Estabilidade Relativa dos Dienos Dr Wolfs CHM 201

Estabilidade Relativa dos Dienos Dr. Wolf's CHM 201 & 202 10 -44
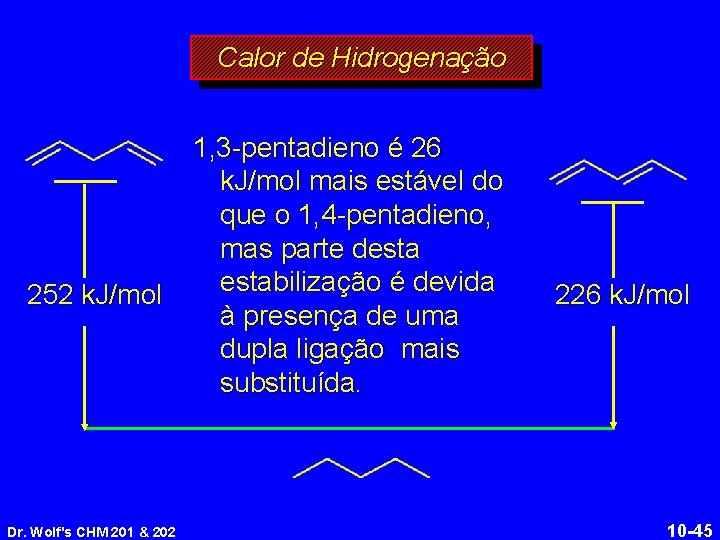
Calor de Hidrogenação 252 k. J/mol Dr. Wolf's CHM 201 & 202 1, 3 -pentadieno é 26 k. J/mol mais estável do que o 1, 4 -pentadieno, mas parte destabilização é devida à presença de uma dupla ligação mais substituída. 226 k. J/mol 10 -45
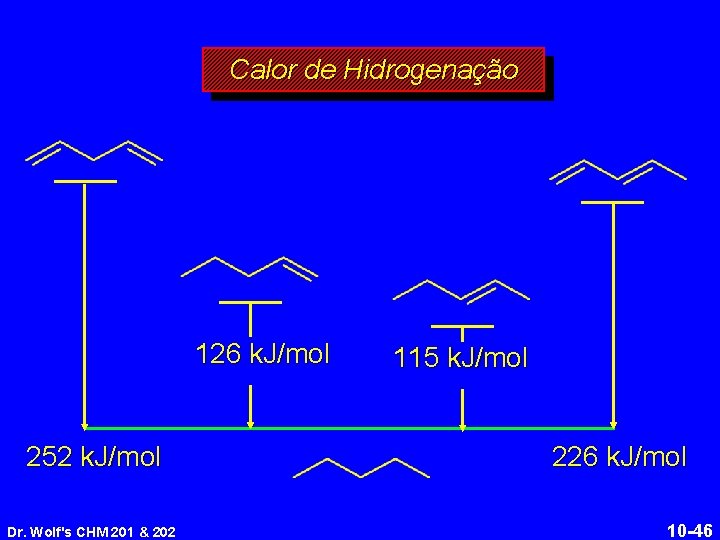
Calor de Hidrogenação 126 k. J/mol 252 k. J/mol Dr. Wolf's CHM 201 & 202 115 k. J/mol 226 k. J/mol 10 -46
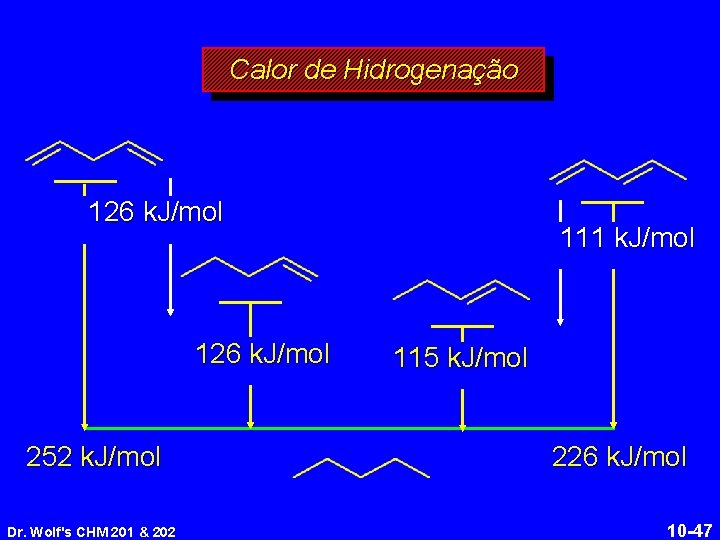
Calor de Hidrogenação 126 k. J/mol 252 k. J/mol Dr. Wolf's CHM 201 & 202 111 k. J/mol 115 k. J/mol 226 k. J/mol 10 -47
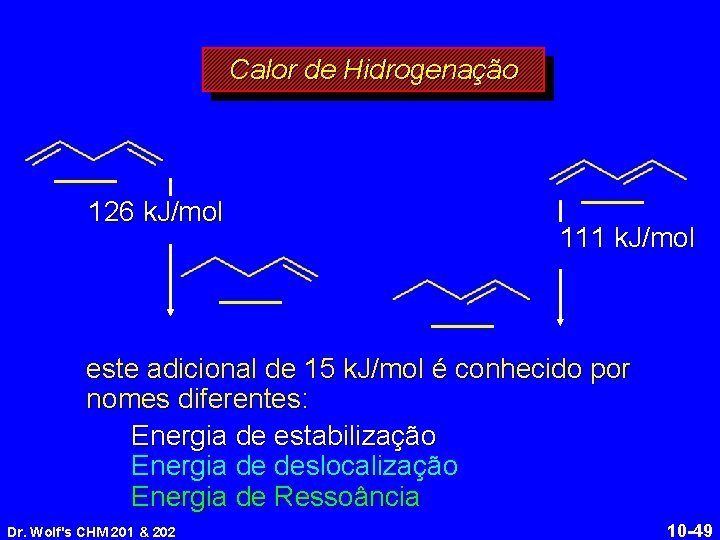
