Ethics review by SMEC GDPR check for research

Ethics review by SMEC & GDPR -check for research Laurens Vangeel

Outline 1. Ethical review boards KU Leuven & mission 2. PRET application platform • Privacy/GDPR check • Ethics check • • Social and scientific value Informed consent COVID-19 Other points of interest • Outcome 3. Cases

Ethical review boards KU Leuven • https: //www. kuleuven. be/english/research/ethics/committees • Ethics Committee Research UZ/KU Leuven (EC Research) * • Ethical Committee for Animal Experimentation (ECD) * • Ethics Committee on Dual Use, Military Use & Misuse of Research (EC DMM) • Social and Societal Ethics Committee (SMEC) * These commissions have legal authority: research that involves animals or medical/health related research are legally required to be reviewed by these commissions their advice is legally binding

Mission SMEC evaluates research from different disciplines on human subjects that does not fall under the scope of the Belgian Law regarding research on human subjects dd May 7, 2004 If the primary goal of the study concerns gaining knowledge that will benefit the practice of a health care profession, EC Research is the designated EC. For example: • Research with medical tools that are subject of research (e. g. , robots, scanners) • Clinical trials and invasive experiments on patients and human volunteers (e. g. , surgery, testing of treatments)

Mission We also refer to EC Research if the location of the research is a campus site of UZ Leuven or involves UZ Leuven personnel or patients. Decision tree EC Research – SMEC : https: //www. kuleuven. be/english/research/ethics/committees/smec/ Beslissingsboom In case of any doubt, the promotor can contact SMEC at smec@kuleuven. be.

Mission SMEC includes a multidisciplinary panel of experts for ethical review of research in the humanities and the behavioral or social science research traditions. Also protocols in engineering, natural or life science may be submitted to the SMEC panel. Top 8 Faculty/Department 2018 % Psychology and Educational Sciences 42 Social Sciences 18 Law 4 Arts 4 Economic and Business Sciences 4 Movement Sciences 3 Computer Sciences 3 KU Leuven Association 3

Outline 1. Ethical review boards KU Leuven & mission 2. PRET application platform • Privacy/GDPR check • Ethics check • • Social and scientific value Informed consent COVID-19 Other points of interest • Outcome 3. Cases
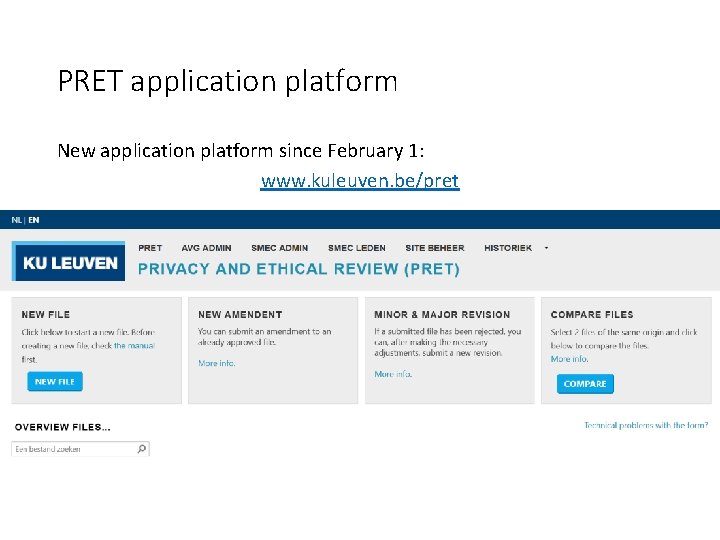
PRET application platform New application platform since February 1: www. kuleuven. be/pret
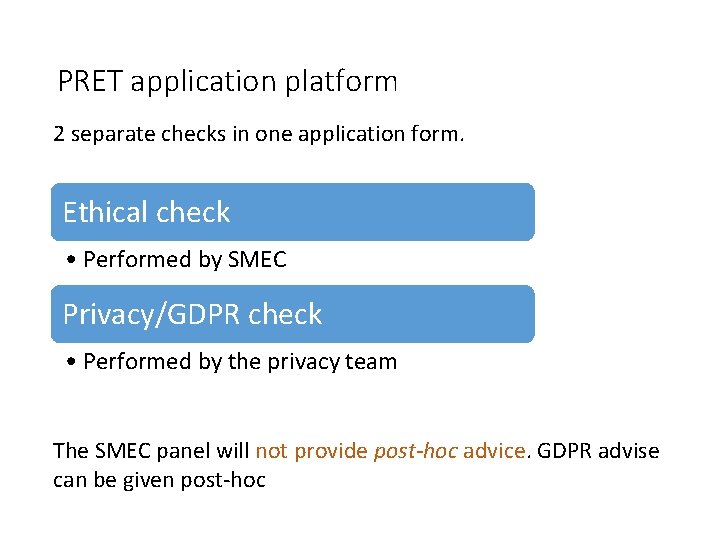
PRET application platform 2 separate checks in one application form. Ethical check • Performed by SMEC Privacy/GDPR check • Performed by the privacy team The SMEC panel will not provide post-hoc advice. GDPR advise can be given post-hoc

