go to: http: //lisaherbinger. participoll. com

http: //lisaherbinger. participoll. com I do peds anesthesia… https: //doctoryg. blogspot. com/2016/12/g eneral-anesthesia-in-young-children. html A. B. C. D. …all day, every day. …a few times a week. …a few times a month. …never! A B C D 0 vote at lisaherbinger. participoll. com

Review: Non-Depolarizing NMBs § Non-depolarizing NMBs are competitive antagonists § NMBs bind to the α subunit of the acetylcholine receptor neuromuscular blockade § 2 classes: – Steroidal (rocuronium, vecuronium, pancuronium) – Benzylisoquinoline (atracurium, cisatracurium, mivacurium) https: //basicmedicalkey. com/acetylcholine-receptor-agonists/
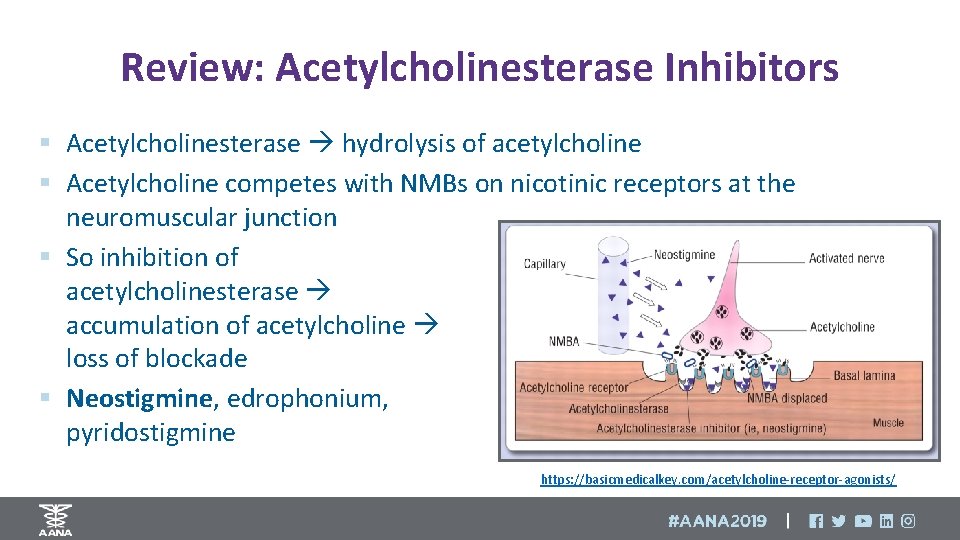
Review: Acetylcholinesterase Inhibitors § Acetylcholinesterase hydrolysis of acetylcholine § Acetylcholine competes with NMBs on nicotinic receptors at the neuromuscular junction § So inhibition of acetylcholinesterase accumulation of acetylcholine loss of blockade § Neostigmine, edrophonium, pyridostigmine https: //basicmedicalkey. com/acetylcholine-receptor-agonists/

So What Exactly is Sugammadex? § A modified, three-dimensional cyclodextrin § Conical doughnut-shape with a hydrophobic cavity and a hydrophilic ring § The hydrophobic cavity tightly traps steroidal (hydrophilic) NMB molecules a 1: 1 ratio water soluble complex. § Estimate: For every 30 million sugam/roc complexes, one dissociates. § No recurarization reported in human studies. § (Naguib, Stoelting’s Pharm. 5 th ed: 337) https: //www. researchgate. net

http: //lisaherbinger. participoll. com When was the first batch of sugammadex produced? A. 1999 B. 2002 C. 2005 D. 2008 E. 2012 www. google. com/search? q=witch+stirring+cauldron A B C D E 0 vote at lisaherbinger. participoll. com

https: //www. apsf. org/article/the-development-and-regulatory-history-of-sugammadex-in-the-united-states/

Sugammadex History § FDA rejected Merck 3 times between 2008 and 2015. § 1˚concern: Hypersensitivity anaphylaxis § U. S. FDA approval: December, 2015 § Currently in >60 countries, used in >10 million patients. https: //www. medline. com/product/Bridion-Injection/Z 05 -PF 156243 https: //www. medline. com/produ ct/Bridion-Injection/Z 05 PF 156243

