Evidence Based Guide to Gestational Diabetes Robert Fraser

Evidence Based Guide to Gestational Diabetes Robert Fraser University of Sheffield. UK.

Treatments for Gestational Diabetes and Impaired Glucose Tolerance ( Cochrane Systematic Reviews 2003) “ It is uncertain whether intensive treatment can influence birth weight and reduce perinatal morbidity i. e. is there any benefit to treating women with GDM or IGT in pregnancy? ”

NICE: Antenatal Care Guidelines (2003) “there is an absence of evidence to support routine screening for gestational diabetes mellitus and therefore it is not recommended”.

So - Is GDM a disease capable of causing adverse perinatal outcomes? • If so – is there effective treatment available? • If so – are there effective screening protocols?


ACHOIS Trial: Research Question Does treatment of women with screen detected impaired glucose tolerance in pregnancy reduce perinatal morbidity, without increasing maternal physical and psychological morbidity?
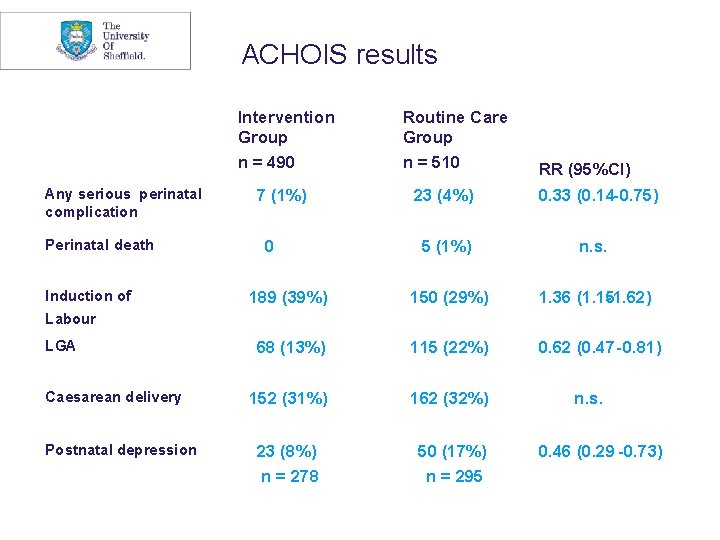
ACHOIS results Any serious perinatal complication Perinatal death Induction of Intervention Group Routine Care Group n = 490 n = 510 7 (1%) 0 23 (4%) RR (95%CI) 0. 33 (0. 14 -0. 75) 5 (1%) n. s. 189 (39%) 150 (29%) 1. 36 (1. 15 - 1. 62) 68 (13%) 115 (22%) 0. 62 (0. 47 -0. 81) 152 (31%) 162 (32%) 23 (8%) 50 (17%) n = 278 n = 295 Labour LGA Caesarean delivery Postnatal depression n. s. 0. 46 (0. 29 -0. 73)

ACHOIS Trial: Conclusions “ Treatment of gestational diabetes reduces serious perinatal morbidity and may also improve the women’s health –related quality of life. ”