PRET application platform If you work with personal data the privacy check is a legal requirement under GDPR
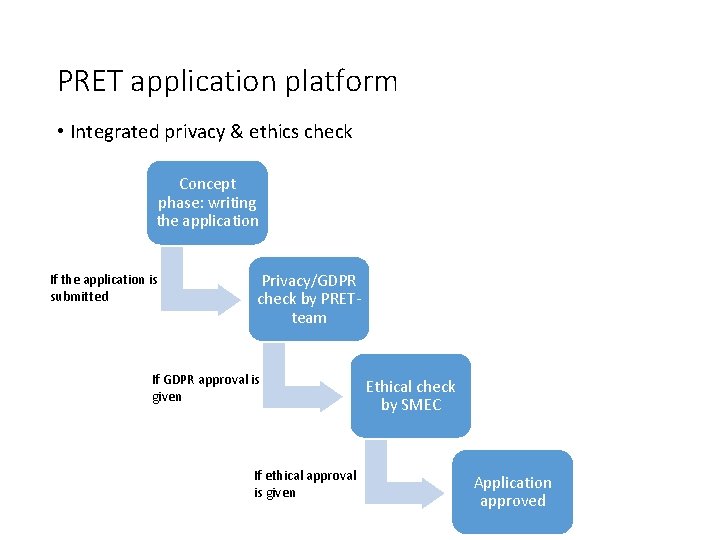
PRET application platform • Integrated privacy & ethics check Concept phase: writing the application If the application is submitted Privacy/GDPR check by PRETteam If GDPR approval is given If ethical approval is given Ethical check by SMEC Application approved

Outline 1. Ethical review boards KU Leuven & mission 2. PRET application platform • Privacy/GDPR check • Ethics check • • Social and scientific value Informed consent COVID-19 Other points of interest • Outcome 3. Cases

