Everolimuseluting Bioresorbable Vascular Scaffolds in Patients with Coronary

Everolimus-eluting Bioresorbable Vascular Scaffolds in Patients with Coronary Artery Disease: The ABSORB III trial Dean J. Kereiakes, MD, Stephen G. Ellis, MD, D. Christopher Metzger, MD, Ronald P. Caputo, MD, David G. Rizik, MD, Paul S. Teirstein, MD, Marc R. Litt, MD, Annapoorna Kini, MD, Ameer Kabour, MD, Steven O. Marx, MD, Jeffrey J. Popma, MD, Robert Mc. Greevy, Ph. D, Zhen Zhang, Ph. D, Charles Simonton, MD and Gregg W. Stone, MD for the ABSORB III Investigators

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Modest Consulting Fees • Significant Consulting Fees • Significant Consulting Fees • Major Stock Shareholder/Equity Company • HCRI • Boston Scientific • Abbott Vascular • Svelte Medical Systems, Inc. • Janssen Research & Development LLC • Sanofi-Aventis U. S. LLC • Ablative Solutions, Inc.
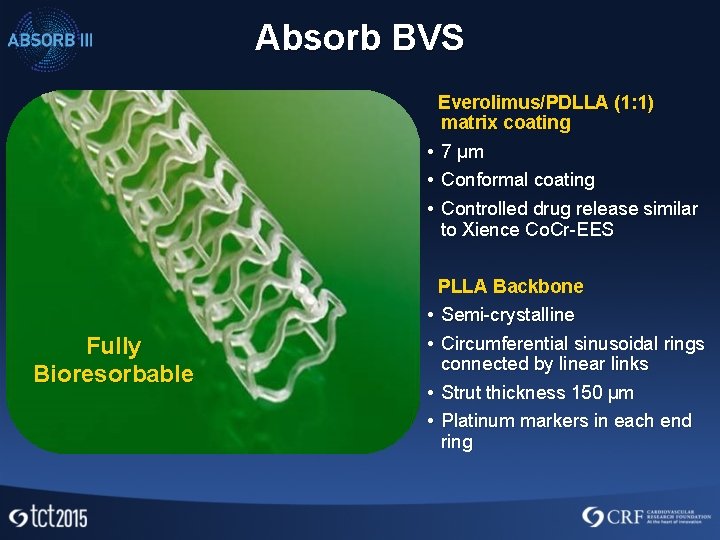
Absorb BVS Everolimus/PDLLA (1: 1) matrix coating • 7 µm • Conformal coating • Controlled drug release similar to Xience Co. Cr-EES PLLA Backbone • Semi-crystalline Fully Bioresorbable • Circumferential sinusoidal rings connected by linear links • Strut thickness 150 µm • Platinum markers in each end ring

Phases of Absorb Functionality Revascularization Restoration Resorption Mechanical Support Mass loss Drug Elution 1 3 6 Months Oberhauser JP et al. Euro. Int 2009; 5: F 15 -22 24
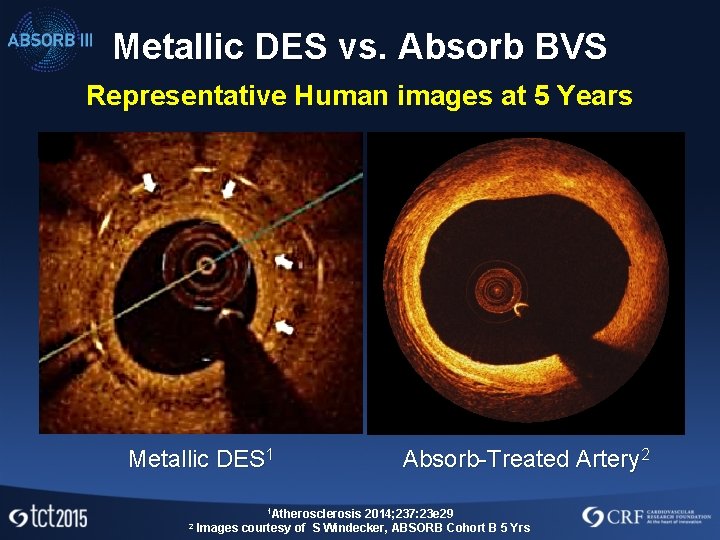
Metallic DES vs. Absorb BVS Representative Human images at 5 Years Metallic DES 1 1 Atherosclerosis 2 Absorb-Treated Artery 2 2014; 237: 23 e 29 Images courtesy of S Windecker, ABSORB Cohort B 5 Yrs

