Ethylene Carbonate Synthesis by Coupling Reaction of Ethylene

Ethylene Carbonate Synthesis by Coupling Reaction of Ethylene Oxide & Carbon Dioxide in the presence of Ionic Liquids Based Catalysts Activity & Stability of [Emim]2 Zn. Br 2 Cl 2 & [Bu 4 N]2 Zn. Br 2 Cl 2 Catalysts due to Weight Ratio of Substrate/Catalyst & Temperature Variations Presented by: Jelliarko P. Green & Sustainable Chemistry Lab

Coupling Reaction in ethylene carbonate production What is coupling reaction of ethylene oxide and carbon dioxide ? O O + O Cat T O O P Advantage - One step reaction. - No solvent is required except the product itself. - High molar substrate/catalyst ratio. - Simple distillation in product separation A Progress Report A progres Introduction A Progress Report
![2 [Emim]2 Zn. Br 2 Cl 2 H 3 C + N N CH 2 [Emim]2 Zn. Br 2 Cl 2 H 3 C + N N CH](http://slidetodoc.com/presentation_image_h2/8ed248dba0ea89d5acfe1d4be182c680/image-3.jpg)
2 [Emim]2 Zn. Br 2 Cl 2 H 3 C + N N CH 3 H 3 C Transition metal-Ionic Liquids Catalysts Zn. Br 2 Cl 2 2 CH 3 N [Bu 4 N]2 Zn. Br 2 Cl 2 + Zn. Br 2 Cl 2 H 3 C CH 3 A Progress Report A progres Introduction A Progress Report

Objective 1. Investigating decomposition of ethylene carbonate alone under vacuum and high temperature. Objective 2. Investigating decomposition of ethylene carbonate in the presence of catalyst under vacuum and high temperature. Objective 3. Investigating activity and stability of catalysts in multiple coupling reactions. A Progress Report A progres ective Obje A Progress Report

1. Investigating Decomposition of Ethylene Carbonate Alone The reactor was charged with an appropriate amount of EC and was fully vacuumed. Then, temperature was increased step by step to specific value (100 – 170 o. C) After cooling, the mixture was weighted and was taken for GC-MS analysis 2. Investigating Decomposition of EC in the Presence of Catalyst The reactor was charge with an appropriate amount of EC and catalyst and was fully vacuumed. Then, temperature was increased step by step to specific value (100 – 170 o. C) After cooling, the mixture was weighted and was taken for GC-MS analysis A Progress Report A progres Experiments A Progress Report
![[Bu 4 N]2 Zn. Br 2 Cl 2 catalyst 0. 5 gr of catalyst [Bu 4 N]2 Zn. Br 2 Cl 2 catalyst 0. 5 gr of catalyst](http://slidetodoc.com/presentation_image_h2/8ed248dba0ea89d5acfe1d4be182c680/image-6.jpg)
[Bu 4 N]2 Zn. Br 2 Cl 2 catalyst 0. 5 gr of catalyst (1. 50 %wt) was loaded into 200 m. L stainless-steel bomb reactor. 50 gr of EC acted as solvent was also put in. Then, the reactor was charged with 33. 6 gr of ethylene oxide. The bomb then was pressurized with CO 2 and heated to specific reaction temperature (100 o. C), with the addition of CO 2 from a reservoir tank to maintain a constant pressure. After cooling the reactor, the mixture was weighted and was transferred into distillation apparatus for separation process. The above procedure was repeated 6 times. A Progress Report A progres Experiments 3. Investigating activity and stability of catalysts in multiple coupling reactions A Progress Report