NICHD Maternal Fetal Medicine Units : Prospective multicenter Randomised Treatment Trial of Mild Gestational Diabetes. 958 women with GDM by ADA criteria (100 g GTT) Double blind randomisation. Primary endpoint: Composite of PNM, and Neonatal Morbidity (Hyperbiliruinaemia, hypoglycaemia, hyperinsulinaemia, birth trauma) (Landon et al 2009)
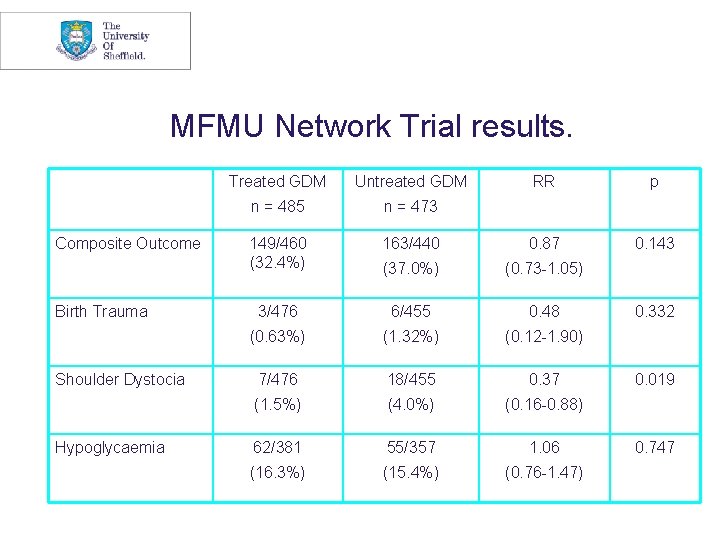
MFMU Network Trial results. Composite Outcome Birth Trauma Shoulder Dystocia Hypoglycaemia Treated GDM Untreated GDM RR p n = 485 n = 473 149/460 (32. 4%) 163/440 0. 87 0. 143 (37. 0%) (0. 73 -1. 05) 3/476 6/455 0. 48 (0. 63%) (1. 32%) (0. 12 -1. 90) 7/476 18/455 0. 37 (1. 5%) (4. 0%) (0. 16 -0. 88) 62/381 55/357 1. 06 (16. 3%) (15. 4%) (0. 76 -1. 47) 0. 332 0. 019 0. 747
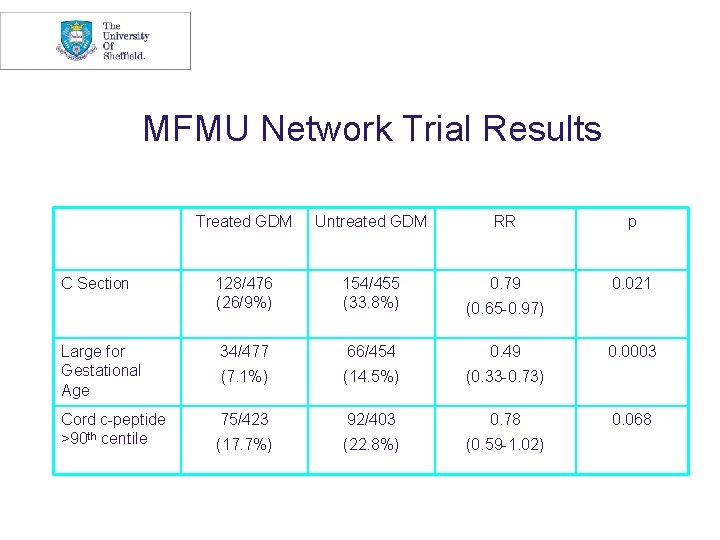
MFMU Network Trial Results Treated GDM Untreated GDM RR p 128/476 (26/9%) 154/455 (33. 8%) 0. 79 0. 021 (0. 65 -0. 97) Large for Gestational Age 34/477 66/454 0. 49 (7. 1%) (14. 5%) (0. 33 -0. 73) Cord c-peptide >90 th centile 75/423 92/403 0. 78 (17. 7%) (22. 8%) (0. 59 -1. 02) C Section 0. 0003 0. 068

• Two recently published RCTs suggest treatment of mild GDM is effective. • If GDM causes pathological outcomes which respond to treatment it follows that screening is justified.

Gestational Diabetes Mellitus • How should screening be performed? • Does GDM cause an increase in Congenital Malformations? • Can GDM management be more effective? • Does timing of delivery in GDM affect outcomes?

Screening for GDM by Risk Factors ( Helton 1997) Obesity, FH Diabetes, Previous pregnancy failure, Previous macrosomia. Sensitivity Specificity 69% 68% PPV 5% (High risk ethnic minority groups)

Screening for GDM by 50 g Glucose Challenge Test 50 g glucose in 150 ml H 2 O: Unprepared subject at 24 -28 weeks gestation Positive Test – Plasma Glucose >7. 8 mmol/l Sensitivity 79% Specificity 87% PPV 15% ( O’Sullivan 1973)

Diagnostic Screening by 75 g glucose load after overnight fast Community based population screening in Southern Sweden 2 h value only recorded: interpreted by WHO Criteria ( Aberg 2001)

ADA: Low Risk Criteria for GDM • Age < 25 yrs • Normal weight • From low prevalence ethnic group Can be excluded from screening


HAPO Study 23, 325 women were recruited at random. 75 g OGTT at 24 -32 weeks gestation. Results unblinded if FPG > 5. 8 mmol/l 0 r 2 hr >11. 1 mmol/l. If symptomatic may have Random Plasma Glucose at 34 -37 weeks – unblinded if >8. 9 mmol/l.