Absorb Program Objectives A Series of Randomized Trials Designed to: • Demonstrate similar (non-inferior) results with ABSORB BVS compared to Xience Co. Cr-EES at 1 year • Demonstrate superior results with ABSORB BVS compared to Xience Co. Cr-EES between 1 and 5 years

ABSORB III Study Design Prospective, multicenter, single-blind, trial ~2, 000 patients randomized 2: 1 Absorb BVS vs. Xience Co. Cr-EES Clinical follow-up: 30 d 6 mo 12 mo 24 mo 36 mo 48 mo 60 mo No routine angiographic follow-up 7

193 Enrolling Centers U. S. Australia

Top Enrollers (N patients) 1. Dr. Metzger (76) 8. Dr. De. Gregorio (38) 15. Dr. Waksman (26) Holston Valley Wellmont Medical Center Englewood Hospital and Medical Center Med. STAR Washington Hospital Center 2. Dr. Caputo (52) St. Joseph's Hospital Health Center 9. Dr. Cannon (36) 16. Dr. Abbas (24) Northern Michigan Hospital William Beaumont Hospital 3. Dr. Rizik (49) 10. Drs. Cambier & Stein (35) Scottsdale Healthcare Morton Plant Hospital, 17. Dr. Zidar (24) Rex Hospital, Inc 4. Dr. Teirstein (45) 11. Dr. Newman (34) 18. Dr. Dearing (24) Scripps Green Hospital Wake. Med Thomas Hospital 5. Dr. Litt (42) 12. Dr. Feldman (31) 19. Dr. Williams (23) Baptist Medical Center Munroe Regional Medical Center Presbyterian Hospital 6. Dr. Kini (41) 13. Dr. Broderick (28) Mount Sinai Medical Center The Christ Hospital Baylor Jack and Jane Hamilton Heart and Vascular Hospital 7. Dr. Kabour (41) 14. Dr. Jain (28) 21. Dr. Moses (23) Mercy St. Vincent Medical Center Washington Hospital, Fremont, CA Columbia University Medical Center 20. Dr. Choi (23)

Study Leadership • Principal Investigators ¡ Dean Kereiakes, MD, The Christ Hospital, Cincinnati, OH ¡ Stephen G. Ellis, MD, Cleveland Clinic, Cleveland, OH • Study Chairman ¡ Gregg W. Stone, MD, Columbia University Medical Center, NY • Clinical Events Committee ¡ Cardiovascular Research Foundation, New York, NY Steven Marx, MD, chair • Angiographic Core Laboratory ¡ Beth Israel Deaconess Medical Center, Boston, MA Jeff Popma, MD, director • Data Safety Monitoring Board § Axio Research, Seattle, WA; Robert N. Piana, MD, chair • Sponsor ¡ Abbott Vascular, Santa Clara, CA

Major Endpoints at 1 Year Primary Endpoint: Target Lesion Failure (non-inferiority) • Cardiac death, or • Myocardial infarction attributed to the target vessel (TV-MI), or • Peri-procedural MI: CK-MB >5 x ULN w/i 48 hours • Ischemia-driven target lesion revascularization (ID-TLR) Powered Secondary Endpoints (superiority) • Angina • All revascularization • Ischemia-driven target vessel revascularization (ID-TVR)

Statistical Design Primary Endpoint Non-inferiority analysis for TLF at 1 year with the following assumptions: ¡ 1 -year TLF rate of 7% ¡ Non-inferiority margin of 4. 5% § “Putative placebo”, preserving ≥ 50% of the treatment effect of Xience vs. BMS ¡ 1 -sided ¡ 95% ¡ 2000 alpha of 0. 025 (equivalent to 2 -sided 0. 05) 1 -year follow-up subjects → 96% power Maximum observed difference in order to pass non-inferiority = 2%

