Everything MPL 1203 Early Postulates What is everything














- Slides: 24

Everything MPL 1203

Early Postulates What is everything (Matter)made of? Greek Mythology –Fire, Air, Water, Earth Leucippius (Born-50 BCE) Democritus (Born – 460 BCE) Postulated an indivisible form of matter THE ATOM Aristotle in “Metaphysics”----- The shape and existence of all things are determined by void and atoms through the differences in shape, position and arrangement of the atoms and the proportion of void in the substance.

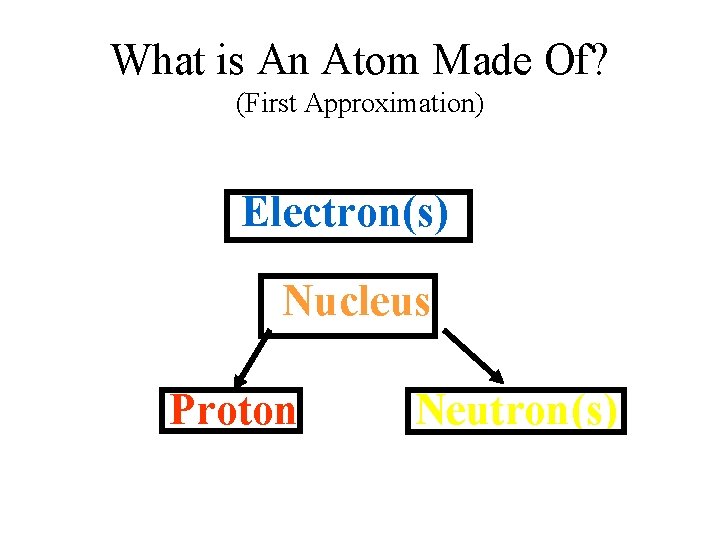
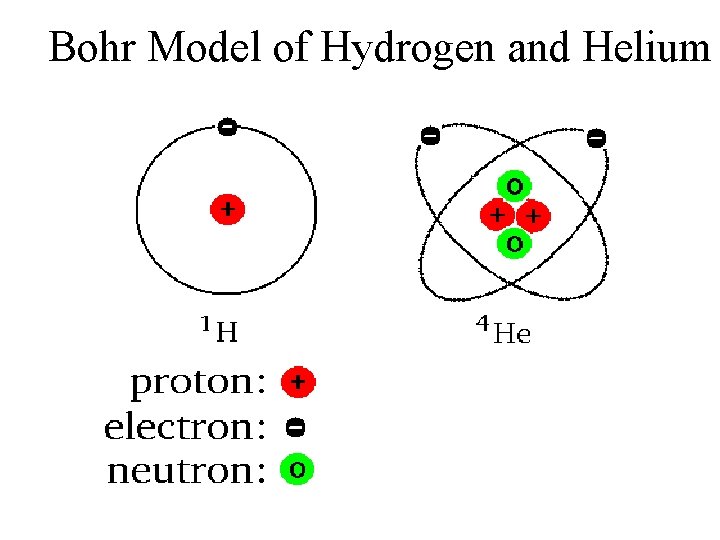
What is An Atom Made Of? (First Approximation) Electron(s) Nucleus Proton Neutron(s)

Discovery of the Electron 1897 J. J. Thompson 1899 Charge of the Electron Measured Electrons Missing Target Electrons Hitting Target Crookes Tube Vacuum Tube

Electrons, Electricity? ? ? Electricity was thought of as a stream, (analogous to water flowing as a pipe) Now thought of as a the motion of Discrete Particles having mass (Electrons) Carrying the smallest possible unit of charge