Calor de Hidrogenação 126 k. J/mol 111 k. J/mol este adicional de 15 k. J/mol é conhecido por nomes diferentes: Energia de estabilização Energia de deslocalização Energia de Ressoância Dr. Wolf's CHM 201 & 202 10 -49

Heats of Hydrogenation Cumulated double bonds have relatively high heats of hydrogenation H 2 C C CH 2 + 2 H 2 CH 3 CH 2 CH 3 DH° = -295 k. J H 2 C CH 2 CH 3 + H 2 CH 3 CH 2 CH 3 DH° = -125 k. J Dr. Wolf's CHM 201 & 202 10 -50

10. 7 Bonding in Conjugated Dienes Dr. Wolf's CHM 201 & 202 10 -51
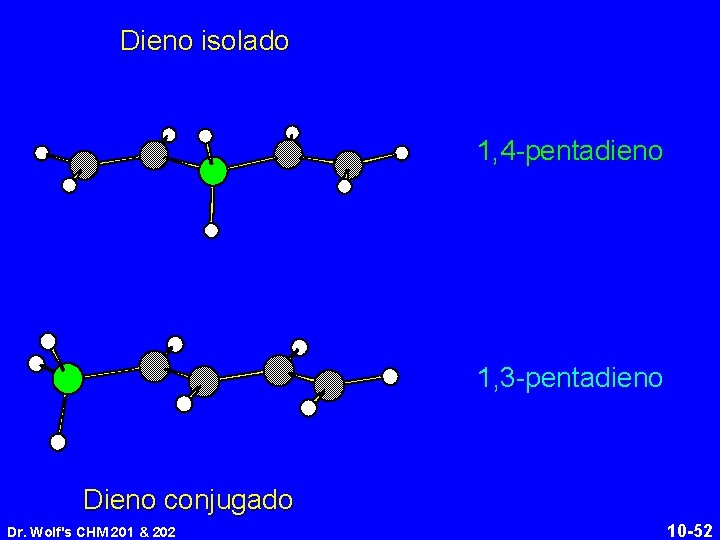
Dieno isolado 1, 4 -pentadieno 1, 3 -pentadieno Dieno conjugado Dr. Wolf's CHM 201 & 202 10 -52
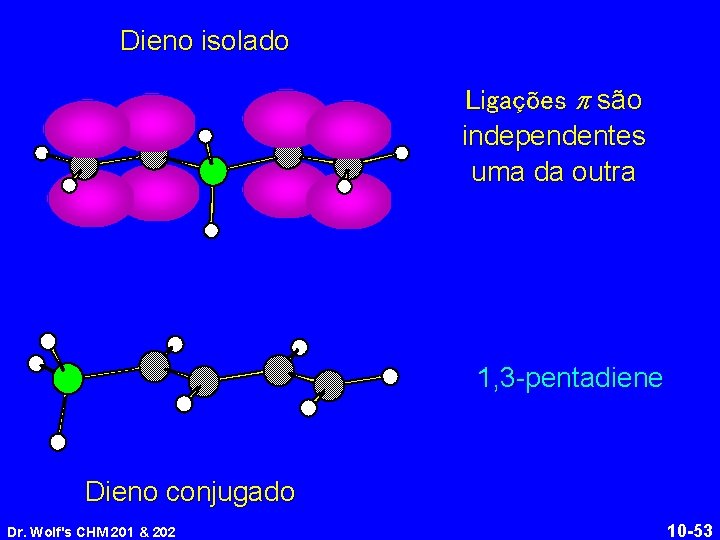
Dieno isolado Ligações p são independentes uma da outra 1, 3 -pentadiene Dieno conjugado Dr. Wolf's CHM 201 & 202 10 -53
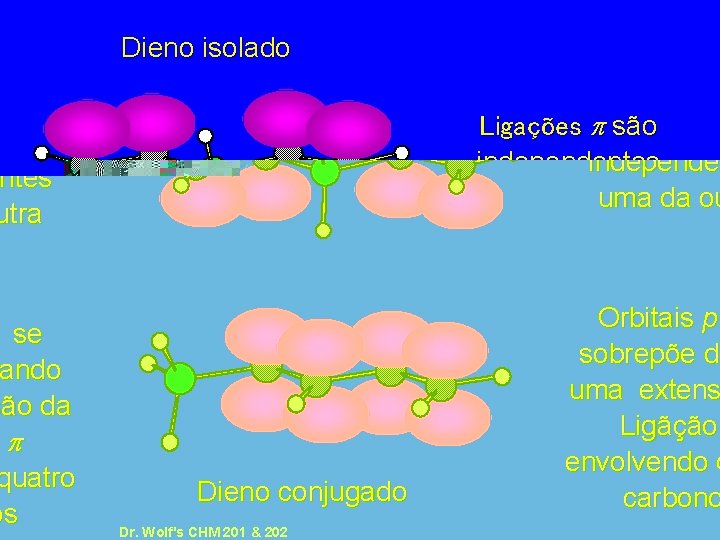