HAPO Study-Primary Outcomes Caesarean delivery Increased Fetal Size Neonatal Hypoglycaemia Fetal Hyperinsulinism
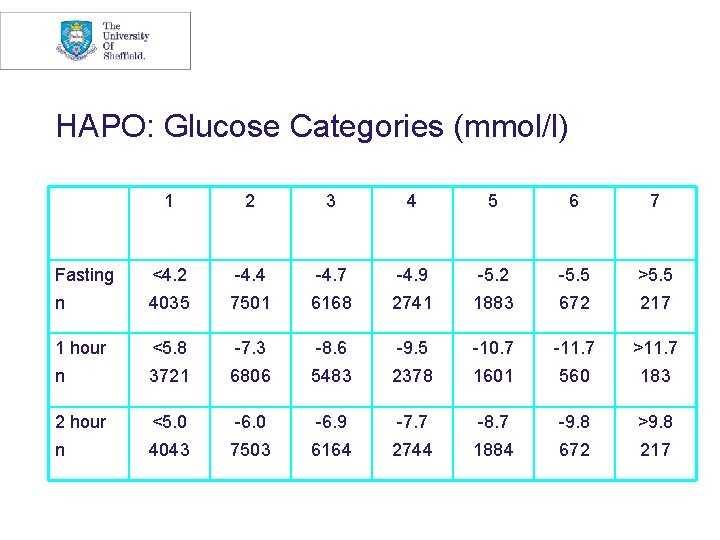
HAPO: Glucose Categories (mmol/l) 1 2 3 4 5 6 7 Fasting <4. 2 -4. 4 -4. 7 -4. 9 -5. 2 -5. 5 >5. 5 n 4035 7501 6168 2741 1883 672 217 1 hour <5. 8 -7. 3 -8. 6 -9. 5 -10. 7 -11. 7 >11. 7 n 3721 6806 5483 2378 1601 560 183 2 hour <5. 0 -6. 9 -7. 7 -8. 7 -9. 8 >9. 8 n 4043 7503 6164 2744 1884 672 217
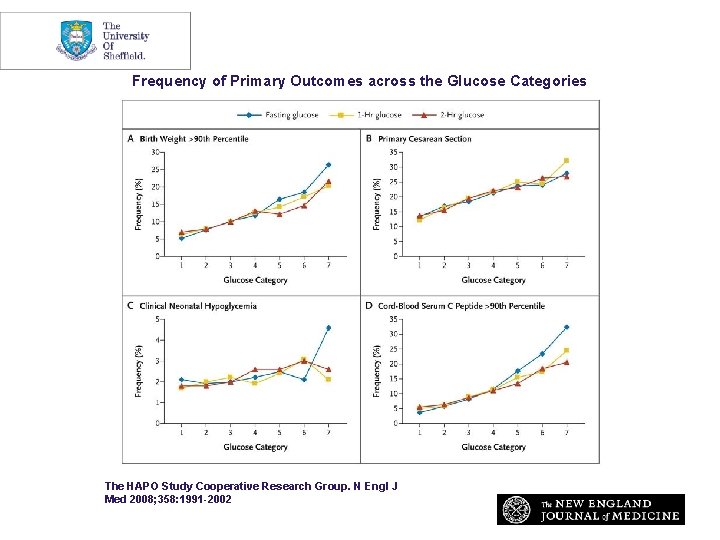
Frequency of Primary Outcomes across the Glucose Categories The HAPO Study Cooperative Research Group. N Engl J Med 2008; 358: 1991 -2002

HAPO Study Concensus on revised diagnostic criteria. • Risk factor positive; if FPG >7. 0 mmol/l, or random glucose above 11. 1 mmol/l, or HB A 1 c >6. 5% consider as likely overt diabetes and treat accordingly. • Or if universal blood testing : 75 g load • Fasting plasma glucose >5. 1 mmol/l • And/or 1 hr level >10. 0 mmol/l • ? ? include 2 hr > 8, 5 mmol/l
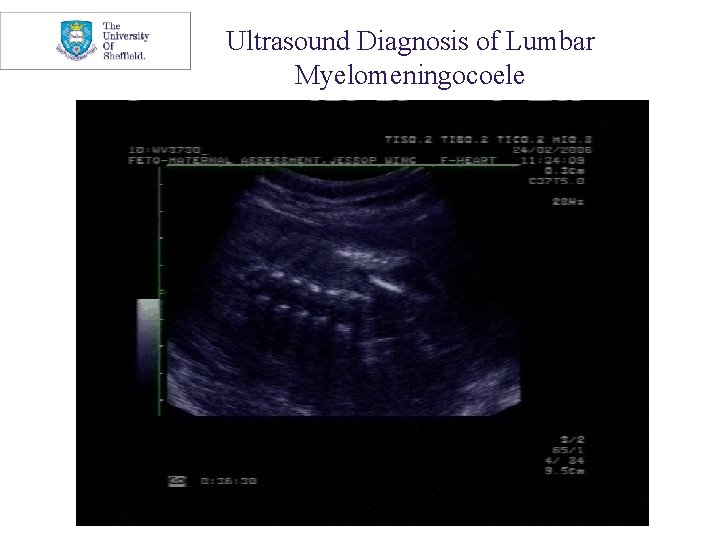
Ultrasound Diagnosis of Lumbar Myelomeningocoele