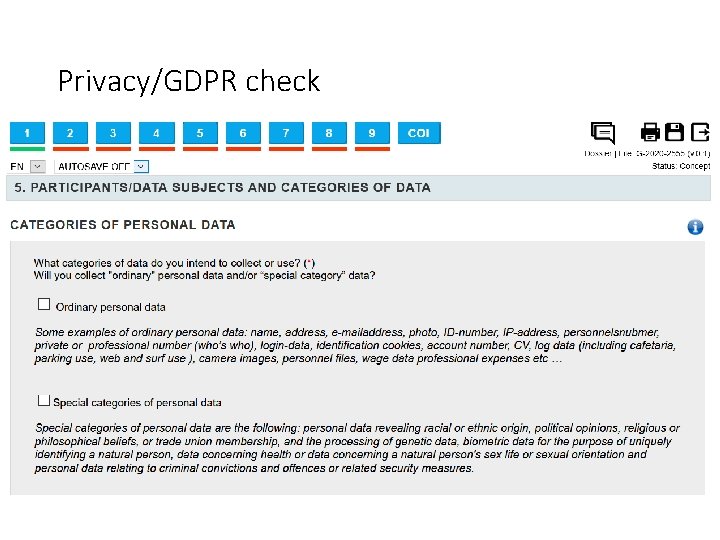
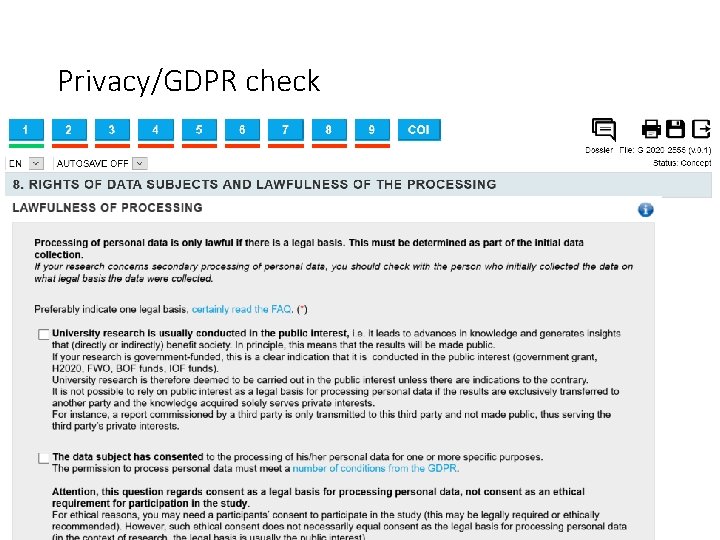
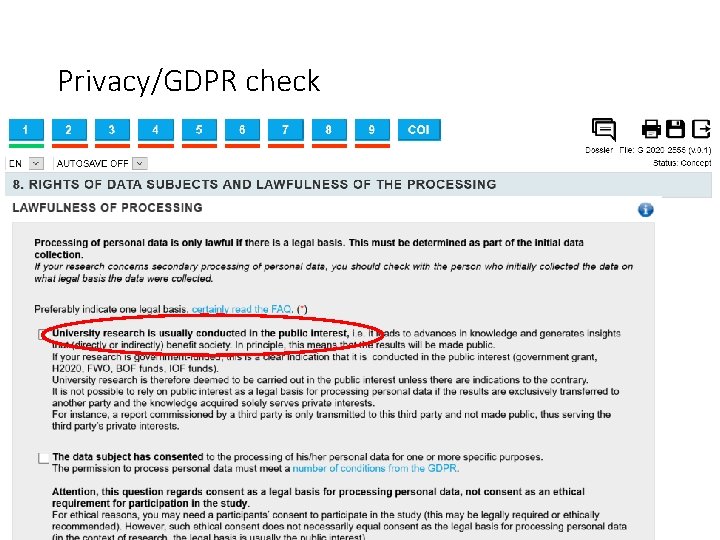
Privacy/GDPR check Personal data is any information relating to an identified or identifiable natural person. An identifiable natural person is one who can be directly or indirectly identified. Examples of ordinary personal data include a person’s name, address, e-mail address, photo, national register number, ID number, IP address, staff number, personal or work phone number (Who’s who), health data, login data, identification cookies, account number, CV, log data (e. g. cafeteria, car park, web browsing), camera images, staff files, payroll data, professional expenses, etc. )
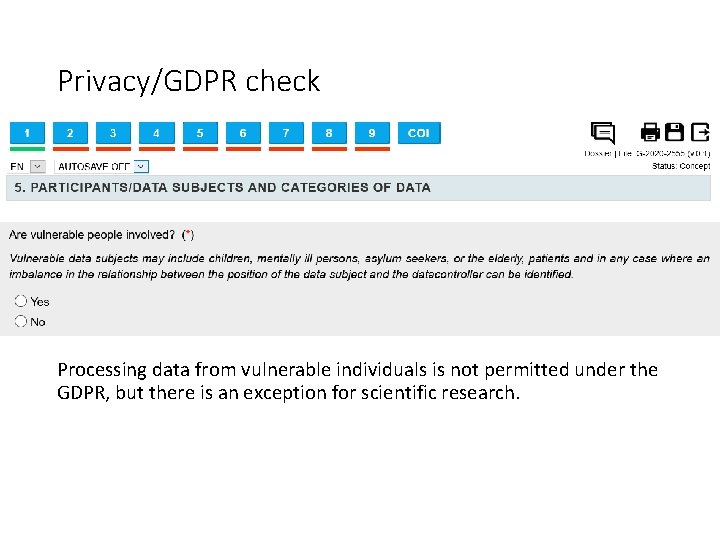
Privacy/GDPR check Processing data from vulnerable individuals is not permitted under the GDPR, but there is an exception for scientific research.
Privacy/GDPR check
Privacy/GDPR check
Privacy/GDPR check