Dieno isolado Ligações p são independentes uma da outra Dieno conjugado Dr. Wolf's CHM 201 & 202 Orbitais p se sobrepõe dando uma extensão da Ligãção p envolvendo quatro carbonos 10 -54
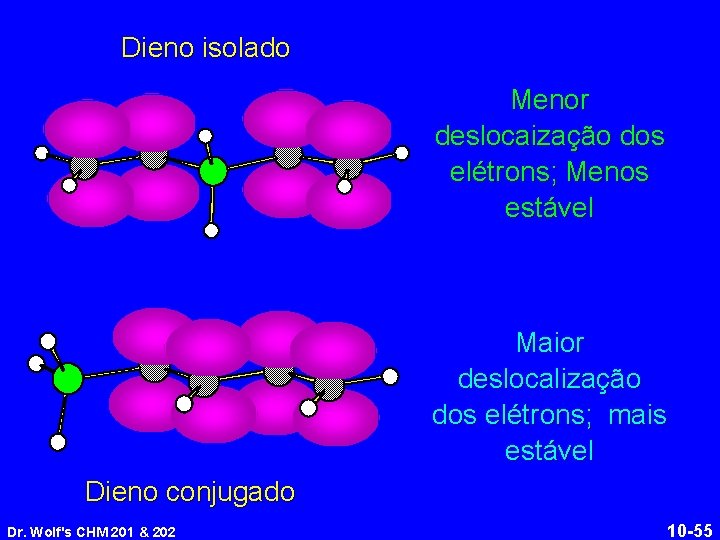
Dieno isolado Menor deslocaização dos elétrons; Menos estável Maior deslocalização dos elétrons; mais estável Dieno conjugado Dr. Wolf's CHM 201 & 202 10 -55
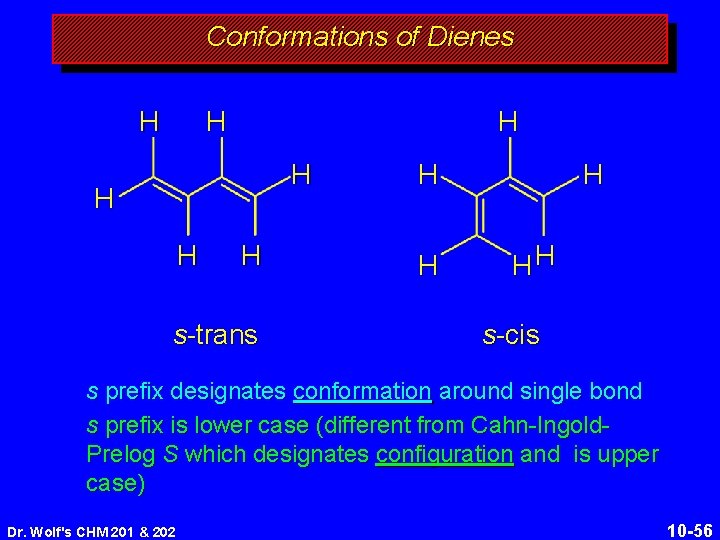
Conformations of Dienes H H H H s-trans H HH s-cis s prefix designates conformation around single bond s prefix is lower case (different from Cahn-Ingold. Prelog S which designates configuration and is upper case) Dr. Wolf's CHM 201 & 202 10 -56
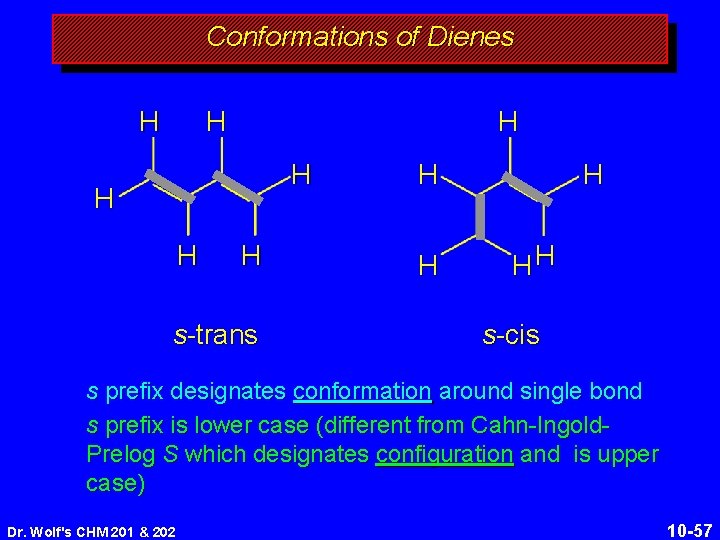
Conformations of Dienes H H H H s-trans H HH s-cis s prefix designates conformation around single bond s prefix is lower case (different from Cahn-Ingold. Prelog S which designates configuration and is upper case) Dr. Wolf's CHM 201 & 202 10 -57
