Establishing a Clinical Genomics Program at an Academic

Establishing a Clinical Genomics Program at an Academic Medical Center Jason Merker, M. D. , Ph. D. Co-director, Stanford Clinical Genomics Service Regional APC/PDAS Meeting, Kauai 10/23/2014

Introduction to Clinical Genomics (in 5 Slides)

Wellcome Collection – Medicine Now 3. 4 billion units of DNA code: • 127 volumes • 1, 000 pages per volume

Size of targeted regions in assays Genome – 127 volumes Exome – 2. 5 volumes 500 gene panel – 40 pages 50 gene panel – 4 pages Single gene – <1/10 of a page

Tumor/Normal or Trio Genome Sequencing Data
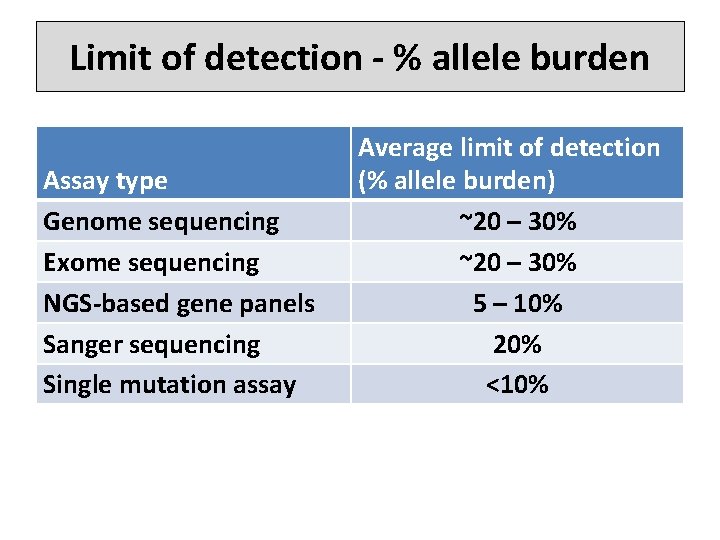
Limit of detection - % allele burden Assay type Genome sequencing Exome sequencing NGS-based gene panels Sanger sequencing Single mutation assay Average limit of detection (% allele burden) ~20 – 30% 5 – 10% 20% <10%
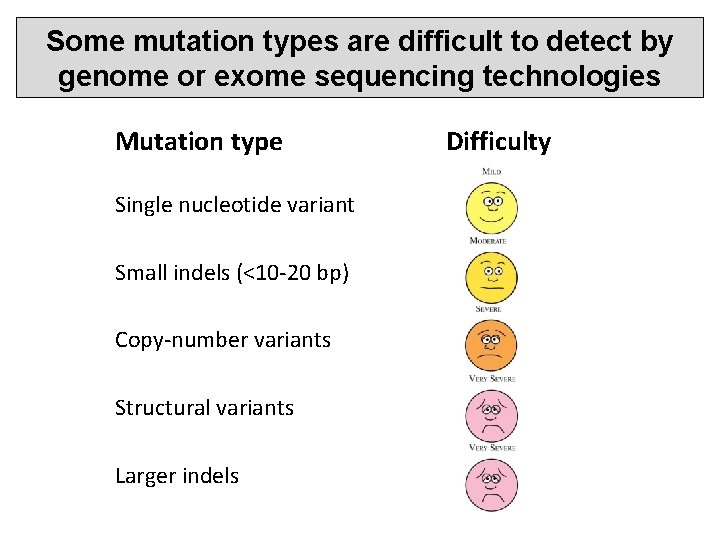
Some mutation types are difficult to detect by genome or exome sequencing technologies Mutation type Single nucleotide variant Small indels (<10 -20 bp) Copy-number variants Structural variants Larger indels Difficulty