Congenital Malformation Rates - Sheffield Audit Type 1/2 GDM/IGT 4 1 Major malformations at birth 12 7 Percent major malformations (7. 6%) (2. 0%) Minor malformations at birth 1 12 TOP for congenital malformations

Congenital Malformations in Gestational Diabetes Major Congenital Malformations (%) Type I diabetes (95% CI) 5. 9 (3. 2 – 9. 8) Type 2 diabetes 4. 4 (2. 4 – 7. 3) Gestational diabetes 1. 4 (0. 9 – 2. 0) but New onset Type 2 in GDM group 4. 6 (2. 3 – 8. 2) (13% of 1822) (Farrell et al 2002)

Patterns of Malformations and Relationships to Initial Maternal Fasting Serum Glucose Levels in Gestational Diabetes Initial FPG No Malformations Major Malformations Aneuploidy n = 3895 n = 143 n = 21 6. 4 ± 2. 0 8. 0 ± 3. 1 6. 4 ± 2. 1 level mmol/L ± SD (Schaefer-Graf et al 2000)
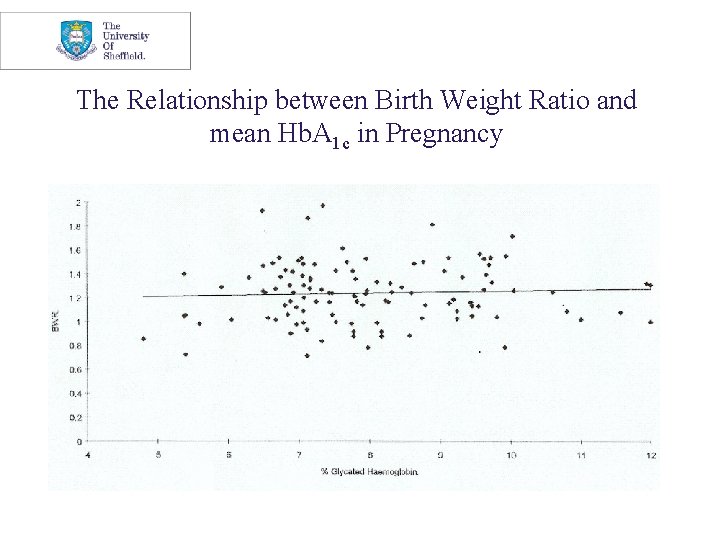
The Relationship between Birth Weight Ratio and mean Hb. A 1 c in Pregnancy
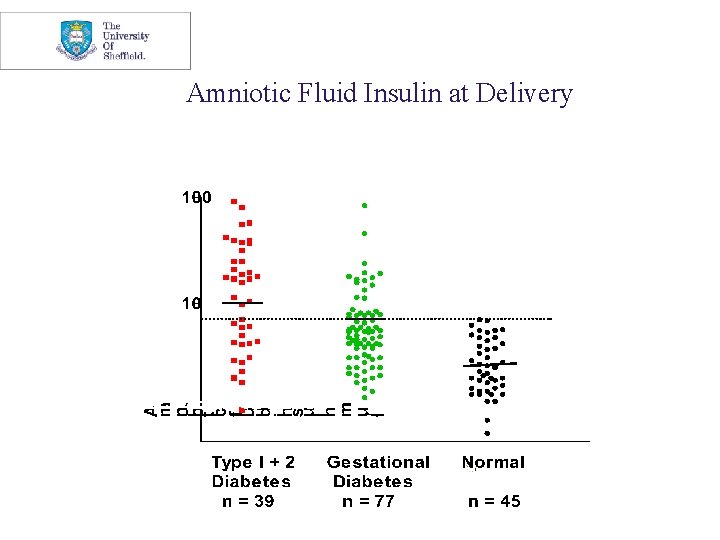
Amniotic Fluid Insulin at Delivery
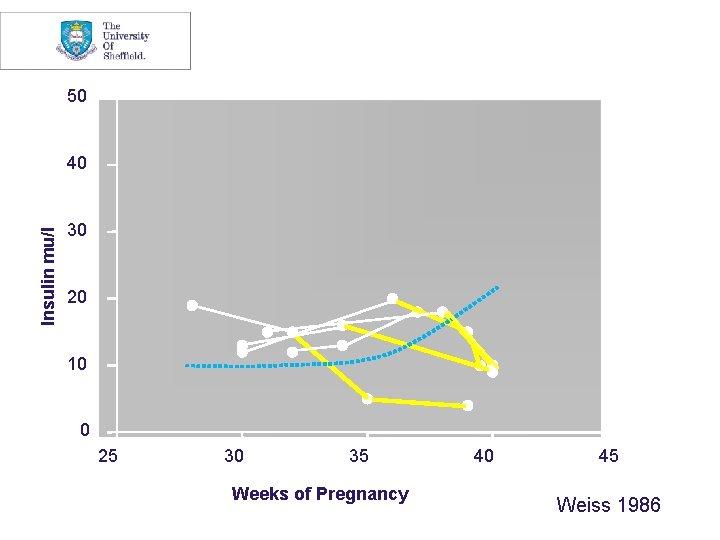
50 Insulin mu/l 40 30 20 10 0 25 30 35 Weeks of Pregnancy 40 45 Weiss 1986

