ETHICS Birgit Schwenzer SSMC and MRSECDMR Young Investigator

ETHICS Birgit Schwenzer, SSMC and MRSEC/DMR Young Investigator Workshop 2017 June 8 th, 2017

Jan-Hendrik Schön (Bell Labs) Patrick G. Harran (UCLA)

Why do we Need Ethics? • Honesty and integrity are requirements for the sane practice of science • Represent accepted practices and norms • Good citizenship It is the law: If you submit proposals to the Federal Government, strict set of rules determine acceptable conduct.

Two Aspects of Ethics in Research 1. Ethical considerations when submitting proposals and doing research 2. The ethical proposal review
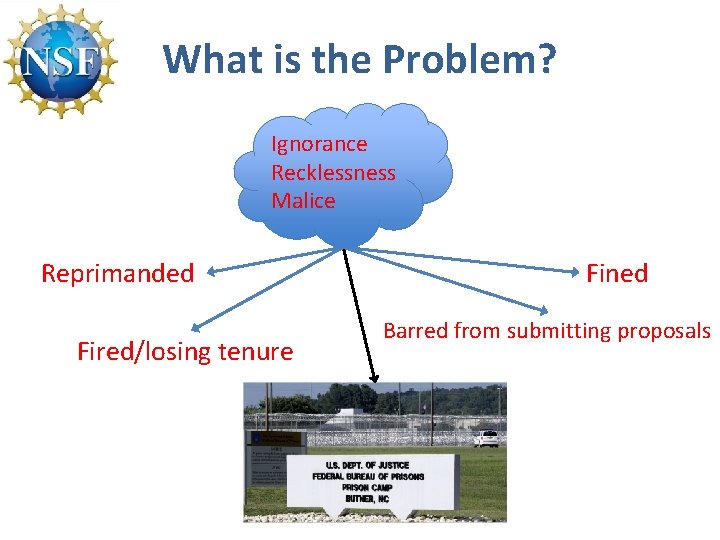
What is the Problem? Ignorance Recklessness Malice Reprimanded Fired/losing tenure Fined Barred from submitting proposals

NSF Office of Inspector General • Provides independent oversight of the agency’s programs and operations (audits/investigations) • Prevents and detects fraud, waste, and abuse • Submits semiannual summary report of its activities to the Congress www. nsf. gov/oig

(Research) Misconduct An instance of fabrication, falsification, or plagiarism in proposing/performing/reviewing research, or in reporting research results – Fabrication: making up results and recording or reporting them – Falsification: manipulating research materials, equipment, or processes or changing or omitting data or results such that the research is not accurately represented in the research record. – Plagiarism: the appropriation of another person’s ideas, processes, results, or words without giving appropriate credit Retaliation against a person who in good faith reported or provided information about misconduct

False/Inappropriate Charges • Padding travel expenses • Commingling funds – Mixing business and pleasure expenses – Mixing grant funds and personal business expenses • • Charging for time not spent on a grant Charging for work already done Billing for inappropriate items Billing for alcohol or entertainment

Misconduct Investigation Examples (from OIG Report to Congress)

Misconduct Investigation Examples (from OIG Report to Congress)

2. Proposal Review Ethics • Confidentiality • Conflict of Interest • Intentional or implicit bias We know you share our desire to ensure that proposals receive a fair and thoughtful review Confidentiality and Impartiality are important to maintaining the integrity of the review process

Confidentiality • • The contents of proposals are confidential Reviews and reviewers are confidential Ratings and recommendations are confidential Panel membership is confidential Do not discuss proposals, reviews, or any related business with others without the presence of an NSF official You are welcome to share general information about the NSF reviewer/panel experience and process with colleagues

Confidentiality • Principal Investigators will receive anonymous copies of reviews • NSF protects reviews from public disclosure to the extent allowed by law (NSF does not usually disclose reviews in response to Freedom of Information Act requests. ) • Copies of reviews may be shared with NSF funding partners, if any

Conflict of Interest Institutional Individual Family/Household Financial Other WHAT TO DO: If you think you have a COI or other circumstance that could lead to questions about your impartiality, contact the managing program director before you submit a review.