Introduction to Clinical Genomics at Stanford
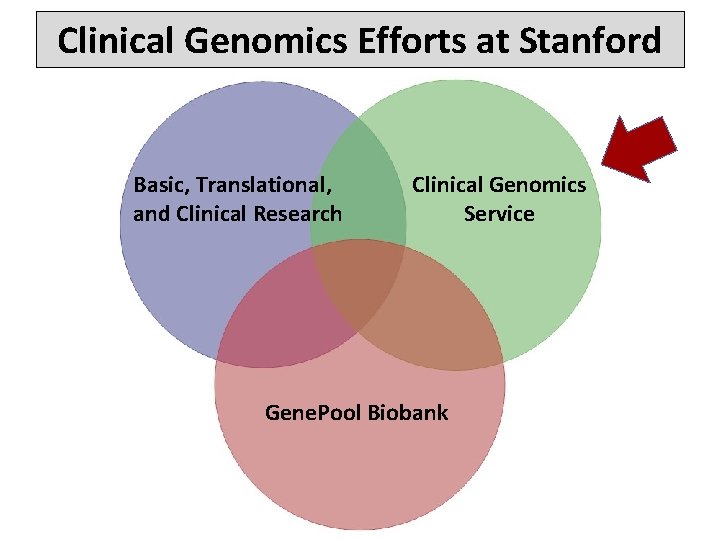
Clinical Genomics Efforts at Stanford Basic, Translational, and Clinical Research Clinical Genomics Service Gene. Pool Biobank
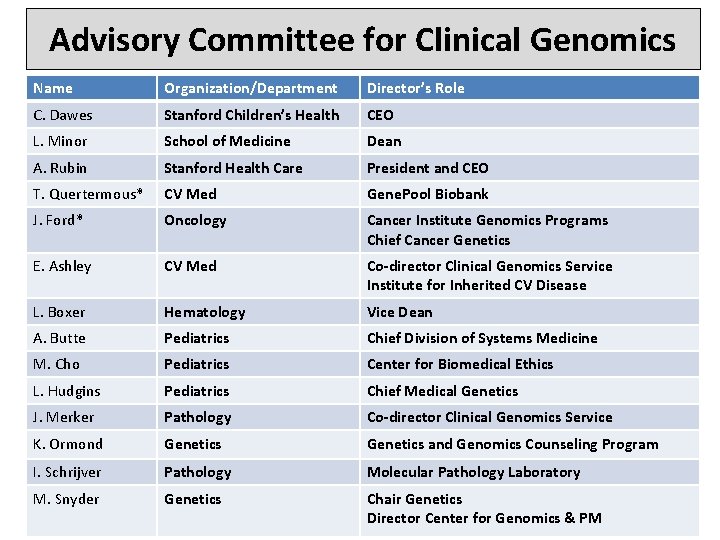
Advisory Committee for Clinical Genomics Name Organization/Department Director’s Role C. Dawes Stanford Children’s Health CEO L. Minor School of Medicine Dean A. Rubin Stanford Health Care President and CEO T. Quertermous* CV Med Gene. Pool Biobank J. Ford* Oncology Cancer Institute Genomics Programs Chief Cancer Genetics E. Ashley CV Med Co-director Clinical Genomics Service Institute for Inherited CV Disease L. Boxer Hematology Vice Dean A. Butte Pediatrics Chief Division of Systems Medicine M. Cho Pediatrics Center for Biomedical Ethics L. Hudgins Pediatrics Chief Medical Genetics J. Merker Pathology Co-director Clinical Genomics Service K. Ormond Genetics and Genomics Counseling Program I. Schrijver Pathology Molecular Pathology Laboratory M. Snyder Genetics Chair Genetics Director Center for Genomics & PM