200 m. L stainless-steel bomb reactor was charged with an appropriate amount of catalyst and EC acted as solvent. Then, the reactor was charged with ethylene oxide. The bomb was pressurized with CO 2 and heated to specific reaction temperature with the addition of CO 2 from a reservoir tank to maintain a constant pressure. After cooling the reactor, the mixture was purged with nitrogen to remove dissolved CO 2 and EO. The reactor and its content were weighted and the second reaction was performed. Experiments A Progress Report A progres Experiments 4. Investigating activity and stability of catalysts in multiple coupling reactions A Progress Report
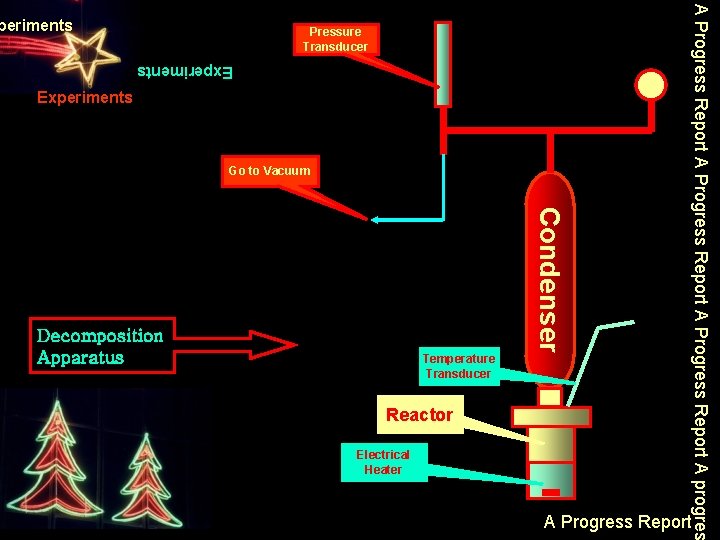
Pressure Transducer Experiments Go to Vacuum Temperature Transducer Reactor Electrical Heater Condenser Decomposition Apparatus A Progress Report A progres periments A Progress Report
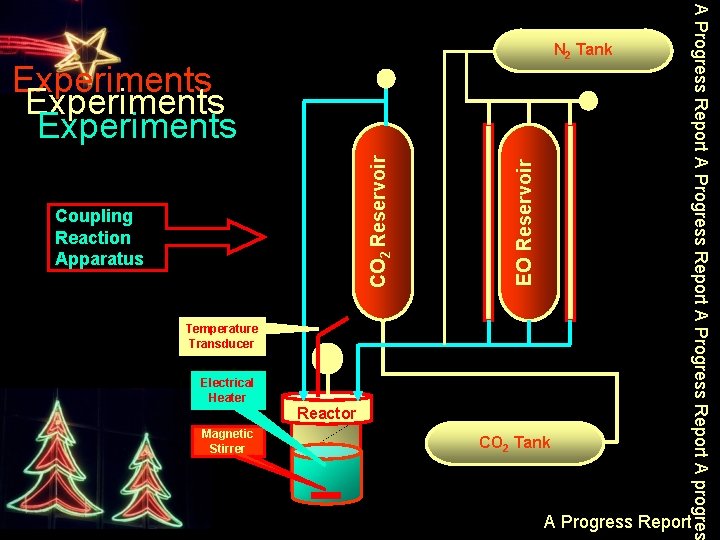
Coupling Reaction Apparatus EO Reservoir CO 2 Reservoir Experiments Temperature Transducer Electrical Heater Reactor Magnetic Stirrer CO 2 Tank A Progress Report A progres N 2 Tank A Progress Report
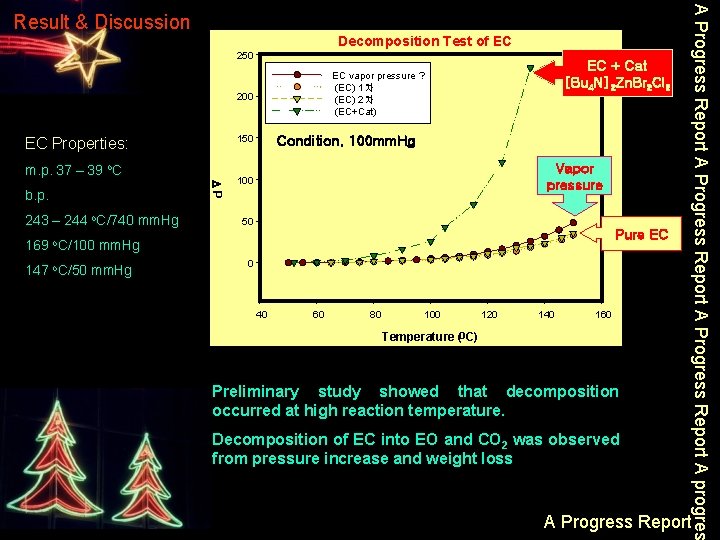
Decomposition Test of EC 250 200 150 EC Properties: m. p. 37 – 39 o. C 243 – 244 o. C/740 mm. Hg DP b. p. EC + Cat [Bu 4 N]2 Zn. Br 2 Cl 2 EC vapor pressure ? (EC) 1차 (EC) 2차 (EC+Cat) Condition. 100 mm. Hg Vapor pressure 100 50 Pure EC 169 o. C/100 mm. Hg 147 o. C/50 mm. Hg 0 40 60 80 100 120 140 160 Temperature (o. C) Preliminary study showed that decomposition occurred at high reaction temperature. Decomposition of EC into EO and CO 2 was observed from pressure increase and weight loss A Progress Report A progres Result & Discussion A Progress Report
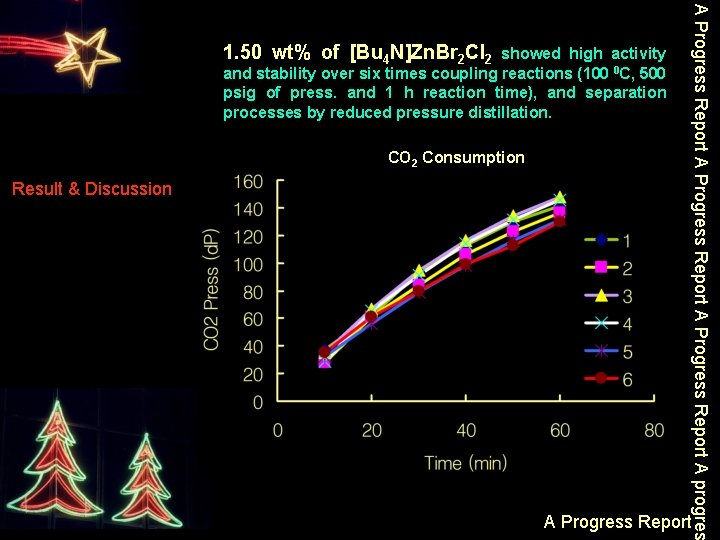
showed high activity and stability over six times coupling reactions (100 0 C, 500 psig of press. and 1 h reaction time), and separation processes by reduced pressure distillation. CO 2 Consumption Result & Discussion A Progress Report A progres 1. 50 wt% of [Bu 4 N]Zn. Br 2 Cl 2 A Progress Report
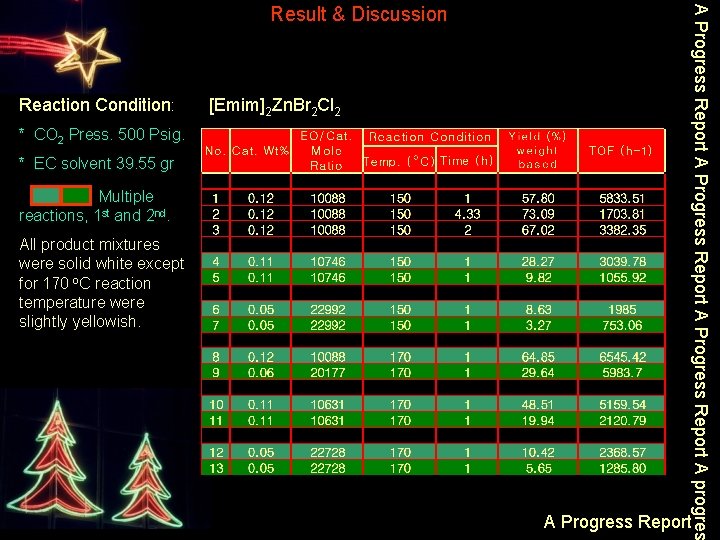
Reaction Condition: * CO 2 Press. 500 Psig. * EC solvent 39. 55 gr Multiple reactions, 1 st and 2 nd. All product mixtures were solid white except for 170 o. C reaction temperature were slightly yellowish. [Emim]2 Zn. Br 2 Cl 2 A Progress Report A progres Result & Discussion A Progress Report
