Establishing a Compliance Program Presented by Andrew Farhat

Establishing a Compliance Program Presented by Andrew Farhat and Jennifer Buoniconti UL / PPAI Product Safety Consultants

This information is being furnished by PPAI for educational and informational purposes only. The Association makes no warranties or representations about specific dates, coverage or application. Consult with appropriate legal counsel about the specific application of the law to your business and products.

Compliance Programs • Not an option in today’s regulatory environment • Must actively manage and document their compliance processes and procedures • Provide transparency

Compliance Program Objectives 1. Provide confidence when dealing with regulators, recalls and other quality or safety incidents 2. Provide brand protection for your clients 3. Reassurance in knowing that you can provide answers and proof of due diligence in the event of a quality or safety incident 4. Documented compliance with regulatory requirements

Where Do I Begin? 1. Establish a code of conduct 2. Create a supplier vetting and approval process 3. Supplier training 4. Establish a database or system for managing and maintaining all documents associated with your compliance program 5. Develop a plan to handle product safety or quality incidents and recalls 6. Form a relationship with a third party laboratory 7. Keep up to date with regulatory requirements and industry activity
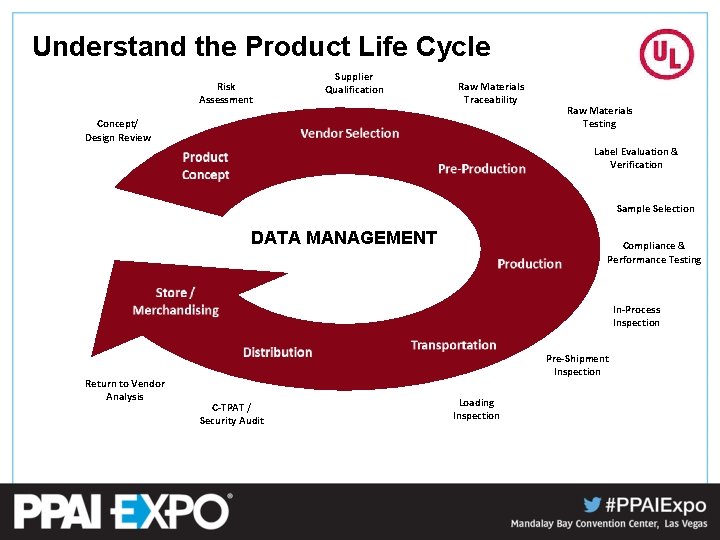
Understand the Product Life Cycle Risk Assessment Supplier Qualification Raw Materials Traceability Concept/ Design Review Raw Materials Testing Label Evaluation & Verification Sample Selection DATA MANAGEMENT Compliance & Performance Testing In-Process Inspection Return to Vendor Analysis Pre-Shipment Inspection C-TPAT / Security Audit Loading Inspection

Concept / Design Review • Can be internal or external but should be done by someone with experience and a human factors background • Goes beyond the standards • Try to anticipate foreseeable use and unintended use scenarios • Identify steps associated with product use (assembly, use, cleaning, storage etc. ) • Understand enhance interaction between users and products • Work with product designers to remove foreseeable hazards prior to manufacture

Risk Assessment • • NEISS data Human factors research journals CPSC publications and data NIH publications – medical literature Social sciences publications Anthropometric data (e. g. CHILDATA) CPSC public consumer product safety database Company internal feedback
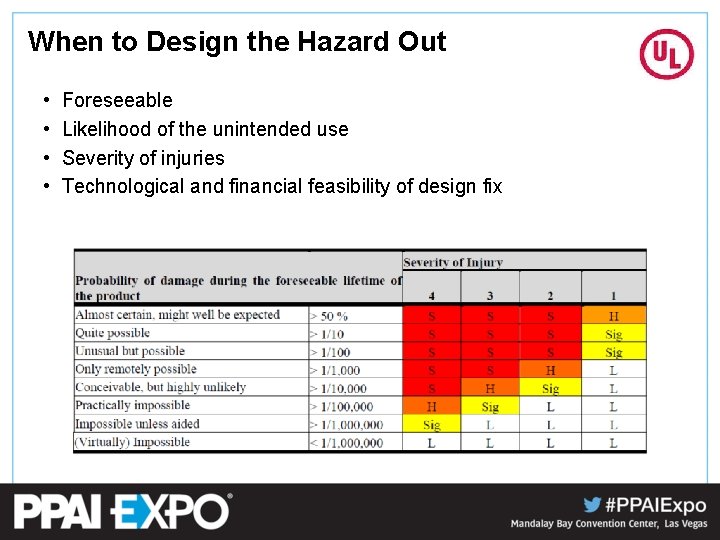
When to Design the Hazard Out • • Foreseeable Likelihood of the unintended use Severity of injuries Technological and financial feasibility of design fix