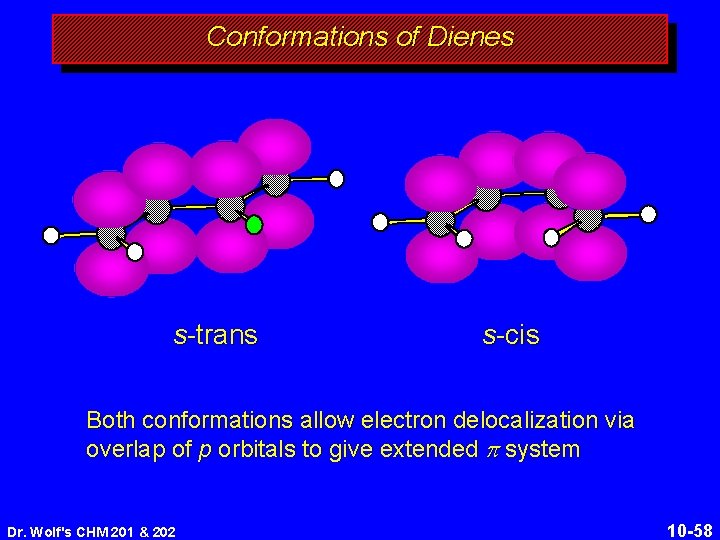
Conformations of Dienes s-trans s-cis Both conformations allow electron delocalization via overlap of p orbitals to give extended p system Dr. Wolf's CHM 201 & 202 10 -58

s-trans is more stable than s-cis Interconversion of conformations requires two p bonds to be at right angles to each other and prevents conjugation 12 k. J/mol Dr. Wolf's CHM 201 & 202 10 -59
Dr. Wolf's CHM 201 & 202 10 -60
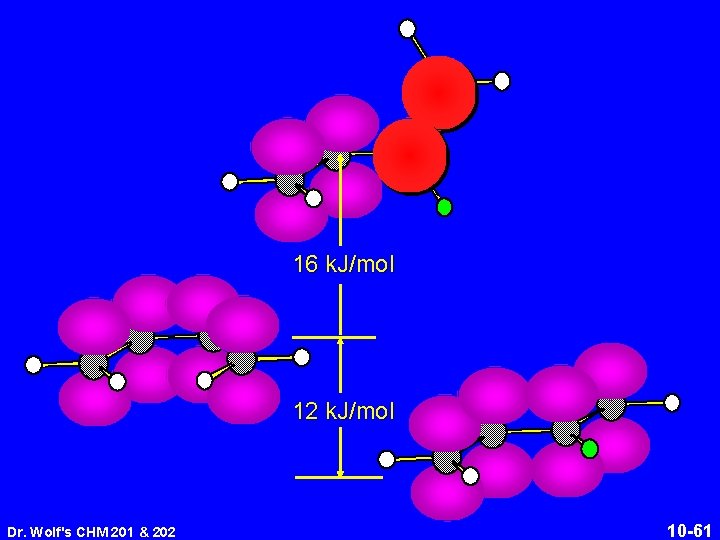
16 k. J/mol 12 k. J/mol Dr. Wolf's CHM 201 & 202 10 -61

10. 8 Bonding in Allenes Dr. Wolf's CHM 201 & 202 10 -62
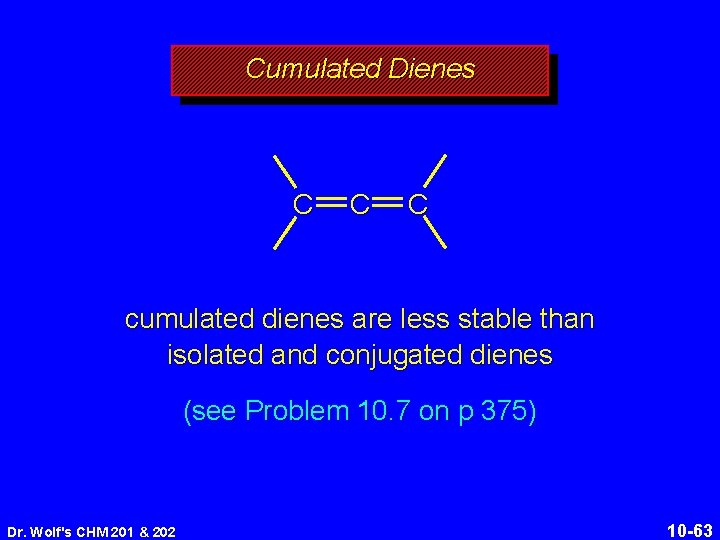