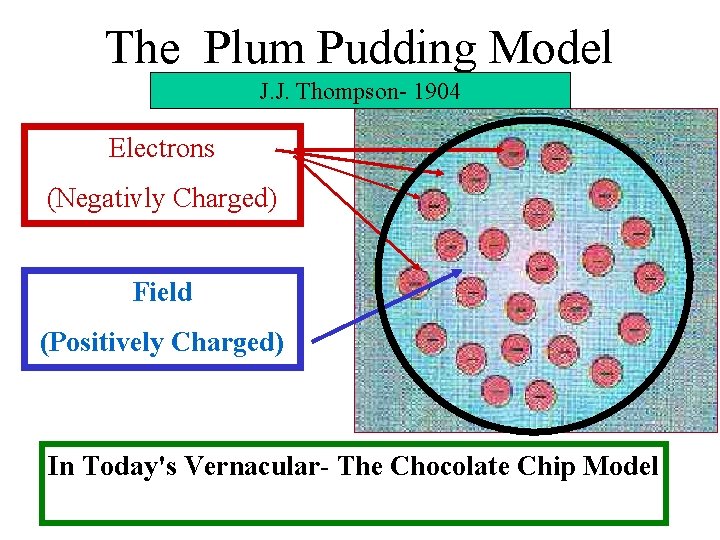
The Plum Pudding Model J. J. Thompson- 1904 Electrons (Negativly Charged) Field (Positively Charged) In Today's Vernacular- The Chocolate Chip Model

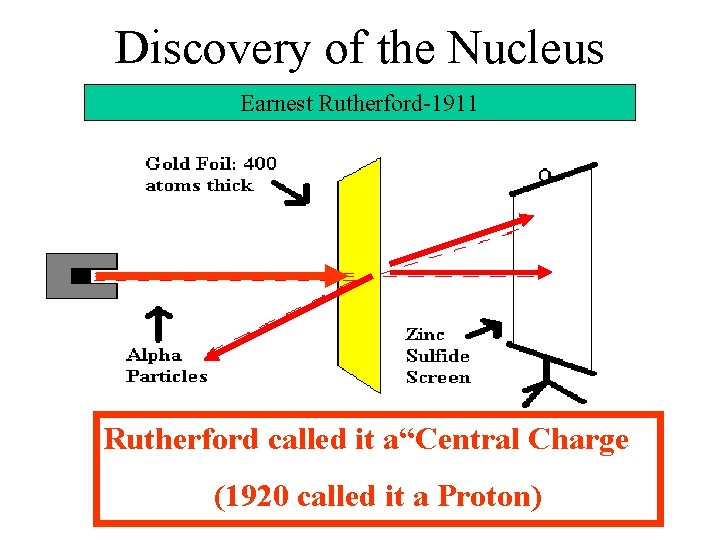
Discovery of the Nucleus Earnest Rutherford-1911 Rutherford called it a“Central Charge (1920 called it a Proton)

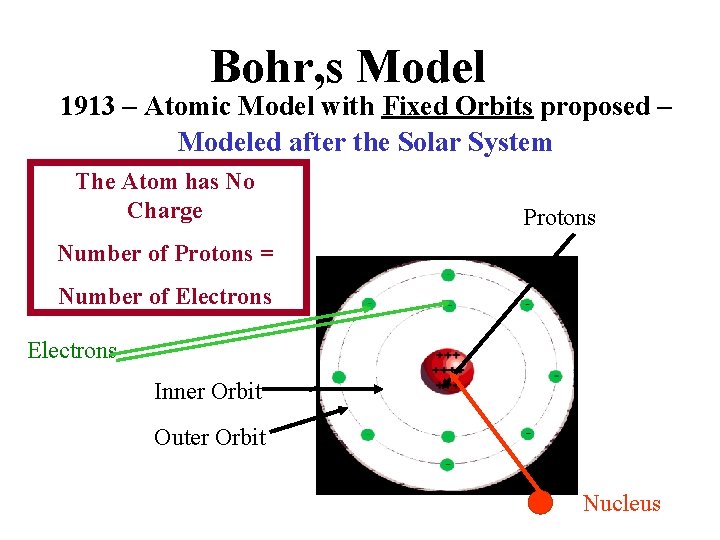
Bohr, s Model 1913 – Atomic Model with Fixed Orbits proposed – Modeled after the Solar System The Atom has No Charge Protons Number of Protons = Number of Electrons Inner Orbit Nucleus Outer Orbit Nucleus

What Did Bohr’s Model Accomplish ? Stability Electrical Forces balanced by Centrifugal Forces Identity Change in number of Protons/Electrons Changes the Element Regeneration Once Pulled Apart, the Atom Reforms as before

How Big is It, (Are They)? 1 Proton ( or Neutron)----1. 67 x 10 -24 Grams =0. 0000000000001. 67 Grams 1 electron-----9. 1 x 10 -28 Grams =0. 000000000000009. 1 Grams The Proton weighs 1800 x the Electron The Atom is about 5 X 10 -8 Cm