Supplier Qualification Audits • • • Initial factory qualifications Quality & Capability audit Quality assessment systems Production monitoring c. GMPs HACCP SQF Sanitation Product safety OSHA Social Compliance audit

Quality & Capability Audit • Identifies strengths and weaknesses • Determine whether products being manufactured will meet your quality standards and expectations • Assess whether production volume and schedules will be met • Provides objective evidence of processes • Allow the supplier to initiate corrective and preventative action

Social Compliance Audit • Evaluate supplier’s social and environmental compliance with local labor standards, local workplace practices and industry standards • Provide key insight and intelligence needed to make critical business sourcing decisions • Measure compliance with national legislation and international standards for: » » » • Child labor Collective bargaining Discrimination Document review Dormitories Environment Freedom of association Harassment and abuse Health and safety Prison or forced labor Wages Work hours Allow the supplier to initiate corrective and preventative action

Raw Materials Traceability • Determine ethical sourcing by tracing product components, verifying documentation of origin and trade, assessing workplace conditions • Objectively confirm the source of raw materials • Help comply with industry accreditations or specific client requirements

Raw Materials Testing • Confirm finished product will meet regulatory requirements and/or client specific requirements • Determine whether product will meet specifications for intended use and expected life cycle • Allow for corrective action prior to production

Label Evaluation & Verification • Labeled claim substantiation • Ensure package and product label meets regulatory requirements for the intended market • Allow for corrective action prior to production

Random/Representative Sample Selection • Avoid ‘golden samples’ • Basis for verifying compliance of entire lot • Typically selected from product in final packaged state

Compliance & Performance Testing • Meet or exceed existing standards and regulations • Standards and regulations are the minimum requirements • Over half of all toys recalled in previous years complied with mandatory and voluntary standards!

Compliance & Performance Testing • • • Safety Performance Quality Regulatory Requirements Function and Durability Claims Substantiation Sensory Evaluation Product Comparisons Consumer Studies Focus Groups

Quality Inspections • Top of Production (TOP) » Raw Materials • Initial Production (IPI) / First Line Inspections » First 200 units produced • In-Line / In-Process Inspections (DUPRO) » Between 20% and 40% produced • Final Random Inspections (FRI) » Minimum; 100% produced, 80% packed • Loading Inspections » Count, confirm load, seal container at factory or port • Production Monitoring » On-site monitoring of each operation throughout the production Inspections are typically conducted using ANSI/ASQ-Z 1. 4 -2008 sampling procedures.

Quality Inspection Benefits • Reduction of non-compliant products reaching your customers • Prevents defective products from being shipped from the manufacturer • Higher quality levels = higher customer satisfaction • Reduction in product returns • Cost effective means to help ensure that products meet your quality standards prior to shipment

Customs-Trade Partnership Against Terrorism (C-TPAT) • Voluntary supply chain security program, launched in November of 2001 • Over 10, 000 certified partners • Partners must ensure the integrity of their supply chain • Partners are considered low-risk • C-TPAT is base requirement for Importer Self Assessment