Key Patient Eligibility Criteria • >18 years old • Evidence of myocardial ischemia (stable/unstable/postinfarction angina or silent ischemia) • No elevation of CK-MB • 1 or 2 de novo target lesions in up to 2 native coronary arteries (max 1 lesion per artery) • Diameter stenosis ≥ 50% and <100% with TIMI flow ≥ 1 ¡ If <70%, abnormal functional test (including FFR ≤ 0. 80), unstable angina or post-infarct angina • RVD ≥ 2. 50 mm and ≤ 3. 75 mm (site-determined) • Lesion length ≤ 24 mm (site-determined)
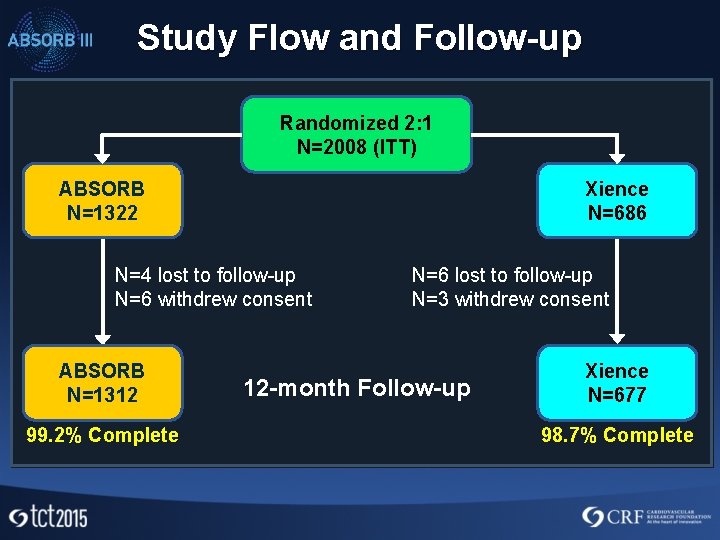
Study Flow and Follow-up Randomized 2: 1 N=2008 (ITT) ABSORB N=1322 Xience N=686 N=4 lost to follow-up N=6 withdrew consent ABSORB N=1312 99. 2% Complete N=6 lost to follow-up N=3 withdrew consent 12 -month Follow-up Xience N=677 98. 7% Complete
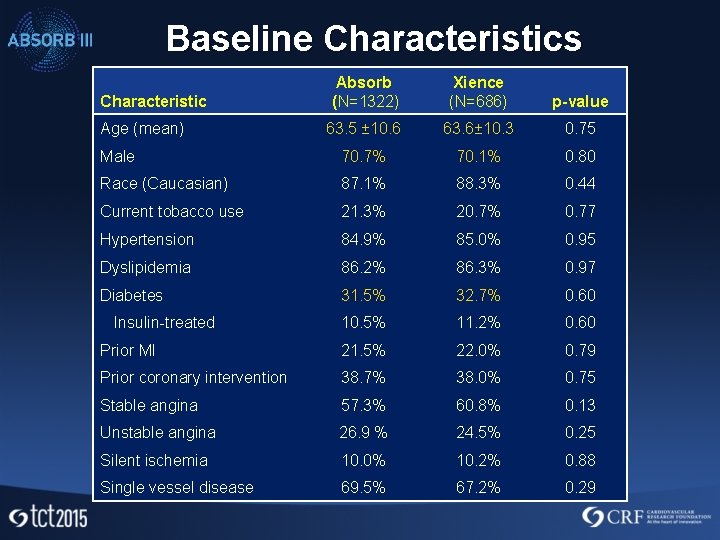
Baseline Characteristics Absorb (N=1322) Xience (N=686) p-value 63. 5 ± 10. 6 63. 6± 10. 3 0. 75 Male 70. 7% 70. 1% 0. 80 Race (Caucasian) 87. 1% 88. 3% 0. 44 Current tobacco use 21. 3% 20. 7% 0. 77 Hypertension 84. 9% 85. 0% 0. 95 Dyslipidemia 86. 2% 86. 3% 0. 97 Diabetes 31. 5% 32. 7% 0. 60 10. 5% 11. 2% 0. 60 Prior MI 21. 5% 22. 0% 0. 79 Prior coronary intervention 38. 7% 38. 0% 0. 75 Stable angina 57. 3% 60. 8% 0. 13 Unstable angina 26. 9 % 24. 5% 0. 25 Silent ischemia 10. 0% 10. 2% 0. 88 Single vessel disease 69. 5% 67. 2% 0. 29 Characteristic Age (mean) Insulin-treated
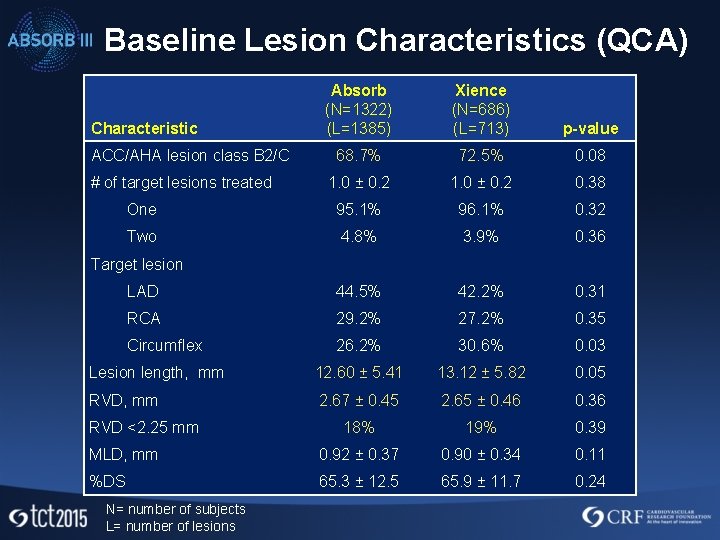