Percentage of LGA in infants according to Quintiles of Carbohydrate Intake in GDM LGA <173 g 173 -186 186 -211 211 -243 >243 5 (37%) 2 (12%) 4 (25%) 0 (0%) 2 (13%) 0 (0%) 1 (6%) SGA 0 (0%) Romon et al 2001

Gestational Diabetes; can a low glycemic index diet reduce the need for insulin? A randomised trial. • 63 women randomised to low GI diet or conventional High Fibre diet. • Low GI 9/31 (29%) required insulin. • High Fibre 19/32 (59%)required insulin • (P= 0. 023) (Moses et al 2009)

RCT of Glycaemic Parameters ± Fetal Ultrasound to Determine Insulin Therapy in GDM • 98 women with FGP 5. 8 – 6. 7 mmol/L • Experimental group – insulin only if AC ≥ 70 th centile or Plasma Glucose > 6. 7 mmol/L • Control group – all received insulin (targets: preprandial ≤ 5. 0, 2 h postprandial ≤ 6. 7) (Kjos et al 2001)

RCT of Glycaemic Parameters ± Fetal Ultrasound to Determine Insulin Therapy in GDM Outcomes Controls Experimental Group Insulin Rx 100% 62% Birthweight 3. 27 Kg ± 0. 5 3. 37 ± 0. 5 BWT >90 th centile 6. 3% 8. 3% Neonatal Morbidity 25%