Anaphylaxis has occurred in 0. 3% of healthy volunteers. Bradycardia/cardiac arrest has occurred within minutes after administration. Doses up to 16 mg/kg increased PT, PTT and INR of up to 25% for up to 1 hour in healthy volunteers. Precautions Not recommended with severe renal impairment. Most common adverse reactions (≥ 10%, all doses): Nausea, vomiting, pain, hypotension, headache. https: //www. merck. com/product/usa /pi_circulars/b/bridion_pi. pdf Patients on hormonal contraceptives must use an additional, non-hormonal method for 7 days post-administration.

http: //lisaherbinger. participoll. com The safety and effectiveness of sugammadex have not been established in patients less than _______ of age. A. B. C. D. 17 years 12 years 6 months https: //www. ks. childcareaware. org A B C D 0 vote at lisaherbinger. participoll. com

Safety and effectiveness have not been established in patients less than 17 years of age. https: //www. merck. com/product/usa/pi_circulars/b/bridion_pi. pdf

Ethics and Peds Drug Research § Paradigm shift: “We must protect children from research” to “We must protect children through research” § Current legislation requires pediatric trials be done. – Best Pharmaceuticals for Children’s Act (2002) and Pediatric Research Equity Act (2003) – Provide additional 6 months patent time for “pediatric exclusivity” § Since 1998: – 1, 000 peds clinical drug trials – Over 25% failed to show efficacy in children – Over 700 drug label updates specific to children (Baum, Pediatr Anes, 2019) http: //www. childrenshospital. org

http: //lisaherbinger. participoll. com What exactly does “not approved” mean? A. B. C. D. The drug is considered contraindicated. Administration would be considered unethical. Administration would be considered illegal. The drug has not moved through the FDA clinical trial process. A B C D 0 vote at lisaherbinger. participoll. com

Until officially approved… § Despite labeling, we are within medical and legal rights to use these drugs. § Not approved does NOT mean improper, illegal, contraindicated or investigational. https: //www. google. com/imghp? hl=en § American Academy of Pediatrics: Practitioners should use their professional judgment to determine these uses. . . Therapeutic decision making must always rely on the best available evidence and the importance of the benefit for the individual patient. (AAP, 2014)

Consider this… Your next patient is a 5 year old in the ER who arrived vomiting blood. He is 4 days post-tonsillectomy. The ENT surgeon has posted him for an emergent cauterization. You do not have access to his previous anesthetic records. Mom tells you, “My 3 year old nephew has muscular dystrophy so my son had special anesthesia for his tonsil surgery. Are you going to do that again today? ” 4 Assumptions § Emergent § High risk for aspiration § ↑K+, rhabdomyolysis & CV arrest susceptible § Less than 10 -minute procedure time https: //www. medicalnewstoday. co m

“Current evidence for the use of sugammadex in children” (educational review) § Approx. 200 patients (2 – 12 years) from 3 RCTs: More rapid and effective reversal than neostigmine § Recommended dosing – 2 mg/kg with ≥ 2 TOF twitches – 4 mg/kg with 1 -2 post-tetanic twitches – 16 mg/kg (max dose) if immediately following an intubating dose (can’t intubate; can’t ventilate) § After 2, 4 or 16 mg/kg, return to TOF ratio of 0. 9 averaged 2. 2 minutes. (Tobias, Ped Anesth, 2017)

What if you cannot trust (or use) your NMB monitor? § 2 TOF twitches = 75 -80% block = approx. 28 minutes (0. 6 mg/kg) § Roc 0. 6 – 1 mg/kg 60 minute duration in infants and children § In neonates, roc 0. 6 mg/kg 90 minute duration with ↑variability (Anderson, Cote 6 th ed: 150) § The Lisa Method § For 0. 6 - 1 mg/kg (infant or child): 4 mg/kg if <30 minutes. § Neonates: Use at least 4 mg/kg. (Tobias, Ped Anesth, 2017)