Baseline Lesion Characteristics (QCA) Absorb (N=1322) (L=1385) Xience (N=686) (L=713) p-value 68. 7% 72. 5% 0. 08 1. 0 ± 0. 2 0. 38 One 95. 1% 96. 1% 0. 32 Two 4. 8% 3. 9% 0. 36 LAD 44. 5% 42. 2% 0. 31 RCA 29. 2% 27. 2% 0. 35 Circumflex 26. 2% 30. 6% 0. 03 Lesion length, mm 12. 60 ± 5. 41 13. 12 ± 5. 82 0. 05 RVD, mm 2. 67 ± 0. 45 2. 65 ± 0. 46 0. 36 18% 19% 0. 39 MLD, mm 0. 92 ± 0. 37 0. 90 ± 0. 34 0. 11 %DS 65. 3 ± 12. 5 65. 9 ± 11. 7 0. 24 Characteristic ACC/AHA lesion class B 2/C # of target lesions treated Target lesion RVD <2. 25 mm N= number of subjects L= number of lesions
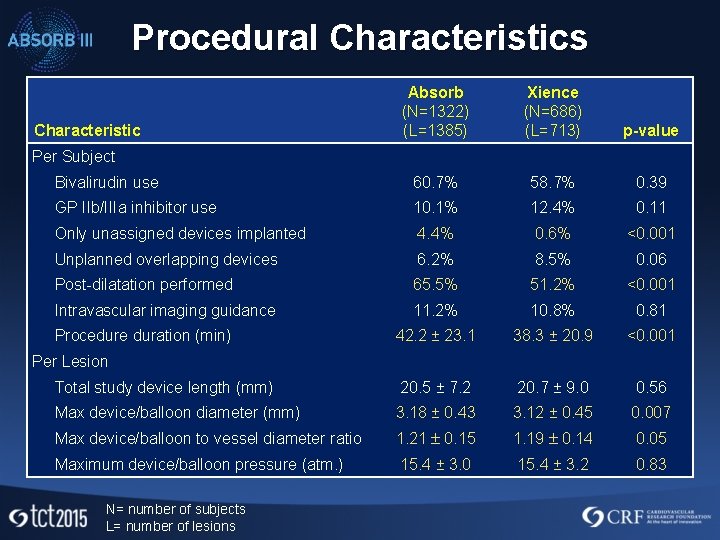
Procedural Characteristics Absorb (N=1322) (L=1385) Xience (N=686) (L=713) p-value Bivalirudin use 60. 7% 58. 7% 0. 39 GP IIb/IIIa inhibitor use 10. 1% 12. 4% 0. 11 Only unassigned devices implanted 4. 4% 0. 6% <0. 001 Unplanned overlapping devices 6. 2% 8. 5% 0. 06 Post-dilatation performed 65. 5% 51. 2% <0. 001 Intravascular imaging guidance 11. 2% 10. 8% 0. 81 42. 2 ± 23. 1 38. 3 ± 20. 9 <0. 001 Total study device length (mm) 20. 5 ± 7. 2 20. 7 ± 9. 0 0. 56 Max device/balloon diameter (mm) 3. 18 ± 0. 43 3. 12 ± 0. 45 0. 007 Max device/balloon to vessel diameter ratio 1. 21 0. 15 1. 19 0. 14 0. 05 Maximum device/balloon pressure (atm. ) 15. 4 ± 3. 0 15. 4 ± 3. 2 0. 83 Characteristic Per Subject Procedure duration (min) Per Lesion N= number of subjects L= number of lesions
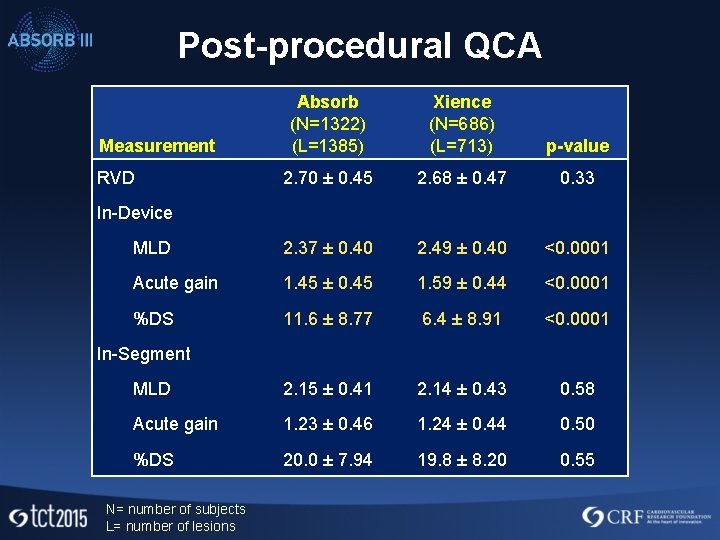