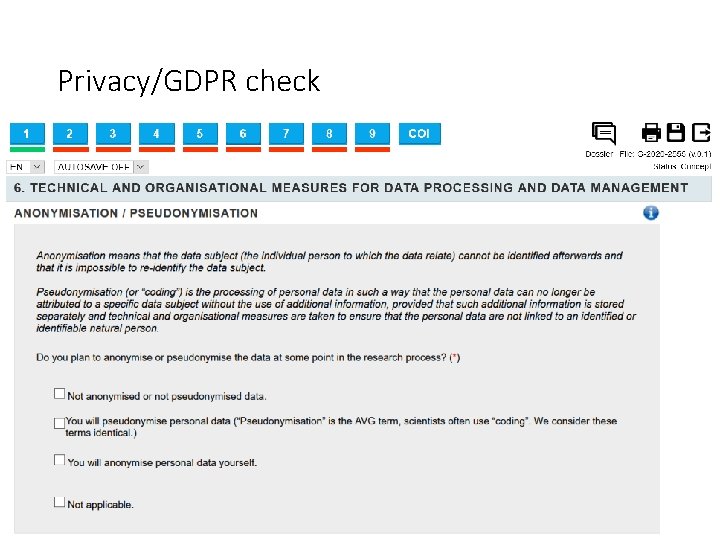
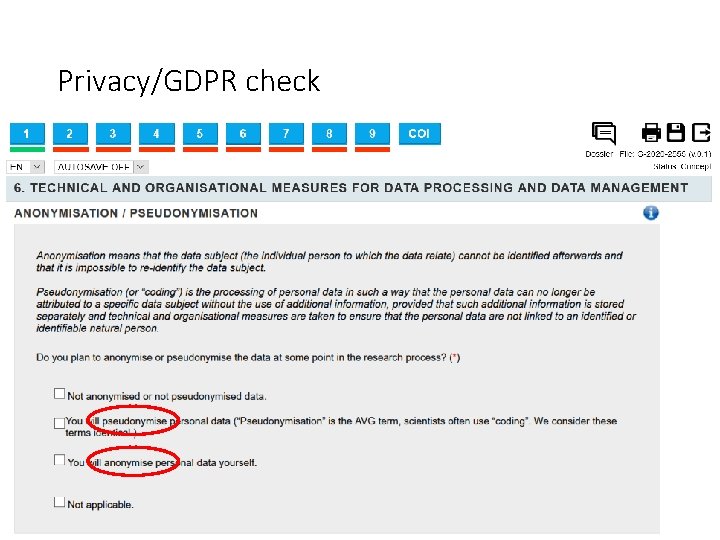
Privacy/GDPR check • Pseudonymization: The data cannot be identified immediately, but a link between data and identities still exists. • Anonymization: Nobody can link the data back to specific individuals
Privacy/GDPR check
Privacy/GDPR check

Outline 1. Ethical review boards KU Leuven & mission 2. PRET application platform • Privacy/GDPR check • Ethics check • • Social and scientific value Informed consent COVID-19 Other points of interest • Outcome 3. Cases

Ethics check • The application identifies and weighs the potential costs and benefits to all stakeholders, with an emphasis on the consequences for the participants and their communities. • In case the research is conducted in a country other than Belgium, the PI is responsible for ensuring that the research is conducted with due regard for local laws, habits and customs. However, the research always adheres to the ethics standards of Belgium/the EU.

Social and scientific value • The research will plausibly lead to relevant insights in the research domain. • The same insights cannot be obtained by using alternative means of research that are less intrusive. • The plausible new insights from the research are in proportion to conceivable burden and risks imposed on participants. • (The research makes use of sound methodology)

Informed consent (1/4) • Consent should be obtained before the participant enters the research (prospectively). Language: aim is that the participant understands what the research is and what they are consenting to in participant’s native language • Inform participants extensively about the research procedures before signing the informed consent forms. Make sure to include information about what participants are actually expected to do during the course of the study. Participants should be warned about anything that might be aversive or distressing (e. g. , explicit materials, surprising or startling events). • Participants cannot be coerced in any way voluntariness of participation will be checked extensively

Informed consent (2/4) • Other topics to be addressed: • Any recordings of voices and images should be mentioned • The right to decline or withdraw without negative consequences • Incentives/reimbursement • Contact information of the experimenter and SMEC should be on the forms – also indicate that procedures have been reviewed by the SMEC panel, and that the commission can be contacted in case of uncertainties about ethical issues.