![[Bu 4 N]2 Zn. Br 2 Cl 2 * CO 2 Press. 500 Psig. [Bu 4 N]2 Zn. Br 2 Cl 2 * CO 2 Press. 500 Psig.](http://slidetodoc.com/presentation_image_h2/8ed248dba0ea89d5acfe1d4be182c680/image-13.jpg)
[Bu 4 N]2 Zn. Br 2 Cl 2 * CO 2 Press. 500 Psig. * EC solvent 39. 55 gr Multiple reactions, 1 st and 2 nd. A portion was taken from decomposition test at 150 o. C. All product mixtures were solid white. Result & Discussion A Progress Report A progres Reaction Condition: A Progress Report
![[Emim]2 Zn. Br 2 Cl 2 Reaction Temp. Activity Wt% Ratio Activity A Progress [Emim]2 Zn. Br 2 Cl 2 Reaction Temp. Activity Wt% Ratio Activity A Progress](http://slidetodoc.com/presentation_image_h2/8ed248dba0ea89d5acfe1d4be182c680/image-14.jpg)
[Emim]2 Zn. Br 2 Cl 2 Reaction Temp. Activity Wt% Ratio Activity A Progress Report A progres Result & Discussion A Progress Report
![Reaction Temp. Activity A Progress Report A progres [Bu 4 N]2 Zn. Br 2 Reaction Temp. Activity A Progress Report A progres [Bu 4 N]2 Zn. Br 2](http://slidetodoc.com/presentation_image_h2/8ed248dba0ea89d5acfe1d4be182c680/image-15.jpg)
Reaction Temp. Activity A Progress Report A progres [Bu 4 N]2 Zn. Br 2 Cl 2 Wt% Ratio Result & Discussion A Progress Report

Conclusion • Moderate EC decomposition occurred at high temperature and severe decomposition occurred in the presence of catalysts. • 1. 50 wt% of catalyst ratio showed high durability over multiple coupling reaction and separation processes. ([Bu 4 N]2 Zn. Br 2 Cl 2 – study case). • Above 140 o. C, [Emim]2 Zn. Br 2 Cl 2 showed reduced activity as wt% decreased. However, the activity increased as reaction temperature was raised. • Above 120 o. C, [Bu 4 N]2 Zn. Br 2 Cl 2 showed reduced activity as wt% decreased. However, the activity increased as reaction temperature was raised. Plans • Finding tune the weight ratio of catalysts over EO that will give the most economical value. • Recycling coupling reaction test. A Progress Report A progres “c. HRistm. As” Co. Nc. Lu. Sion & Plans A Progress Report

Saint “재준” Claus says……… Ho…. . ho…. Dear all…. . Happy Christmas & Merry New Year !!! Be cool & Enjoy your works But I will have long vacation ho…ho. .
- Slides: 17