Post-procedural QCA Absorb (N=1322) (L=1385) Xience (N=686) (L=713) p-value 2. 70 ± 0. 45 2. 68 ± 0. 47 0. 33 MLD 2. 37 ± 0. 40 2. 49 ± 0. 40 <0. 0001 Acute gain 1. 45 ± 0. 45 1. 59 ± 0. 44 <0. 0001 %DS 11. 6 ± 8. 77 6. 4 ± 8. 91 <0. 0001 MLD 2. 15 ± 0. 41 2. 14 ± 0. 43 0. 58 Acute gain 1. 23 ± 0. 46 1. 24 ± 0. 44 0. 50 %DS 20. 0 ± 7. 94 19. 8 ± 8. 20 0. 55 Measurement RVD In-Device In-Segment N= number of subjects L= number of lesions
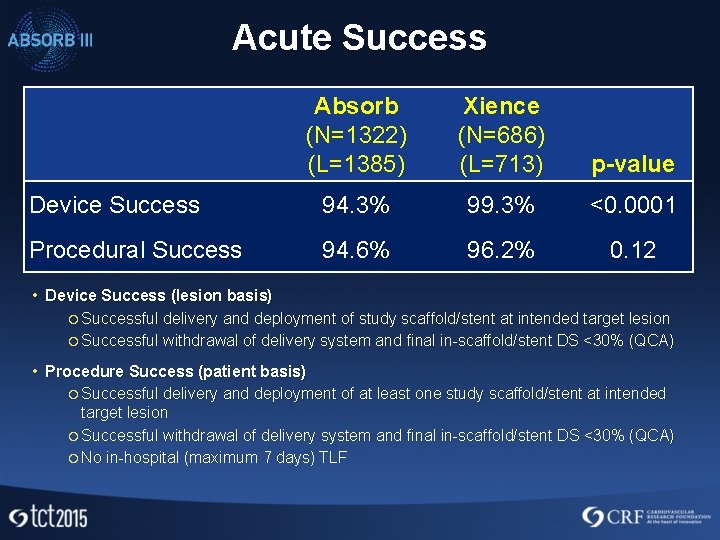
Acute Success Absorb (N=1322) (L=1385) Xience (N=686) (L=713) p-value Device Success 94. 3% 99. 3% <0. 0001 Procedural Success 94. 6% 96. 2% 0. 12 • Device Success (lesion basis) ¡ Successful delivery and deployment of study scaffold/stent at intended target lesion ¡ Successful withdrawal of delivery system and final in-scaffold/stent DS <30% (QCA) • Procedure Success (patient basis) ¡ Successful delivery and deployment of at least one study scaffold/stent at intended target lesion ¡ Successful withdrawal of delivery system and final in-scaffold/stent DS <30% (QCA) ¡ No in-hospital (maximum 7 days) TLF
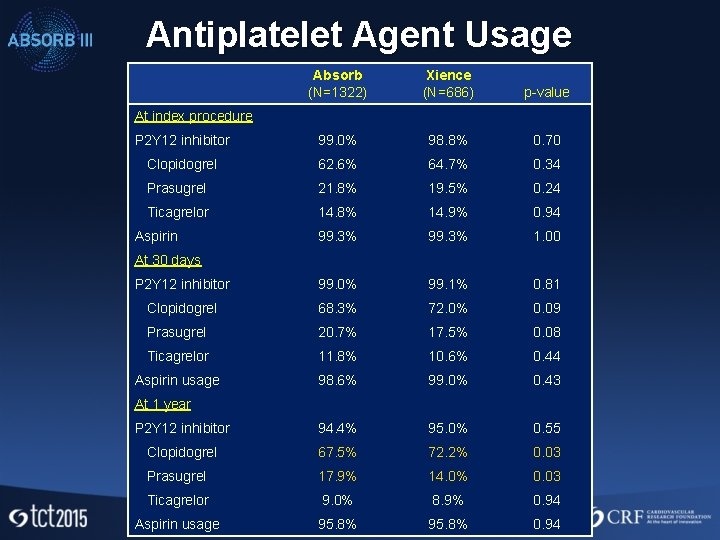
Antiplatelet Agent Usage Absorb (N=1322) Xience (N=686) p-value P 2 Y 12 inhibitor 99. 0% 98. 8% 0. 70 Clopidogrel 62. 6% 64. 7% 0. 34 Prasugrel 21. 8% 19. 5% 0. 24 Ticagrelor 14. 8% 14. 9% 0. 94 99. 3% 1. 00 P 2 Y 12 inhibitor 99. 0% 99. 1% 0. 81 Clopidogrel 68. 3% 72. 0% 0. 09 Prasugrel 20. 7% 17. 5% 0. 08 Ticagrelor 11. 8% 10. 6% 0. 44 Aspirin usage 98. 6% 99. 0% 0. 43 P 2 Y 12 inhibitor 94. 4% 95. 0% 0. 55 Clopidogrel 67. 5% 72. 2% 0. 03 Prasugrel 17. 9% 14. 0% 0. 03 Ticagrelor 9. 0% 8. 9% 0. 94 Aspirin usage 95. 8% 0. 94 At index procedure Aspirin At 30 days At 1 year