Stanford Clinical Genomics Service • Directors – Jason Merker (Path), Euan Ashley (CV Med) • Department – Pathology • Goal – Build a clinical laboratory service at Stanford University Medical Center that uses genome sequencing to evaluate adult and pediatric patients with unexplained genetic diseases. • Pilot – Develop analysis/curation pipeline and perform genome sequencing on 4 patient populations (100 cases total): – – Heritable cancer predisposition Heritable cardiovascular disorders Pediatric syndromes Familial adverse drug reactions or sensitivity
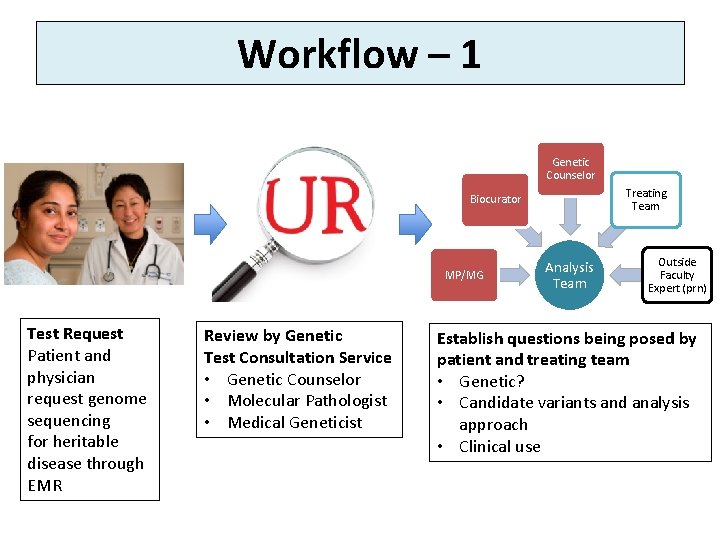
Workflow – 1 Genetic Counselor Treating Team Biocurator MP/MG Test Request Patient and physician request genome sequencing for heritable disease through EMR Review by Genetic Test Consultation Service • Genetic Counselor • Molecular Pathologist • Medical Geneticist Analysis Team Outside Faculty Expert (prn) Establish questions being posed by patient and treating team • Genetic? • Candidate variants and analysis approach • Clinical use
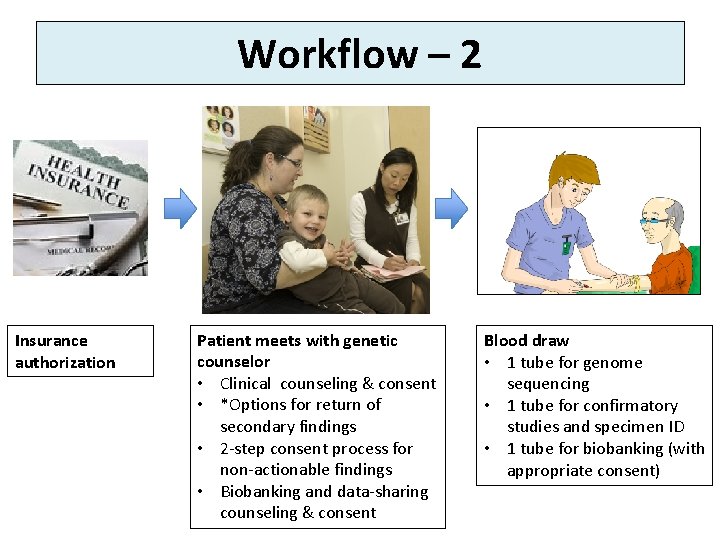
Workflow – 2 Insurance authorization Patient meets with genetic counselor • Clinical counseling & consent • *Options for return of secondary findings • 2 -step consent process for non-actionable findings • Biobanking and data-sharing counseling & consent Blood draw • 1 tube for genome sequencing • 1 tube for confirmatory studies and specimen ID • 1 tube for biobanking (with appropriate consent)
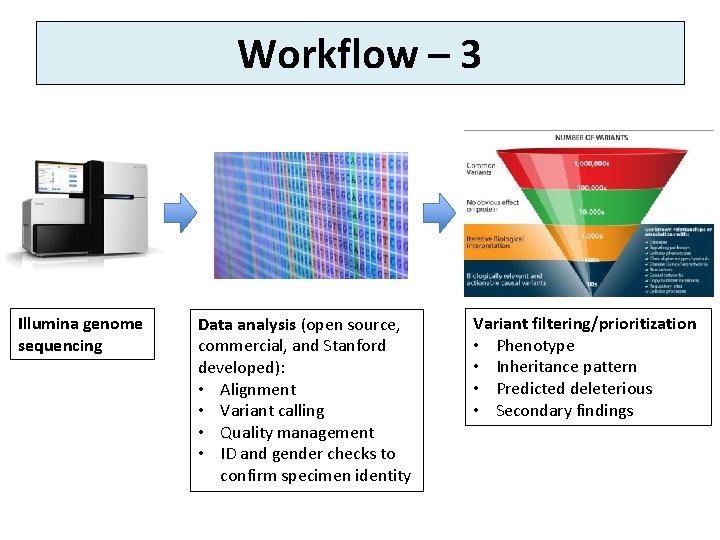
Workflow – 3 Illumina genome sequencing Data analysis (open source, commercial, and Stanford developed): • Alignment • Variant calling • Quality management • ID and gender checks to confirm specimen identity Variant filtering/prioritization • Phenotype • Inheritance pattern • Predicted deleterious • Secondary findings
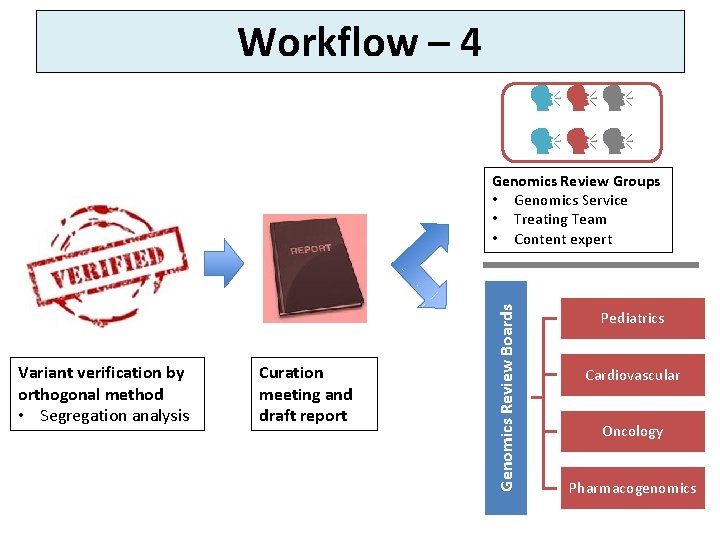
Workflow – 4 Variant verification by orthogonal method • Segregation analysis Curation meeting and draft report Genomics Review Boards Genomics Review Groups • Genomics Service • Treating Team • Content expert Pediatrics Cardiovascular Oncology Pharmacogenomics
Workflow – 5 Final report generated and uploaded to EMR Patient meets with genetic counselor and relevant members of treatment team Yearly re-analysis upon request • Improved analysis • Improved sequencing • Increased medical knowledge
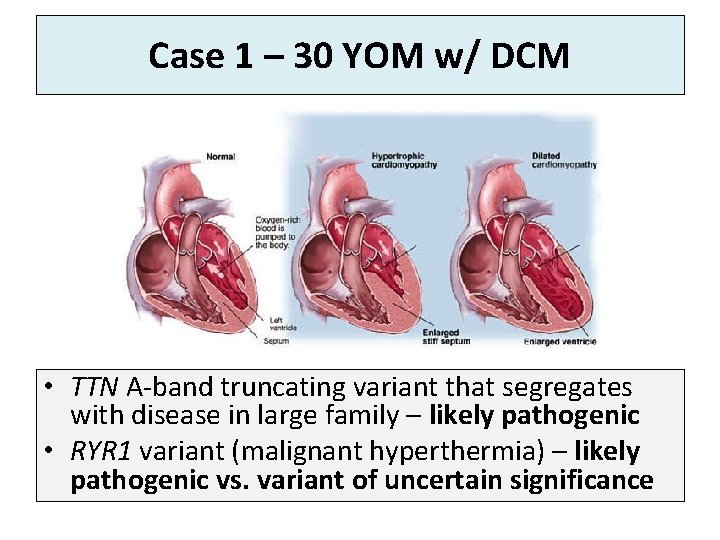
Case 1 – 30 YOM w/ DCM • TTN A-band truncating variant that segregates with disease in large family – likely pathogenic • RYR 1 variant (malignant hyperthermia) – likely pathogenic vs. variant of uncertain significance
Case 2