Comparing Sugammadex to Neostigmine in Children § Faster recovery, faster extubation, less adverse events (Ammar, Acta Anaesth Scandinavica, 2017) § Facilitates earlier extubation; early off-label use in peds has been positive (Buck, Pediatr Pharm, 2017) § Faster recovery with less incidence of bradycardia (Liu, Sci Rep, 2017) § No increased incidence of adverse events (Won, Medicine, 2016) § 4 mg/kg is effective and safe in early reversal of a deep NMB in infants and children (Benigni, Paediatr Anaesth Int Care, 2013)

http: //lisaherbinger. participoll. com Regarding anaphylaxis, what is considered the “critical time” following administration? A. The first minute B. The first 5 minutes C. The first 15 minutes D. The first 30 minutes A B C D 0

What about Sugammadex-Induced Anaphylaxis in Children? § § University of Tokyo Approx. 840, 000 pts, <18 years of age, GA (national database, 2010 -2016) Retrospective, case-controlled matching No association between sugammadex and anaphylaxis § § Ben-Gurion University of the Negev, Be’er Sheva, Israel Review article, characteristics of hypersensitivity reactions with sugammadex Critical 5 -min period immediately following administration Mean time of reaction onset: 1 min 52 sec (Tadakoro, Pediatr Anesth, 2018) (Tsur, Anaesthesia, 2014)

Sugammadex-Induced Bradycardia in Children? § Nationwide Children’s Hospital, Columbus, OH § Prospective, 200 children <18 years of age, HR measured q minute x 15 minutes, then q 5 minutes until out of OR § Bradycardia = HR below 5 th percentile for age § Bradycardia in 15 (8%), 7 of those (47%) had documented heart disease § No change in BP, no treatment needed. § Cardiac co-morbidities and male gender considered risk factors. (Sims, Crit Care Med, 2018)

Dexamethasone and Sugammadex in Children? § Alexandria Faculty of Medicine, Alexandria, Egypt § 80 patients, 1 -6 years of age, ASA I or II, strabismus or T&A § Times to reach 90% TOF (60 vs. 23 sec) and extubation (73 vs. 38 sec) were statistically higher in the dexamethasone (0. 5 mg/kg) group than the control group. (Saleh, Egypt J Anaesth, 2017) § Cukurova University School of Medicine, Adana, Turkey § RCT, 60 patients, 3 -8 years of age, ASA I or II, T&A § No difference in times to reach recovery (98 vs. 91 sec) and extubation (128 vs. 124 sec) in the dexamethasone 0. 5 mg/kg group vs. control respectively. (Gulec, Anesth Analg, 2016)

Sugammadex and Children < 2 years § Retrospective observational study from 2 year period – neostigmine vs. sugammadex § 331 sugammadex doses in children < 2 years of age § Average time to leaving OR not statistically different. https: //basicsofpediatricanesthesia. com § Average time to extubation less with sugammadex (84 sec vs. 103 sec). § Conclusions: Sugammadex equally effective as neostigmine in children <2 years of age with no adverse effects. (Franz, Pediatr Anesth, 2019)

Sugammadex and Infants § Retrospective observational study § 26 infants, 2 -12 months of age, 3 -11 kg, ASA I, II, short neurosurgical procedures § Induction with 0. 6 mg/kg rocuronium, maintenance with sevoflurane, roc re-dosed throughout procedure. § At procedure completion, pts with TOF 0/4 were given sugammadex 3 mg/kg. § Mean recovery time to TOF ratio 0. 9: 113 seconds (< 2 minutes) § No incidence of nausea, vomiting, bradycardia, tachycardia, rash or hypersensitivity. (Ozmete, Clin Anesth, 2016)

Sugammadex and Neonates… § Case report: § 1. 8 kg neonate with TE fistula, 1 mg/kg of rocuronium after awake intubation with dexmedetomidine sedation. § Difficult ventilation/desaturation began a few minutes post-rocuronium with + pressure ventilation air entering the abdomen. § Sugammadex 8 mg/kg given. § Assisted ventilation until spontaneous resumed (1 -2 minutes). § “Future anecdotal experience and prospective trials needed. ” (Wakimoto, J Med Cases, 2018) https: //basicsofpediatricanesthesia. com/ cool-movies/neonatal-mask-ventilation/