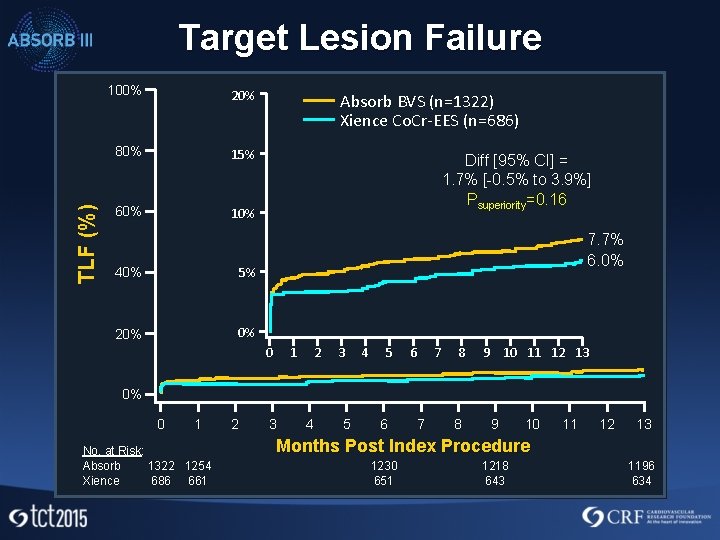
TLF (%) Target Lesion Failure 100% 20% 80% 15% 60% 10% 40% 5% 20% 0% Absorb BVS (n=1322) Xience Co. Cr-EES (n=686) Diff [95% CI] = 1. 7% [-0. 5% to 3. 9%] Psuperiority=0. 16 7. 7% 6. 0% 0 1 2 3 4 5 6 7 8 9 10 11 12 13 0% 0 1 No. at Risk: Absorb 1322 1254 Xience 686 661 2 3 4 5 6 7 8 9 10 11 12 13 Months Post Index Procedure 1230 651 1218 643 1196 634