Lets Try Analogies If the Nucleus were the size of a Garbage Can The Atom would be the size of LA If an electrons weighed as much as a watermelon The proton would weigh as much as an elephant Conclusion; It’s mostly nothing

Strange Things were Happening Sometimes the nucleus of an element weighed differently than another nucleus of the same element Rutherford – 1920 Postulated a Neutron Doublet Irene and Fredrick Joliot- Curie 1932 found radiation they thought was protons James Chadwick Didn’t Believe It

Discovery of the Neutron James Chadwick - 1935 A Unique Particle, (not an electron or a proton ) Essentially the same mass as an Proton Has no Charge With the Proton, Makes up the Nucleus

Bohr Model of Hydrogen and Helium

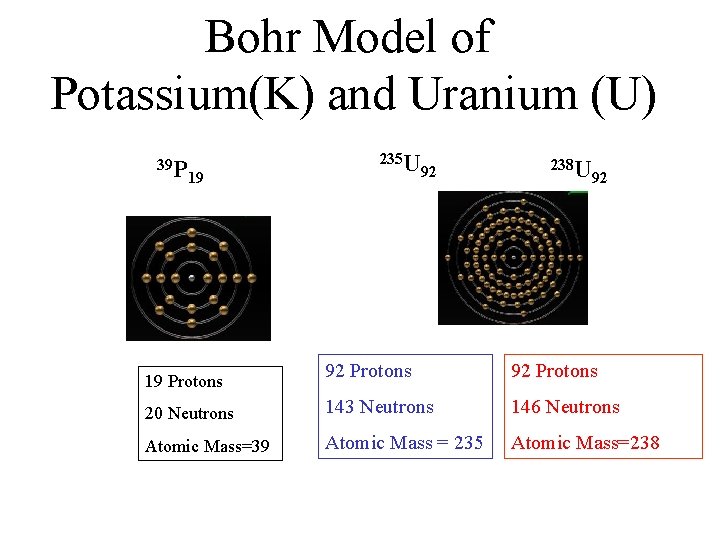
Bohr Model of Potassium(K) and Uranium (U) 39 P 19 235 U 92 238 U 92 92 Protons 20 Neutrons 143 Neutrons 146 Neutrons Atomic Mass=39 Atomic Mass = 235 Atomic Mass=238 19 Protons

So Little From So Much • Marie Curie purified 10, 000 grams of Pitchblende to get 0. 1 Grams of Uranium • Natural uranium is • 99. 3% U 238 • 0. 7% U 235 (The Bomb Type)

Problem – Not Enough 235 U How to increase the. Concentration? Combine the Uranium(s) with Fluorine to get a gas (Uranium Hexafluoride) Pass the gas through a porous filter – More 235 U based gas than 238 U gas gets through. Collect it and do it again, & again------ Centrifuge the Gasses. Heavier 238 U based gas separates from lighter 235 U based gas Collect it and do it again, & again 7 again, & again----Thermal Diffusion- Used Heated, Pressurized Uranium Hexafluoride

How Big was the Job? Oak Ridge Pop. 75, 000 $ 17 Billion (approx $ today)

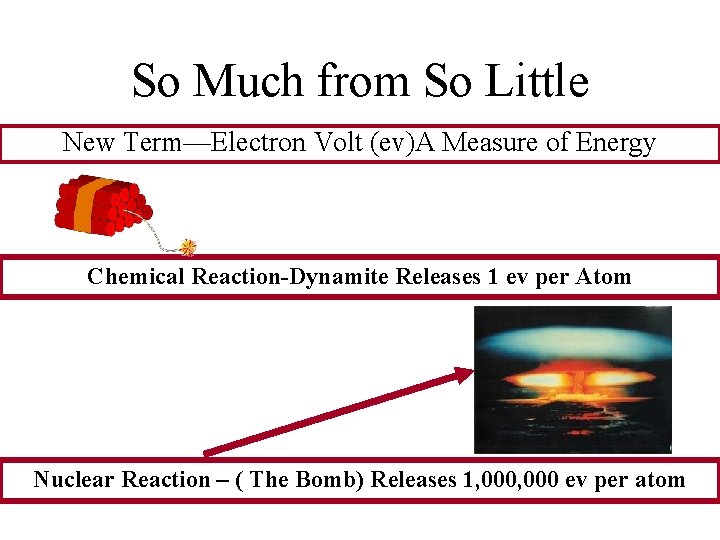
So Much from So Little New Term—Electron Volt (ev)A Measure of Energy Chemical Reaction-Dynamite Releases 1 ev per Atom Nuclear Reaction – ( The Bomb) Releases 1, 000 ev per atom

