EUnet HTA HANDBOOK ON HTA CAPACITY BUILDING Montse

EUnet. HTA HANDBOOK ON HTA CAPACITY BUILDING Montse Moharra; Antoni Parada on behalf of WP 8 of the EUnet. HTA project Catalan Agency for Health Technology Assessment and Research Glasgow, 9 -10 th February 2009 CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH

The EUnet. HTA HANDBOOK ON HTA CAPACITY BUILDING is a product of the EUnet. HTA Workpackage 8 on systems to support HTA in member states with limited institutionalisation of HTA EUnet. HTA WP 8. OBJECTIVES were: 1. To define the recommendable minimum components related to the scope, structure and the work process of an HTA organization. 2. To develop tools for information and education support addressed to institutions or healthcare systems in the process of evolving to an HTA organization. CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH

WP 8 DELIVERABLES & MILESTONES Review on HTA organisations Survey on Information management units December 2006 October 2007 October 2008 Handbook on HTA capacity building HTA Curricula November 2008

HANDBOOK ON HTA CAPACITY BUILDING EUnet. HTA HANDBOOK ON HTA CAPACITY BUILDING WORK PACKAGE 8 OCTOBER 2008 • The WP 8’s main product is the Handbook on HTA capacity building. • Its objective is to provide guidance and support in the process of establishing HTA in countries with limited HTA capacity, and also to serve as a practical tool in other countries where HTA is more consolidated. • The Handbook, which is coordinated by CAHTA, consists of nine chapters. • The handbook is available at www. eunethta. net CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH

CONTENTS OF THE HANDBOOK ON HTA CAPACITY BUILDING I Chapter 1. Introduction Before establishing an HTA programme different aspects should be considered: 1. Preparing the ground Chapter 2. Building of national HTA capacity 2. Identifying suitable professionals and HTA training opportunities 3. Integrating various professional disciplines Chapter 3. Aims and scope 4. Analysing the current scene relevant to HTA 5. Networking and Communication CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH
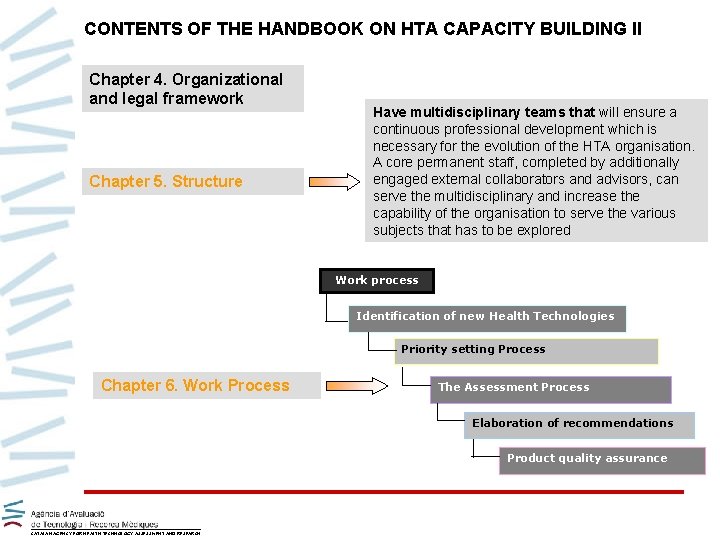
CONTENTS OF THE HANDBOOK ON HTA CAPACITY BUILDING II Chapter 4. Organizational and legal framework Chapter 5. Structure Have multidisciplinary teams that will ensure a continuous professional development which is necessary for the evolution of the HTA organisation. A core permanent staff, completed by additionally engaged external collaborators and advisors, can serve the multidisciplinary and increase the capability of the organisation to serve the various subjects that has to be explored Work process Identification of new Health Technologies Priority setting Process Chapter 6. Work Process The Assessment Process Elaboration of recommendations Product quality assurance CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH
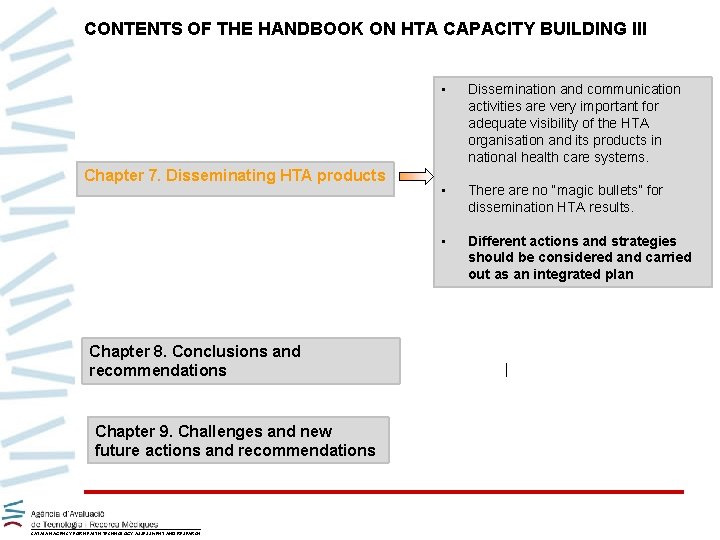
CONTENTS OF THE HANDBOOK ON HTA CAPACITY BUILDING III Chapter 7. Disseminating HTA products Chapter 8. Conclusions and recommendations Chapter 9. Challenges and new future actions and recommendations CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH • Dissemination and communication activities are very important for adequate visibility of the HTA organisation and its products in national health care systems. • There are no “magic bullets” for dissemination HTA results. • Different actions and strategies should be considered and carried out as an integrated plan

WP 8 MAIN FINAL REMARKS Before establishing an HTA programme different aspects should be considered: Preparing the ground Identifying suitable professionals and HTA training opportunities Integrating various professional disciplines Analysing the current scene relevant to HTA Networking and communication Once the new agency has been established, different aspects should be taken into account: Be sensible to the specific setting needs Establish liaisons Be benefited from the ‘core’ information. Look out for high quality products Ensure financial sources Active action on decision makers Have multidisciplinary teams Co-operate at national level Look for international collaboration Achieve legal support CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH

WP 8 MAIN FINAL REMARKS It is recommended that each agency should clearly define its scope of activities and on that basis either identify and/or select technologies for assessment or await other agencies’ assessment of specific technologies, with or without subsequent adaptation to a local context. There are no “magic bullets” for disseminating HTA results. Different actions and strategies should be considered and carried out as an integrated plan which should include: Dissemination HTA results The identification of HTA target audiences Elaboration of the messages adapted to target audiences Formal and complex dissemination strategies CATALAN AGENCY FOR HEALTH TECHNOLOGY ASSESSMENT AND RESEARCH
- Slides: 9