Pre-prandial vs Post-prandial BG monitoring in Insulin treated GDM RCT 66 women with GDM by ADA Criteria : Diagnosed before 30 weeks. Pre-prandial Group : Insulin adjusted to keep fasting glucose 3. 3 - 5. 0 mmol/L and pre-prandial 3. 3 – 5. 9 mmol/L Post-prandial Group: Insulin adjusted to keep fasting glucose 3. 3 - 5. 0 mmol/L and 1 hr post prandial < 7. 8 mmol/l Veciana et al 1995
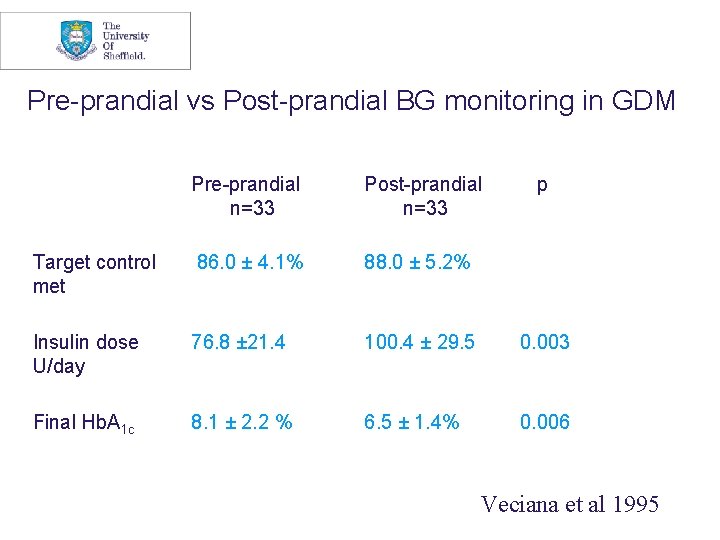
Pre-prandial vs Post-prandial BG monitoring in GDM Pre-prandial n=33 Post-prandial n=33 p Target control met 86. 0 ± 4. 1% 88. 0 ± 5. 2% Insulin dose U/day 76. 8 ± 21. 4 100. 4 ± 29. 5 0. 003 Final Hb. A 1 c 8. 1 ± 2. 2 % 6. 5 ± 1. 4% 0. 006 Veciana et al 1995
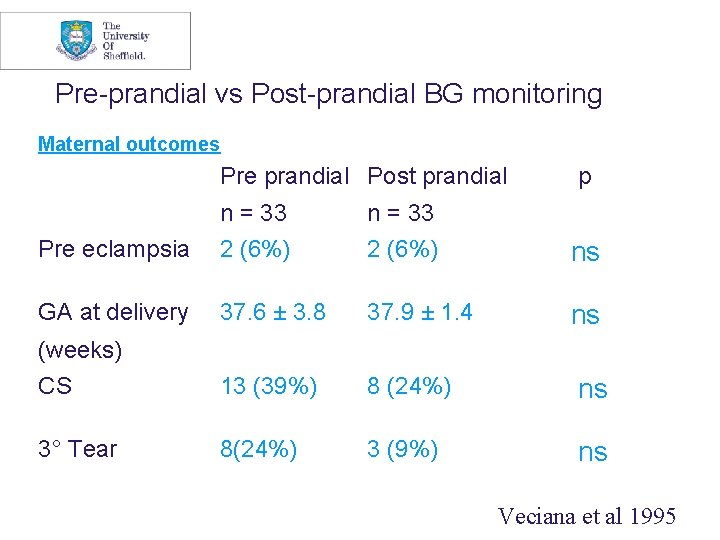
Pre-prandial vs Post-prandial BG monitoring Maternal outcomes Pre prandial Post prandial p n = 33 Pre eclampsia 2 (6%) ns GA at delivery 37. 6 ± 3. 8 37. 9 ± 1. 4 ns (weeks) CS 13 (39%) 8 (24%) 3° Tear 8(24%) 3 (9%) ns ns ns Veciana et al 1995
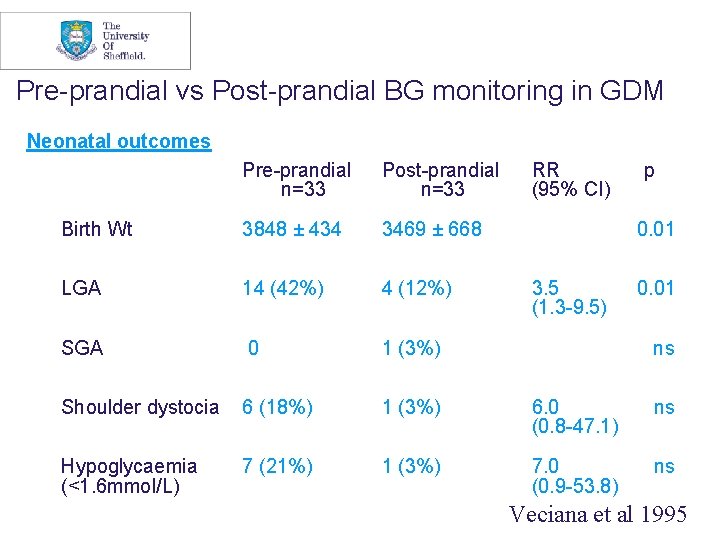
Pre-prandial vs Post-prandial BG monitoring in GDM Neonatal outcomes Pre-prandial n=33 Post-prandial n=33 RR (95% CI) p Birth Wt 3848 ± 434 3469 ± 668 LGA 14 (42%) 4 (12%) SGA 0 1 (3%) Shoulder dystocia 6 (18%) 1 (3%) 6. 0 (0. 8 -47. 1) ns Hypoglycaemia (<1. 6 mmol/L) 7 (21%) 1 (3%) 7. 0 (0. 9 -53. 8) ns 0. 01 3. 5 (1. 3 -9. 5) 0. 01 ns Veciana et al 1995