Informed consent (3/4) • Minors? • • 13 y or younger: informed consent by parents/guardians only 14 -15 y: IC by parents/guardians & informed assent by adolescents 16 -17 y: IC by adolescent & parents have the right to oppose 18 y onwards: IC by participant • The informed consent form is a formally signed agreement between researcher and participant but does not exempt researchers from responsibilities or liabilities. • Informed consent forms are filled out in duplicate (i. e. , for the researcher & the participant). Consent can also be given through for instance an online form

Informed consent (4/4) • Passive Informed Consent relates to the practice of asking participants to signal only if they DO NOT want to participate, and to take consent for granted in the absence of objection. By and large, this procedure is discouraged by the SMEC (especially in minors or vulnerable individuals). Only in special cases, a motivated request for passive consent may be included in the application dossier. • Also for oral rather than written consent, a motivated request will be necessary and is generally discouraged by the SMEC. For a template informed consent form see the SMEC website: https: //www. kuleuven. be/english/research/ethics/committees/smec

COVID-19 • Check the SMEC website (update coming soon) https: //www. kuleuven. be/english/research/ethics/committees/smec Important points • Preference is given to research without face-to-face contact • If face-to-face contact is necessary: • Describe the safety measures in the COVID-19 annex • Add the confirmation of external institutions if applicable

Final points of interest • A study may not employ deception unless the use of deception techniques can be justified by the study’s significant prospective scientific or applied value and when there is no alternative procedure for effectively collecting the data. • Debriefing (in case of deception asap after data collection), feedback & follow-up

Outcome • Accepted Following favorable review of an application dossier, the G-number found in the PRET-platform will signify the approval of the SMEC committee. Approval remains valid up to 4 years of its effective date of issue. • Minor revision Some details are missing to provide you with a final approval. You will receive a list of the (small) adjustments required. The revised application will have to be reviewed by SMEC again. • Major revision In case of rejection of a research protocol or procedure, the applicant will receive a message that mentions the reasons for rejection. Rejected applications can be resubmitted within 6 months.
Need help? • Click the info buttons • Ask your promotor. All applications are treated as a joint document by student and promotor (and other involved researchers) • For further questions: • About the PRET platform and the privacy/GDPR check pret@kuleuven. be • About the ethics check smec@kuleuven. be

Outline 1. Ethical review boards KU Leuven & mission 2. PRET application platform • Privacy/GDPR check • Ethics check • • Social and scientific value Informed consent COVID-19 Other points of interest • Outcome 3. Cases

Case 1 • A lecturer at KU Leuven will collect data from his students during the last half hour of his lecture. The aim of the study is to detect the structure of personality dimensions in young adulthood. No identifiers will be asked for and, as such, the data are 100% anonymous. • What is most likely the advice by the SMEC?

Case 2 • A researcher wants to collect data on the effects of the changing climate in China. He claims that the rural community will not trust him to sign any ICF documents (or will not give truthful info because no critique on the nation’s climate policy is allowed). Therefore, he suggests to only receive an oral consent. • What is most likely the advice by the SMEC?

Case 3 • SMEC receives a complaint from a student: at the end of a collective experiment the researcher distributed one list on which all participants needed to add their name and bank account number in order to receive their reimbursement. • Will SMEC follow-up on this complaint?

Case 4 • A researcher wants to investigate which pornographic material on the internet 10 -12 year olds are interested in. They have to indicate “yes” or “no” whether they ever actively came across a picture on the internet, like the one depicted in the questionnaire. • What is most likely the advice by the SMEC?

Case 5 • A researcher is following up on Quality of Life data in adolescents to better understand the impact of the environment on the development of anxiety disorders. There will be 5 waves of data collection. The researcher claims she uses fully anonymous data. • What is most likely the advice by the SMEC?
Please give your feedback in the evaluation form: https: //ppw. kuleuven. be/onderwijs/studentenportaal/masterproef/ev aluatie-informatiesessie-ethische-toetsing
- Slides: 39