Cumulated Dienes C C C cumulated dienes are less stable than isolated and conjugated dienes (see Problem 10. 7 on p 375) Dr. Wolf's CHM 201 & 202 10 -63
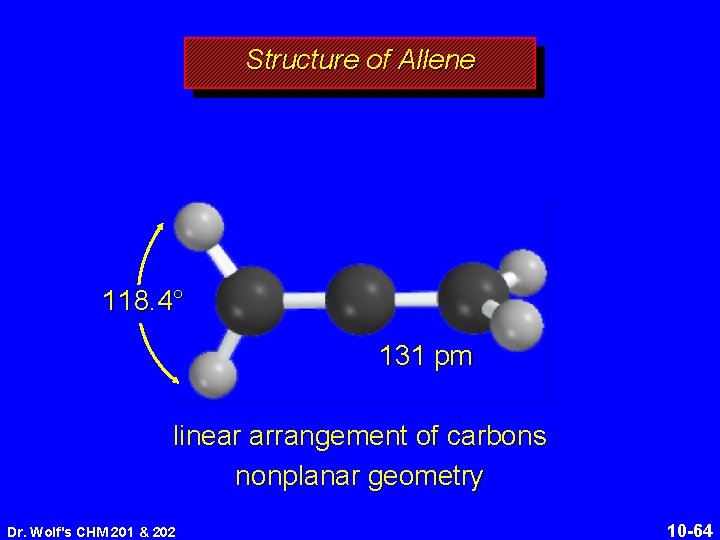
Structure of Allene 118. 4° 131 pm linear arrangement of carbons nonplanar geometry Dr. Wolf's CHM 201 & 202 10 -64
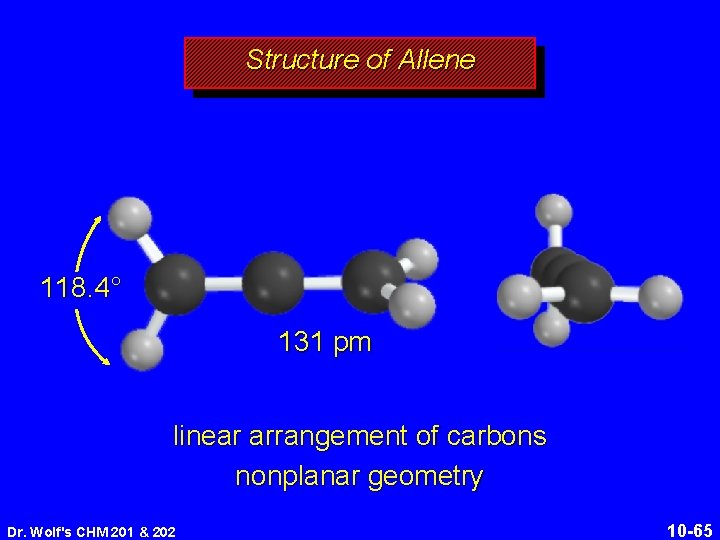
Structure of Allene 118. 4° 131 pm linear arrangement of carbons nonplanar geometry Dr. Wolf's CHM 201 & 202 10 -65
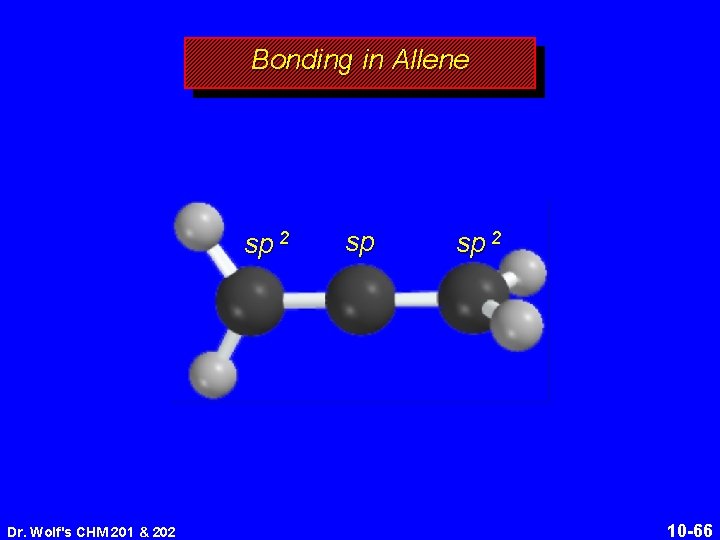
Bonding in Allene sp 2 Dr. Wolf's CHM 201 & 202 sp sp 2 10 -66
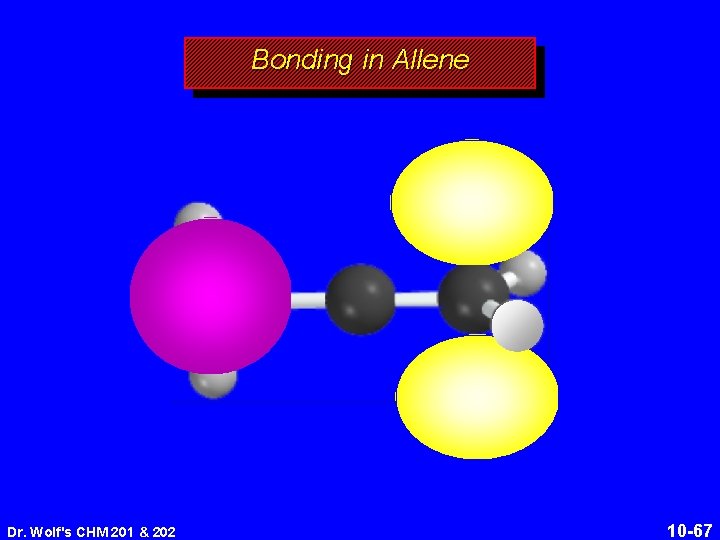
Bonding in Allene Dr. Wolf's CHM 201 & 202 10 -67
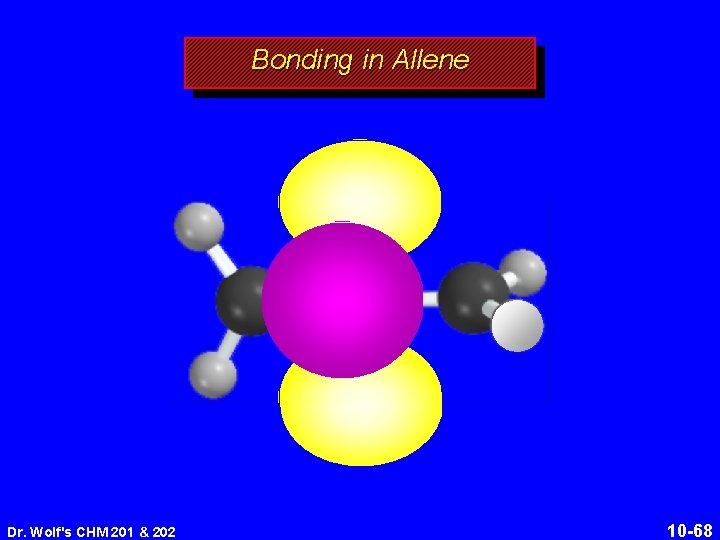
Bonding in Allene Dr. Wolf's CHM 201 & 202 10 -68
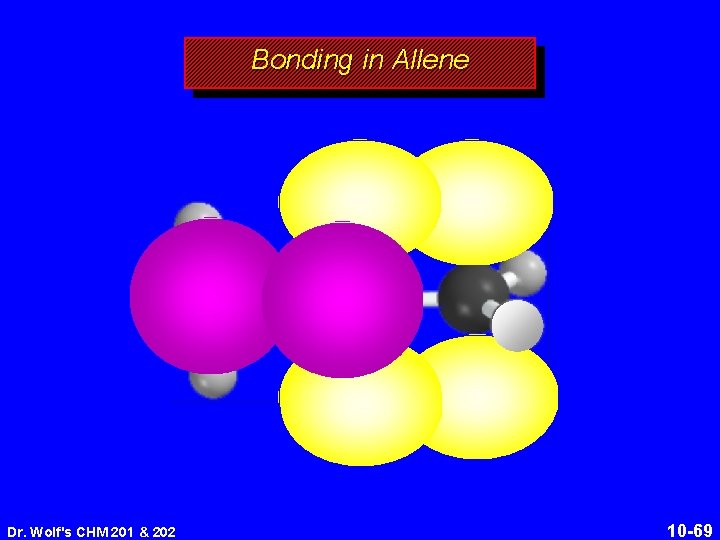
Bonding in Allene Dr. Wolf's CHM 201 & 202 10 -69
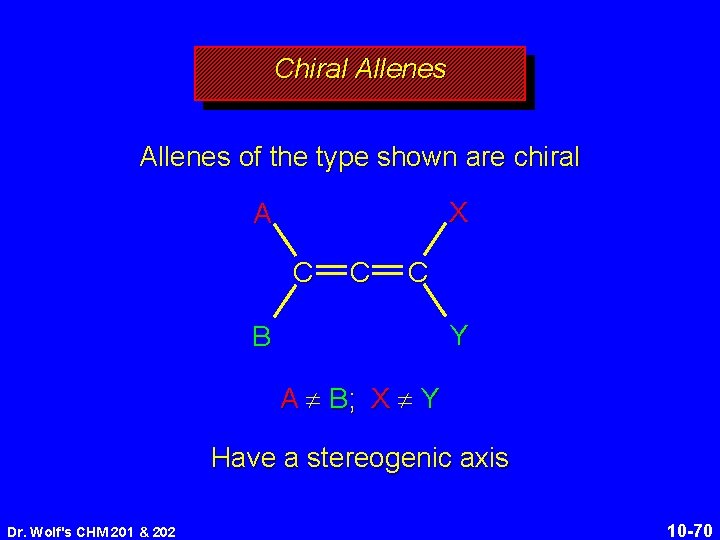