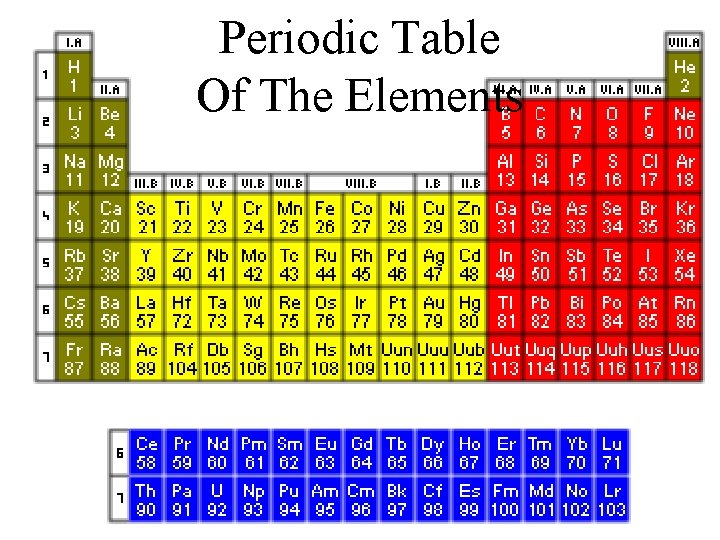
They like Things Orderly How we organize these different Elements? ? ? Mendeleev 1869 Are there Common Characteristics ? ? ? H=1 Li=7 Be=9, 4 B=11 C=12 N=14 O=16 F=19 Na=23 Mg=24 Al=27, 4 Si=28 P=31 S=32 Cl=35, 5 K=39 Ca=40 ? =45 ? Er=56 ? Yt=60 ? In=75, 6 Ti=50 V=51 Cr=52 Mn=55 Fe=56 Ni=Co=59 Cu=63, 4 Zn=65, 2 ? =68 ? =70 As=75 Se=79, 4 Br=80 Rb=85, 4 Sr=87, 6 Ce=92 La=94 Di=95 Th=118? Zr=90 Nb=94 Mo=96 Rh=104, 4 Ru=104, 4 Pd=106, 6 Ag=108 Cd=112 Ur=116 Sn=118 Sb=122 Te=128? J=127 Cs=133 Ba=137 Original Table – 63 Elements ? =180 Ta=182 W=186 Pt=197, 4 Ir=198 Os=199 Hg=200 Au=197? Bi=210? Tl=204 Pb=207

Periodic Table Of The Elements

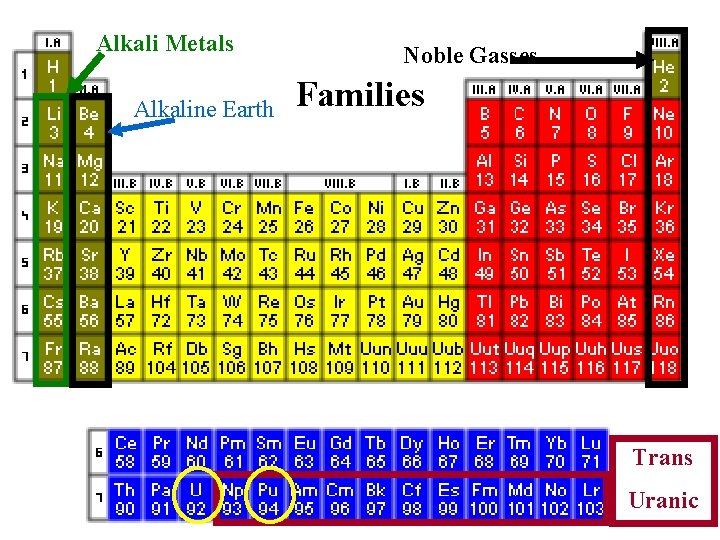
Alkali Metals Alkaline Earth Noble Gasses Families Trans Uranic

The End

Just saving it