Conflict of Interest You have an institutional conflict with a proposal when, for any institution involved in the proposed project, you: • Hold a current position at the institution, such as a professor, adjunct professor, visiting scientist, or similar position • Have a financial arrangement with the institution (such as consulting or a paid advisory arrangement) • Were employed by an institution within the last 12 months • Are seeking employment at the institution • Have a formal or informal reemployment arrangement with the institution

Conflict of Interest You have an institutional conflict with a proposal when, for any institution involved in the proposed project, you: • • Serve on a Visiting Committee or similar body (Conflict is with the part of the organization advised by the committee) Serve as an Officer, Councilor, Trustee, or on a Governing Board (a position with a fiduciary responsibility) Received and retained money from the organization within the last 12 months – e. g. an honorarium or travel expenses Own stock or other security in the organization that is worth more than the de minimus level (currently $15, 000)

Conflict of Interest You have an individual conflict with a proposal which involves a: • • • Spouse or family member Business or professional partner Former employer (within one year) Present or past Ph. D. advisor or student (for M. Sc. or post-doc, see collaborator) A collaborator within the past 48 months A co-editor within the past 24 months

Conflict of Interest You may have a conflict of interest with a proposal which involves: • • The employer or school of a spouse or child A person living in your household The employer of a person living in your household Your parent’s employer (but not if only financial connection was receipt of honoraria)

Catch All – You May Have a Conflict of Interest If there any other circumstances that could cause your impartiality to be questioned • Would a reasonable person, with all the relevant facts, question your impartiality? • For example, do you share a close personal friendship or strong professional rivalry with one of the principals involved in the proposal? When in doubt, ask the program director!

Implicit Bias In a test of implicit bias, most people associate science and math fields with “male” https: //implicit. harvard. edu/implicit

Ethics Training • It is highly recommended that you give your student researchers training in ethics – this protects you in an event of their indiscretion • Do it with all your students/postdocs • Do it before they have a chance to do something bad • Ask them to sign a letter of recognition that you have provided ethics training, that it covers specific elements of ethics, and that they know that you expect appropriate behavior • When you discover misconduct – Contact your institution’s ethics official – Report it to the Office of Inspector General if it involves NSF-related activities

Resources • Office of Research Integrity – http: //ori. hhs. gov • NSF Office of Inspector General – http: //www. nsf. gov/oig • National Center for Professional & Research Ethics – https: //nationalethicsresourcecenter. net/index. php/home • APS guidelines for professional conduct – https: //www. aps. org/policy/statements/02_2. cfm


Misconduct Investigation Examples (from OIG Reports to Congress) • • Debarment recommended for researcher who used NSF-funded supercomputers to mine bitcoins [PI debarred for 3 years, costs recovered] Two Florida scientists (husband wife) sentenced to 15 and 13 years in federal prison following convictions for fraudulently obtaining $10. 6 M SBIR awards [ordered to pay $10. 6 M, in addition to an order of forfeiture of the same amount] Executive director of non-profit debarred for five years for false certification NSF debars PI for 3 months for falsely certifying 100% effort on NSF awards for summer months while teaching at a foreign institution Texas PI and company employee sentenced to prison for making false statements to SBIR program [5 months in prison, $100 K paid in restitution] PI sentenced to 41 months in prison for applying for and accepting an ARPA-E grant for work already funded by NSF Same scrutiny for NSF employees/rotators

Misconduct investigation examples (from OIG reports to Congress) • Plagiarism – A PI from a NY university copied a portion of another scholar’s research goal into a declined NSF proposal and copied a substantial portion of the methodology into a second declined NSF proposal. The university’s investigation concluded that, although the PI’s act was a significant departure from accepted practices, the PI did not commit research misconduct because he acted with careless intent. – OIG’s review of the university’s report found that the university incorrectly interpreted the reckless standard of intent, determining erroneously that recklessness requires a conscious or purposeful element. OIG’s investigation concluded that the PI acted recklessly and thus committed research misconduct.

Misconduct investigation examples (from OIG reports to Congress) • Plagiarism (cont. ) – A PI at a Michigan university submitted an NSF proposal containing three pages of apparently copied text in the proposal’s five-page literature review. The PI asserted that he had used the American Psychological Association (APA) citation style, that common language use was coincidence, and that he used the author’s words to avoid misinterpretation. OIG determined the PI’s citation practices did not meet APA standards, found his other responses contradictory, and referred the investigation to his institution. – The PI asserted that: 1) NSF policies are nuanced and in conflict with his own literal interpretation; 2) NSF’s requirements for quotation are in conflict with other disciplines’ standards; 3) his field is eclectic and not addressed by NSF policy; and 4) the research proposal is not really research. The university refuted all of these assertions and concluded that the PI committed plagiarism, at least recklessly, which was a significant departure from accepted practices. The university required the PI to participate in a supervisory meeting to discuss the seriousness of his actions, identify steps to prevent future occurrences; take training about plagiarism prevention; and submit all grant proposals to a university official for review for two years. – OIG’s investigation determined that the PI knew his actions constituted plagiarism, knew NSF proposals required attention to citation, and was not unfamiliar with the grant writing process. Consequently, OIG determined he acted knowingly. OIG recommended that NSF require the PI submit certifications and assurances for two years, and certify compliance with university-imposed requirements.
- Slides: 26