Clinical Genetic Test Consultation Service
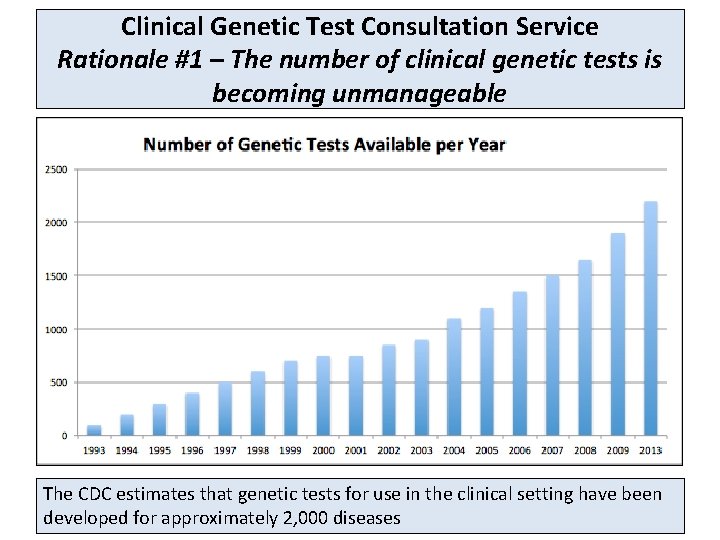
Clinical Genetic Test Consultation Service Rationale #1 – The number of clinical genetic tests is becoming unmanageable The CDC estimates that genetic tests for use in the clinical setting have been developed for approximately 2, 000 diseases