A Comparison of Glyburide (glibenclamide) and Insulin in Women with GDM 404 GDM randomised to Glyburide 2. 5 mg od rising to 20 mg/day – then commenced on insulin (n=8, 4%) or insulin 0. 7 u/Kg with weekly adjustment (Langer 2000)
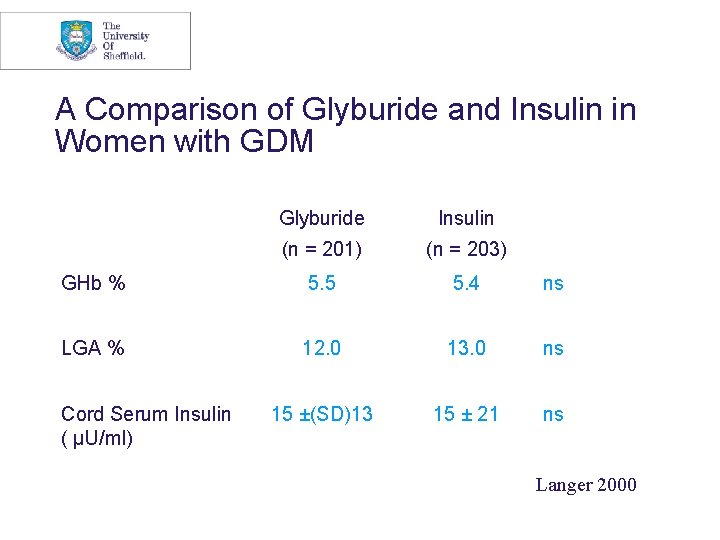
A Comparison of Glyburide and Insulin in Women with GDM Glyburide Insulin (n = 201) (n = 203) GHb % 5. 5 5. 4 ns LGA % 12. 0 13. 0 ns 15 ±(SD)13 15 ± 21 ns Cord Serum Insulin ( µU/ml) Langer 2000
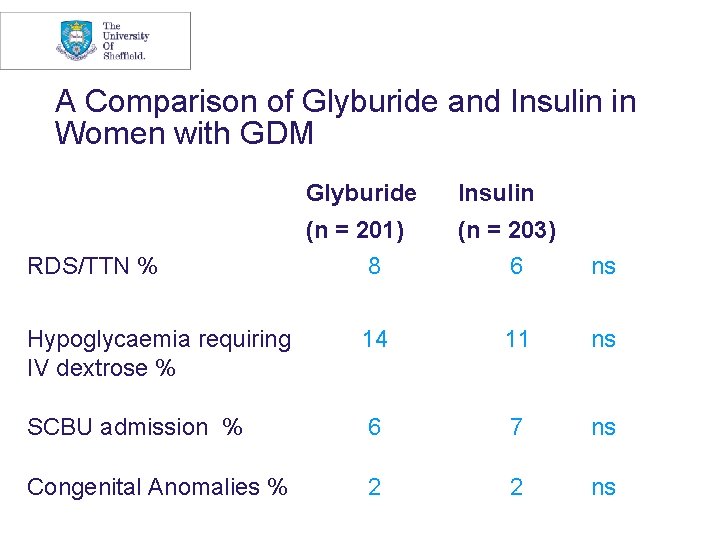
A Comparison of Glyburide and Insulin in Women with GDM Glyburide Insulin (n = 201) (n = 203) RDS/TTN % 8 6 ns Hypoglycaemia requiring IV dextrose % 14 11 ns SCBU admission % 6 7 ns Congenital Anomalies % 2 2 ns

Metformin versus Insulin for the Treatment of Gestational Diabetes (Mi. G) New England Journal of Medicine 2008 358: 2003 -15

Mi. G Study Randomised to Insulin or Metformin If targets not met with Metformin alone – up to 2500 mg/day) – insulin added