Finishing neonates… § Case report: § 2 day old, 3. 3 kg, TE fistula, intubated in OR during rigid bronchoscopy § GA: Desflurane, remifentanil and rocuronium infusions – Rocuronium @ 1 mg/kg/hr for the first 3 hrs – 0. 5 mg/kg/hr for the 4 th hr – D/C’d 30 min prior to the end of surgery. § After 2 post-op hours, VS stable but no spontaneous movement or respirations, bilateral fixed and dilated pupils. § Cerebral ultrasound negative. § Full reversal dose of neostigmine no improvement. § Sugammadex 16 mg/kg given spontaneous ventilation, combative extremity movement and reactive pupils bilaterally within 90 seconds. (Langley, Pediatr Anesth, 2015)

Sugammadex “Others” § Did not affect emergence agitation compared with conventional cholinesterase inhibitors (Kim, J Int Med Res, 2018) § More rapid and safer for asthmatic peds patients having lower abdominal or urogenital surgeries (Nada, Res Opinion Anesth Int Care, 2017) § No adverse events with 2 mg/kg in 11 year old with Duchenne muscular dystrophy (Kim, Medicine, 2017) § Rapid, effective and reliable reversal in children undergoing tonsillectomy with no side effects (El sayed, Egypt J Anaesth, 2016)

Happening now… § Efficacy, Safety, and Pharmacokinetics of Sugammadex for Reversal of Neuromuscular Blockade (NMB) in Pediatric Participants § 8 U. S. and 19 international peds facilities, 2/18 – 7/20 § Phase 4, triple blinded RCT, 2 -16 years of age, ASA I, II or III – – Sugammadex 2 mg/kg Sugammadex 4 mg/kg Neostigmine + Glycopyrrolate Neostigmine + Atropine § 7 primary outcomes, 2 secondary outcomes (National Library of Medicine) https: //clinicaltrials. gov/ct 2/show/NCT 03351608? term=sugammadex+AND+children&recrs=ab&rank=5)

#sugamforkids § Sugammadex is a unique NMB agent that can reverse a deep block to spontaneous ventilation in less than 2 minutes. § Although sugammadex is not approved for children <17 years of age, there is evidence that we can safely and ethically use it for pediatrics. § There is evidence that sugammadex can be safely used for children, infants and neonates. § Studies are ongoing. More peds research is needed. https: //www. google. com

References American Academy of Pediatrics (AAP). Policy statement: Off-label use of drugs in children. Pediatrics. 2014; 133: 563– 567. Ammar AS, Mahmoud KM, Kasemy ZA. A comparison of sugammadex and neostigmine for reversal of rocuronium-induced neuromuscular blockade in children. Acta Anaesthesiol Scandinavica. 2017; 61: 374– 380. doi: 10. 1111/aas. 12868 Anderson BJ, Lerman J, Cote CJ. Pharmacokinetics and pharmacology of drugs used in children. In: Cote CJ, Lerman J, Anderson BJ, eds. Cote and Lerman’s A Practice of Anesthesia for Infants and Children. 6 th ed. Philadelphia, PA: Elsevier; 2019: 150 -152. Baum VC, Bax R, Heon D, Yang Z, Salkyama M. Pediatric drug regulations: International perspectives. Pediatr Anesth. 2019; 29: 572 -582. Benigni A, Mafflioletti M, Spotti A, Benigni AM, Locatelli BG, Sonzogni. Efficacy and safety of sugammadex dose of 4 mg/kg in early reversal of a deep neuromuscular block rocuronium-induced in infants and children: a case series: 10 AP 2 -10. Eur J Anaesthesiol. 2013; 30 Suppl 51: 161 -162. Buck ML. Sugammadex use in infants and children. Ped Pharmacotherapy, Univ VA Child Hosp. 2017; 23(3). https: //med. virginia. edu/pediatrics/wpcontent/uploads/sites/237/2015/12/Mar 17_Sugammadex_Ped. Pharmaco. pdf Accessed on October 22, 2018.