Clinical Genetic Test Consultation Service Rationale #2 – The number of misorders for complex genetic testing is high “Approximately 25% of all requests for complex genetic tests assessing germ line mutations were changed following review. ” Miller CE et al. 2014. Am J Med Genet Part A 164 A: 1094– 1101.

Clinical Genetic Test Consultation Service – Summary of rationales • The number, indications, and complexity of genetic tests offered have been increasing, and will continue to do so for the foreseeable future. • It is therefore not surprising that mistakes often occur in the ordering of complex genetic tests. • Incorrect ordering of genetic tests results in unnecessary costs to the healthcare system, but more importantly adversely affects the care of our patients – Failure or delays in getting the needed test results – Communication of results from the incorrect test – Providing genetic information that was neither requested nor desired by the patient

Clinical Genetic Test Consultation Service – Personnel Molecular Pathologist Genetic Counselor Medical Geneticist

Clinical Genetic Test Consultation Service 1. Provide consultation to SUMC healthcare providers needing further information on available genetic testing. 2. Review all quests for send-out genetic testing from Stanford Clinical Laboratories to identify and help correct genetic test misorders. 3. Work with Genetic Test Utilization Committee develop innovative, provider-friendly ways to educate our physicians about genetic test utilization (e. g. , pop-up windows in EMR offering test consultation or other educational information). 4. Assist departments and divisions with educational activities related to genetic test utilization (e. g. , seminars, presentation to new residents) and with establishing protocols for genetic test ordering for common use cases

Clinical genomics educational efforts

Open Didactic Core Curriculum in Genomic Medicine 1. Experimental methods for measuring and manipulating DNA/RNA 2. Fundamentals of human genetic variation 3. Microarrays and analysis of hybridization data 4. Sequencing methods 5. Heritable genetic disorders 6. Acquired mutations in human cancers I: solid tumors 7. Acquired mutations in human cancers II: hematopoietic malignancies 8. Pharmacogenomics 9. HLA genetics 10. Ethical, legal, and economic implications of clinical genomic testing Schrijver I et al. J Mol Diagn. 2013; 15: 141.

Elective Course in Advanced Genomic Medicine 1. Next-generation sequencing methods 2. 0 2. Human genetic variation 2. 0 3. DNA sequence analysis methods I: sequence databases and files 4. DNA sequence analysis methods II: sequence alignment algorithms 5. DNA sequence analysis methods III: genome assembly and analysis 6. Introduction to scripting programming languages 7. Statistical tools for sequence analysis and genomics Schrijver I et al. J Mol Diagn. 2013; 15: 141.

Elective Course in Advanced Genomic Medicine 1. Next-generation sequencing methods 2. 0 2. Human genetic variation 2. 0 3. DNA sequence analysis methods I: sequence databases and files 4. DNA sequence analysis methods II: sequence alignment algorithms 5. DNA sequence analysis methods III: genome assembly and analysis 6. Introduction to scripting programming languages 7. Statistical tools for sequence analysis and genomics Schrijver I et al. J Mol Diagn. 2013; 15: 141.
New Elective Course in Advanced Genomic Medicine • Genome and exome analysis for heritable disease • Tumor/normal sequencing analysis • RNA sequencing analysis • Unix commands and basic scripting

End
- Slides: 30