Chiral Allenes of the type shown are chiral X A C C C Y B A ¹ B; X ¹ Y Have a stereogenic axis Dr. Wolf's CHM 201 & 202 10 -70

Stereogenic Axis analogous to difference between: a screw with a right-hand thread and one with a left-hand thread a right-handed helix and a left-handed helix Dr. Wolf's CHM 201 & 202 10 -71

10. 9 Preparation of Dienes Dr. Wolf's CHM 201 & 202 10 -72

1, 3 -Butadiene CH 3 CH 2 CH 3 590 -675°C chromiaalumina H 2 C CHCH CH 2 + 2 H 2 More than 4 billion pounds of 1, 3 -butadiene prepared by this method in U. S. each year used to prepare synthetic rubber (See "Diene Polymers" box) Dr. Wolf's CHM 201 & 202 10 -73
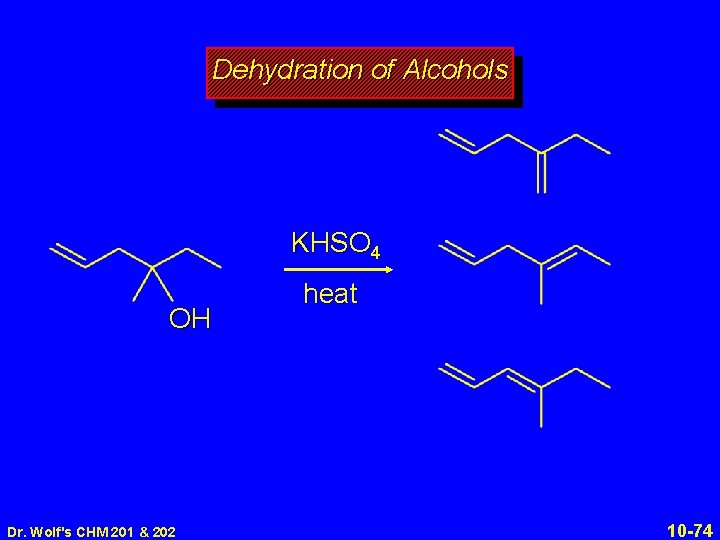
Dehydration of Alcohols KHSO 4 OH Dr. Wolf's CHM 201 & 202 heat 10 -74
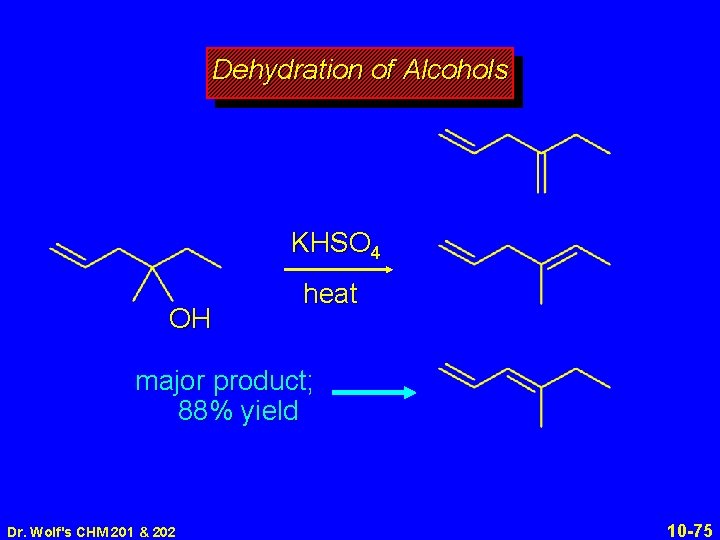