El sayed M, Hassan S. Does sugammadex facilitate recovery after outpatient tonsillectomy in children? Egypt J Anaesth. 2016; 32: 447 -450. Franz AM, Chiem J, Martin LD, Rampersad S, Phillips J, Grigg EB. Case series of 331 doses of sugammadex compared to neostigmine in patients under 2 years of age. Pediatr Anesth. 2019; 29: 591 -596. Gulec E, Biricik E, Turktan M, Hatipoglu Z, Unlugenc H. The effect of intravenous dexamethasone on sugammadex reversal time in children undergoing adenotonsillectomy. Anes Analg. 2016; 122(4): 1147 -1152. Kim JE, Chun HR. Rocuronium-induced neuromuscular block and sugammadex in pediatric patient with Duchenne muscular dystrophy - A case report. Medicine. 2017; 96: 13. Kim YS, Cha JR, Lee YS, Kim WY, Kim JH, Kim YH. Sugammadex affects emergence agitation in children undergoing strabismus surgery. J Int Med Res. 2018: 46(9): 3861– 3872. Langley RJ, Mc. Fadzean J, Mc. Cormack J. The presumed central nervous system effects of rocuronium in a neonate and its reversal with sugammadex. Ped Anesth. 2016; 26: 109– 111.

Liu G, Wang R, Yan Y, Fan L, Xue J, Wang T. The efficacy and safety of sugammadex for reversing postoperative residual neuromuscular blockade in pediatric patients: A systematic review. Scientific Rep. 2017; 7: 5724. doi: 10. 1038/s 41598 -017 -06159 -2 Merck Sharp & Dohme Corporation. Bridion® (sugammadex) injection. https: //www. merck. com/product/usa/pi_circulars/b/bridion_pi. pdf Accessed October 22, 2018. Nada EN. Efficacy of sugammadex compared with neostigmine for reversal of rocuronium-induced neuromuscular blockade and deep extubation in outpatient surgeries for asthmatic pediatric patients. Res Opinion Anesth Intensive Care. 2017; 4: 143– 148. Naguib MA. Neuromuscular blocking drugs and reversal agents. In: Flood P, Rathmell JP, Shafer S, eds. Stoelting’s Pharmacology and Physiology in Anesthetic Practice. 5 th ed. Philadelphia, PA: Wolters Kluwer; 2014: 337 -339. National Library of Medicine. Efficacy, safety, and pharmacokinetics of sugammadex for reversal of neuromuscular blockade (NMB) in pediatric participants (MK-8616 -089). https: //clinicaltrials. gov/ct 2/show/NCT 03351608? term=sugammadex+AND+children&draw=2&rank=13 Accessed July 20, 2019. Ozmete OO, Bali C, Cok OY, et al. Sugammadex given for rocuronium-induced neuromuscular blockade in infants: a retrospectıve study. J Clin Anes. 2016; 35: 497– 501.

Saleh RS, Moustafa MA. Recovery from rocuronium with sugammadex in children premedicated with dexamethasone for prevention of postoperative nausea and vomiting. Egypt J Anaesth 2017; 33: 1– 4. Sims T, Alsuhebani M, Tobias J, et al. Heart rate changes following the administration of sugammadex in children. Crit Care Med. 2018; 47(1 supplement): 988. Tadokoro F, Morita K, Michihata N, Fushimi K, Yasunaga H. Association between sugammadex and anaphylaxis in pediatric patients: A nested case-control study using a national inpatient database. Ped Anesth. 2018; 28: 654– 659. Tobias JD. Current evidence for the use of sugammadex in children. Ped Anesth. 2016; 27: 118– 125. Tsur A, Talansky A. Hypersensitivity associated with sugammadex administration: a systematic review. Anaesthesia. 2014. doi: 10. 1111/anae. 12736 Wakimoto M, Burriera C, Tobias JD. Sugammadex for rapid intraoperative reversal of neuromuscular blockade in a neonate. J Med Cases. 2018; 9(12): 400 -402. Won YJ, Lim BG, Lee DK, Kim H, Kong MH, Lee O. Sugammadex for reversal of rocuronium-induced neuromuscular blockade in pediatric patients - A systematic review and meta-analysis. Medicine. 2016; 95(34): 1 -7.

ml le 2. ht u d o m atc ing/ n i a r t hme : / https /www dd/w d b c n ov/. cdc. g lallison@samford. edu It’s Been Fun Y’all!!! Thank you!

Wood Library Museum of Anesthesiology https: //www. woodlibrarymuseum. org/
- Slides: 38