Mi. G STUDY Metformin Group Insulin Group n = 363 195 Metformin alone n = 370 168 Metformin and Insulin
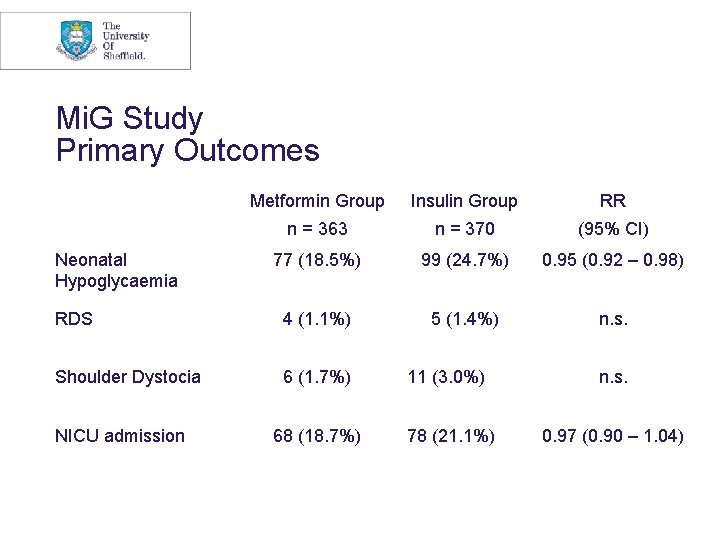
Mi. G Study Primary Outcomes Metformin Group Insulin Group RR n = 363 n = 370 (95% CI) 77 (18. 5%) 99 (24. 7%) 0. 95 (0. 92 – 0. 98) RDS 4 (1. 1%) 5 (1. 4%) n. s. Shoulder Dystocia 6 (1. 7%) 11 (3. 0%) n. s. 68 (18. 7%) 78 (21. 1%) 0. 97 (0. 90 – 1. 04) Neonatal Hypoglycaemia NICU admission

Mi. G Study Secondary Outcomes Maternal Weight (from enrolment to 6 weeks postpartum) Metformin Group Insulin Group n = 363 n = 370 -8. 1 ± 5. 1 Kg -6. 9 ± 5. 3 Kg p 0. 006

Timing of Delivery in Gestational Diabetes

Insulin requiring diabetes in pregnancy: A randomised trial of active induction of labour and expectant management 200 subjects (187 GDM 13 Type 2) Randomised at 38 weeks gestation Active group: Induction within 5 days Expectant group: Twice weekly CTG and weekly Amniotic Fluid Volume measurement until labour (Kjos et al 1993)
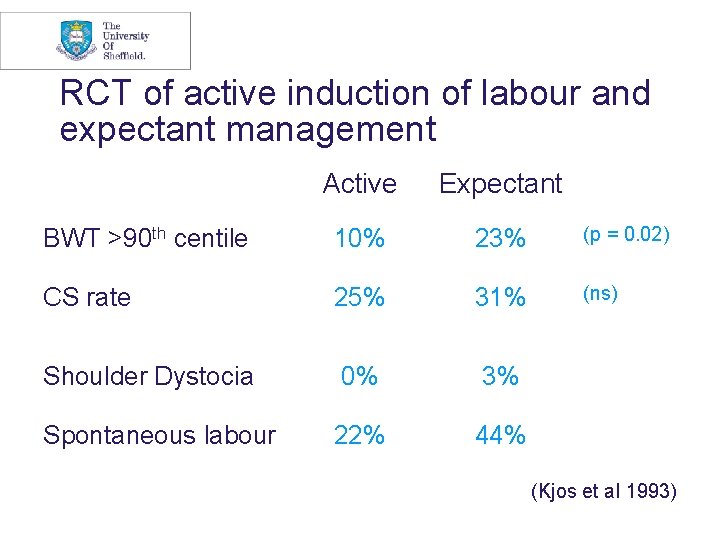
RCT of active induction of labour and expectant management Active Expectant BWT >90 th centile 10% 23% (p = 0. 02) CS rate 25% 31% (ns) Shoulder Dystocia 0% 3% Spontaneous labour 22% 44% (Kjos et al 1993)



Mi. G Hypothesis In women with gestational diabetes metformin treatment, compared to insulin, will: • Result in similar perinatal outcomes • Improve insulin sensitivity in the mother and baby • Be associated with improved treatment acceptability

Mi. G Study 751 women with GDM: Considered for Insulin therapy if Fasting Capillary Glucose >5. 4 mmol/l or 2 h postprandial >6. 7 mmol/l
- Slides: 56