Dehydration of Alcohols KHSO 4 OH heat major product; 88% yield Dr. Wolf's CHM 201 & 202 10 -75
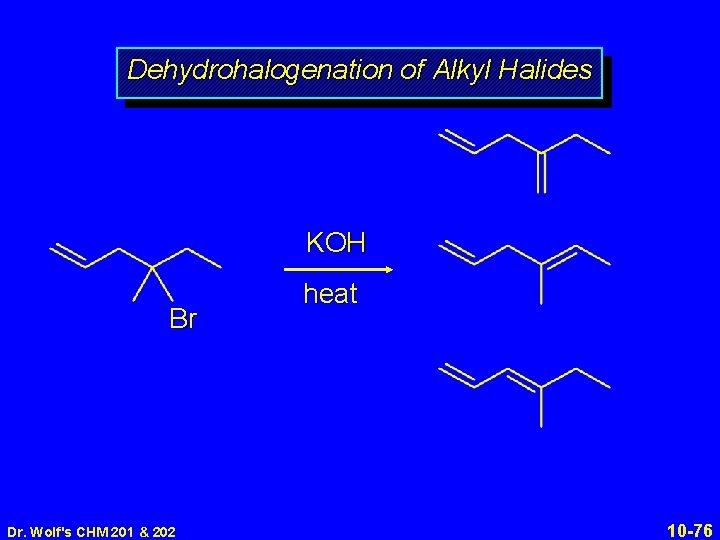
Dehydrohalogenation of Alkyl Halides KOH Br Dr. Wolf's CHM 201 & 202 heat 10 -76
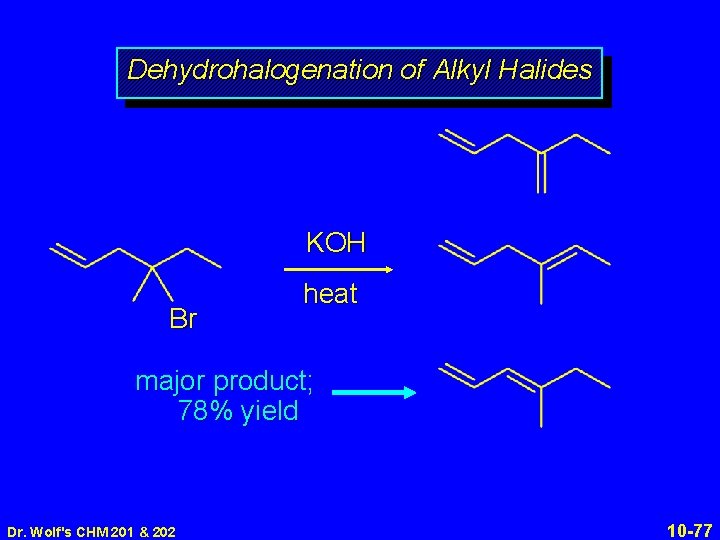
Dehydrohalogenation of Alkyl Halides KOH Br heat major product; 78% yield Dr. Wolf's CHM 201 & 202 10 -77

Reactions of Dienes isolated dienes: double bonds react independently of one another cumulated dienes: specialized topic conjugated dienes: reactivity pattern requires us to think of conjugated diene system as a functional group of its own Dr. Wolf's CHM 201 & 202 10 -78

Tipos de Reações Orgânicas • Reações de : – Adição – duas moléculas se combinam – Eliminação – uma molécula quebra em duas – Substituição – partes de duas moléculas trocam – Rearranjo – a molécula sofre mudanças no modo como seus átomos são conectados.