Peri-Procedural MI by Definition Absorb (N=1322) Xience (N=686) Difference p-value >3 x ULN 6. 8% 6. 6% 0. 2 0. 89 >5 x ULN (protocol) 3. 0% 2. 8% 0. 2 0. 75 >8 x ULN 1. 3% 0. 0 0. 96 >10 x ULN 0. 9% 1. 2% -0. 3 0. 58 SCAI definition* 0. 9% 1. 2% -0. 3 0. 58 CK-MB threshold *>10 x ULN or >5 x ULN with new Q waves or new persistent LBBB J Am Coll Cardiol 2013; 62: 1563 -70

Device Thrombosis to 1 Year Absorb (N=1322) Xience (N=686) p-value 1. 54% 0. 74% 0. 13 - Early (0 to 30 days) 1. 06% 0. 73% 0. 46 - Late (> 30 to 1 year) 0. 46% 0. 00% 0. 10 - Definite* (1 year) 1. 38% 0. 74% 0. 21 - Probable (1 year ) 0. 15% 0. 00% 0. 55 Device Thrombosis (def*/prob) *One “definite ST” in the Absorb arm by ITT was in a pt that was treated with Xience
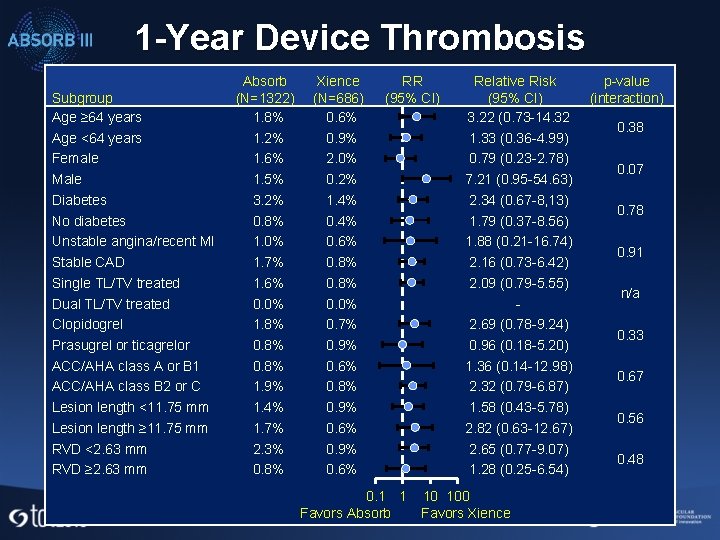
1 -Year Device Thrombosis Subgroup Age ≥ 64 years Age <64 years Female Male Diabetes No diabetes Unstable angina/recent MI Stable CAD Single TL/TV treated Dual TL/TV treated Clopidogrel Prasugrel or ticagrelor ACC/AHA class A or B 1 ACC/AHA class B 2 or C Lesion length <11. 75 mm Lesion length ≥ 11. 75 mm RVD <2. 63 mm RVD ≥ 2. 63 mm Absorb (N=1322) 1. 8% 1. 2% 1. 6% 1. 5% 3. 2% 0. 8% 1. 0% 1. 7% 1. 6% 0. 0% 1. 8% 0. 8% 1. 9% 1. 4% 1. 7% 2. 3% 0. 8% Xience (N=686) 0. 6% 0. 9% 2. 0% 0. 2% 1. 4% 0. 6% 0. 8% 0. 0% 0. 7% 0. 9% 0. 6% 0. 8% 0. 9% 0. 6% RR (95% CI) 0. 1 1 Favors Absorb Relative Risk (95% CI) 3. 22 (0. 73 -14. 32 1. 33 (0. 36 -4. 99) 0. 79 (0. 23 -2. 78) 7. 21 (0. 95 -54. 63) 2. 34 (0. 67 -8, 13) 1. 79 (0. 37 -8. 56) 1. 88 (0. 21 -16. 74) 2. 16 (0. 73 -6. 42) 2. 09 (0. 79 -5. 55) 2. 69 (0. 78 -9. 24) 0. 96 (0. 18 -5. 20) 1. 36 (0. 14 -12. 98) 2. 32 (0. 79 -6. 87) 1. 58 (0. 43 -5. 78) 2. 82 (0. 63 -12. 67) 2. 65 (0. 77 -9. 07) 1. 28 (0. 25 -6. 54) 10 100 Favors Xience p-value (interaction) 0. 38 0. 07 0. 78 0. 91 n/a 0. 33 0. 67 0. 56 0. 48
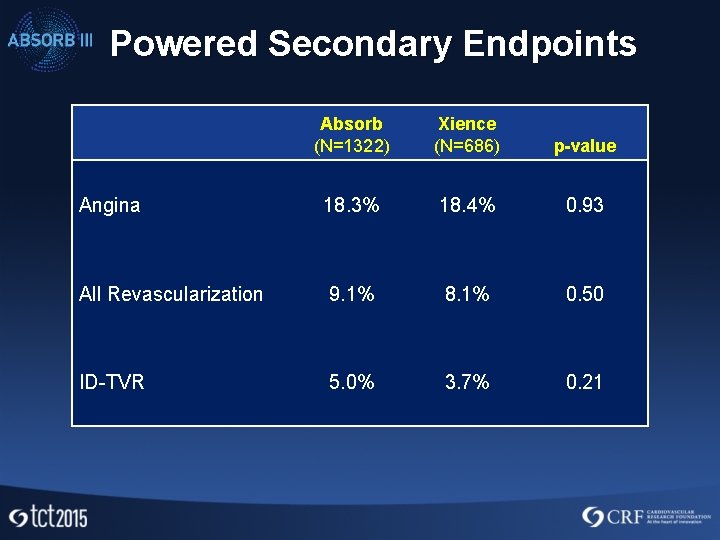
Powered Secondary Endpoints Absorb (N=1322) Xience (N=686) p-value Angina 18. 3% 18. 4% 0. 93 All Revascularization 9. 1% 8. 1% 0. 50 ID-TVR 5. 0% 3. 7% 0. 21