Port Surveillance – FY 2013 • CPSC’s targeting system, known as the RAM (risk assessment methodology), was deployed as a pilot project in late 2011 and is currently being used. • The RAM allows CPSC investigators to analyze certain data provided by the CBP about shipments arriving at U. S. ports of entry, and then make risk-based decisions about which shipments to inspect. • For all of FY 2013, more than 12. 5 million units of violative imports were prevented from reaching the hands of consumers.
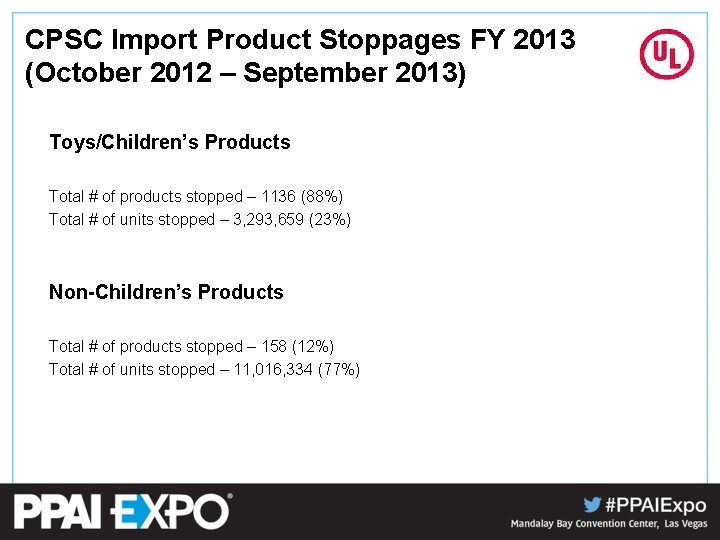
CPSC Import Product Stoppages FY 2013 (October 2012 – September 2013) Toys/Children’s Products Total # of products stopped – 1136 (88%) Total # of units stopped – 3, 293, 659 (23%) Non-Children’s Products Total # of products stopped – 158 (12%) Total # of units stopped – 11, 016, 334 (77%)

CPSC Import Stoppage Details 3 rd/4 th Quarters FY 2013 (April – September 2013) Toys/Children’s Products Total # of products stopped – 548 (89%) Total # of units stopped - 2, 091, 737 (25%) Non-Children’s Products Total # of products stopped – 68 (11%) Total # of units stopped – 6, 123, 150 (75%) Primary Violation/Defect Lead – 225 (Paint 14; Content 211) (41%) Phthalates – 114 (20%) Tracking Labels – 62 (11%) Art Materials - 49 (8%) Small Parts – 39 (7%) Primary Violation/Defect Fireworks – 51 (75%) Cigarette/other lighters – 8 (12%) Mattress Flammability – 6 (9%) Generator Labeling – 2 (3%) Hairdryers – 1 (1%)

Common Issues That May Trigger Inspection • Repetitive use of foreign suppliers found in violation • Incorrect filing of manufacturer ID on entry documents • Un-manifested and un-invoiced merchandise • Missing invoices • Delay of entry documents from broker • Class of product under review by CPSC to monitor compliance • Overlapping IPR and safety concerns • Certificate identifies a non-accredited lab

Avoid Pitfalls That Could Delay Entry • Make certificates readily available and that they include correct citations • Make sure 3 rd party testing body is accredited by CPSC • Ensure goods are classified appropriately • Include product detail on invoice • Join C-TPAT • Sign up for ISA and ISA-PS (Importer Self-Assessment programs) • Don’t import products that are considered Substantial Product Hazards by the CPSC!

Summary Importers • Know your suppliers • Know your product • Stay up-to-date on regulations • Participate in C-TPAT and ISA programs Exporters • Know your suppliers • Know your product • Ensure compliance with applicable safety requirements of country of distribution • Work with trusted partner within country of distribution

Once on the market… • Market surveillance testing • DC inspections • Vendor return analysis • Failure evaluation • Recall assistance • Focus groups

Regulatory Research Monitor Proposed / Pending Regulations & Legislation Westlaw–Capital Watch, Health Canada, US Federal Register, Info. Minder, EUROPA EU, Gov. Track, Australian Government Com. Law Proposed New / Amended Standards BSI, ASTM, CEN, IEC, SABS, UL, SAI Global, RAPEX, AATCC, ISO, CPSC, NIST, Notify. US, NSSN, British Standards On. Line Other Resources Product Safety Daily, Product Safety Australia, CPSC Bulletins, TIA, CA OEHHA Press Releases, SATRA, British Standards On. Line, ECHA, Laboratory Websites/Newsletters

Technical Committees and Meetings • • • ASTM ASQ TIA JVC CPSC ICPHSO

Product Safety Resources PPAI: www. ppai. org Product Safety powered by PPAI: http: //www. ppai. org/inside-ppai/product -safety/ Consumer Product Safety Commission: www. cpsc. gov, www. recalls. gov American National Standards Institute: www. ansi. org or www. astm. org FDA: www. fda. gov UL: www. ul. com; Haley. Sprague@ul. com Questions? Anne. L@ppai. org
- Slides: 31