ABSORB III Very Small Vessel Analysis • ABSORB III eligibility criteria included vessels with RVD 2. 5 mm – 3. 75 mm (visual estimation) • The thicker struts of ABSORB may be of particular concern in very small vessels • Subgroup analysis was therefore performed in vessels with QCA RVD <2. 25 mm vs. ≥ 2. 25 mm (correlates with visual estimate ~2. 5 mm) • ~19% of patients had a target lesion with RVD <2. 25 mm by QCA
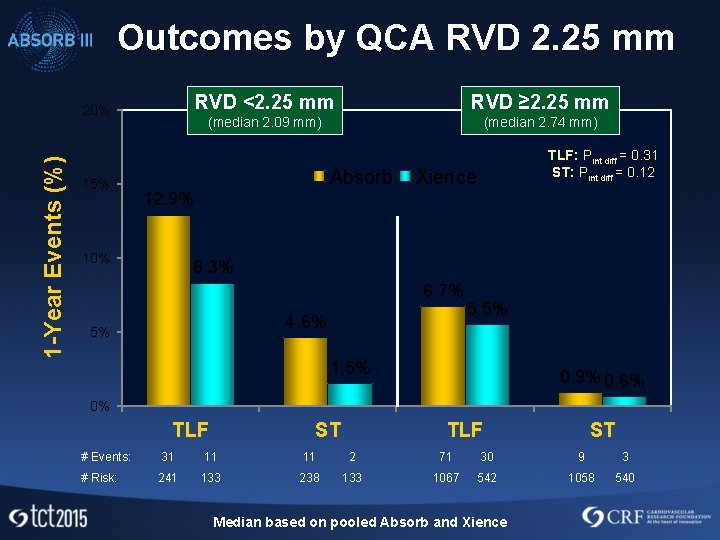
Outcomes by QCA RVD 2. 25 mm 1 -Year Events (%) 20% 15% RVD <2. 25 mm RVD ≥ 2. 25 mm (median 2. 09 mm) (median 2. 74 mm) Absorb TLF: Pint diff = 0. 31 ST: Pint diff = 0. 12 Xience 12. 9% 10% 8. 3% 6. 7% 4. 6% 5% 5. 5% 1. 5% 0. 9% 0. 6% 0% TLF ST # Events: 31 11 11 2 71 30 9 3 # Risk: 241 133 238 133 1067 542 1058 540 Median based on pooled Absorb and Xience

Limitations • ABSORB III enrolled patients with stable ischemic heart disease and stabilized ACS, and excluded specific complex lesions (e. g. CTO, LM, large bif); results may therefore not be generalizable to higherrisk patients and more complex disease • Underpowered for low frequency events • Results should be viewed in context that Xience was the control device which has been associated with the lowest rates of ST and other events • BVS is a first generation device and was used for the first time by most operators within this trial

Summary and Conclusions (1) • ABSORB BVS was non-inferior to Xience Co. Cr. EES for TLF at 1 year (primary endpoint met) • TLF components (cardiac death, TV-MI, ID-TLR) were not significantly different between devices • Angina, all revascularization and ID-TVR were similar between devices • No statistically significant differences in device thrombosis were present

Summary and Conclusions (2) • The ABSORB III trial has demonstrated safety and efficacy of Absorb BVS at 1 year in patients with stable CAD and stabilized ACS • Longer term evaluation is ongoing to determine if ABSORB improves late outcomes compared to Xience

- Slides: 35