10. 10 Addition of Hydrogen Halides to Conjugated Dienes Dr. Wolf's CHM 201 & 202 10 -79
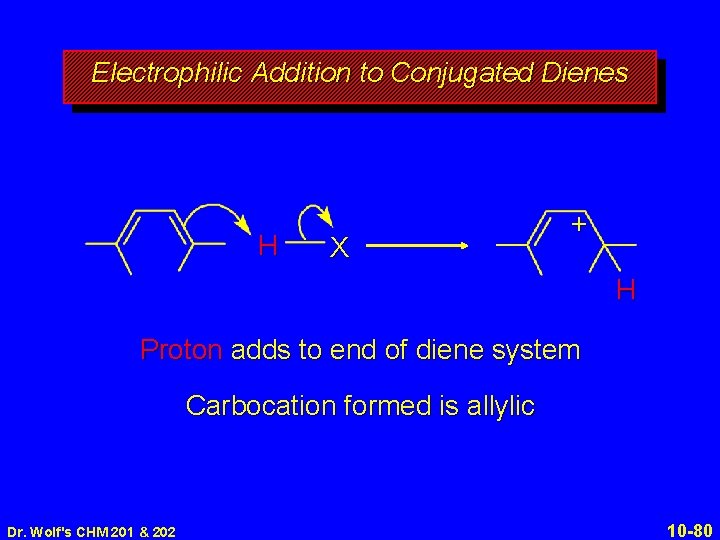
Electrophilic Addition to Conjugated Dienes H X + H Proton adds to end of diene system Carbocation formed is allylic Dr. Wolf's CHM 201 & 202 10 -80
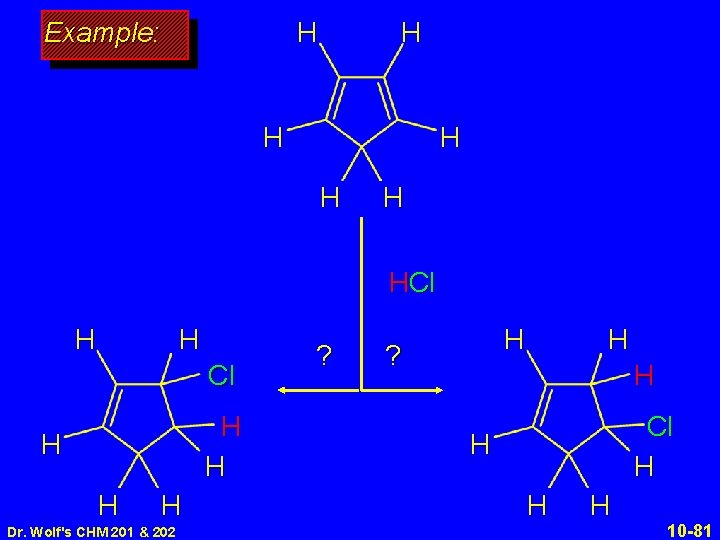
H Example: H H HCl H H H H H Dr. Wolf's CHM 201 & 202 ? H H Cl H H 10 -81
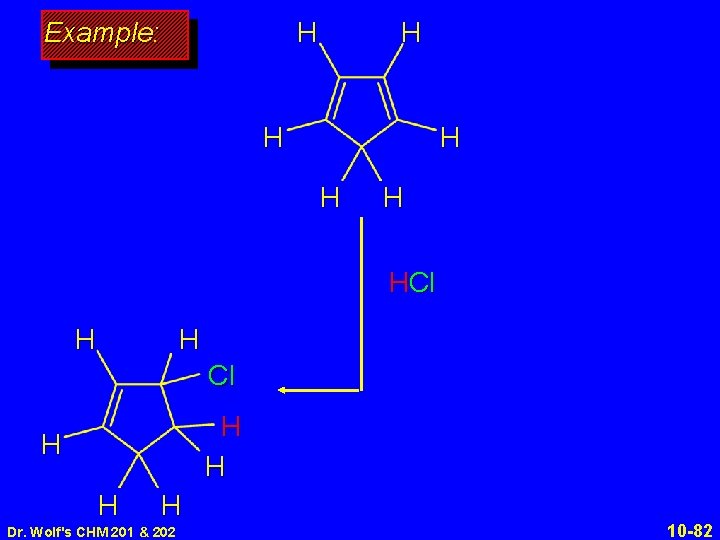
H Example: H H HCl H H H H H Dr. Wolf's CHM 201 & 202 10 -82
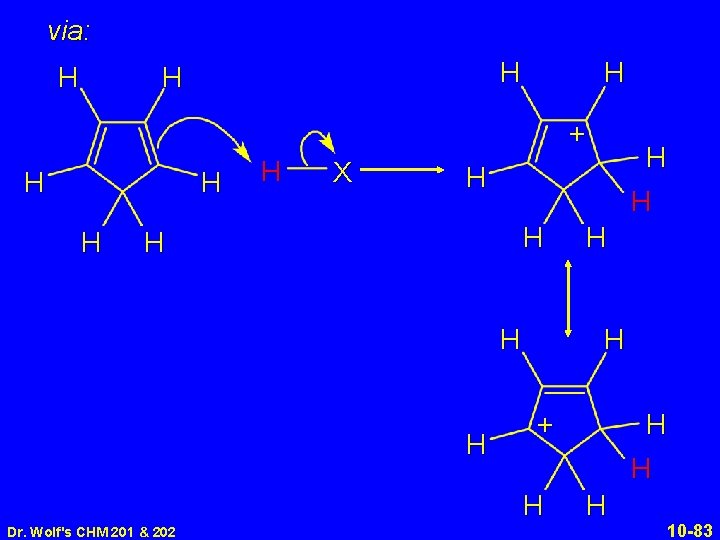
via: H H + H H X H H H H + H H H Dr. Wolf's CHM 201 & 202 H H 10 -83
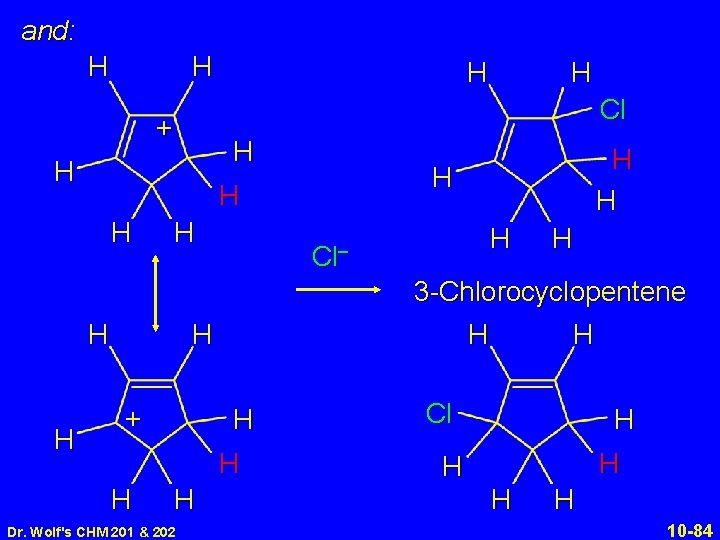
and: H H H H Dr. Wolf's CHM 201 & 202 H 3 -Chlorocyclopentene H H H Cl– H + H H H Cl + H H Cl H H H 10 -84
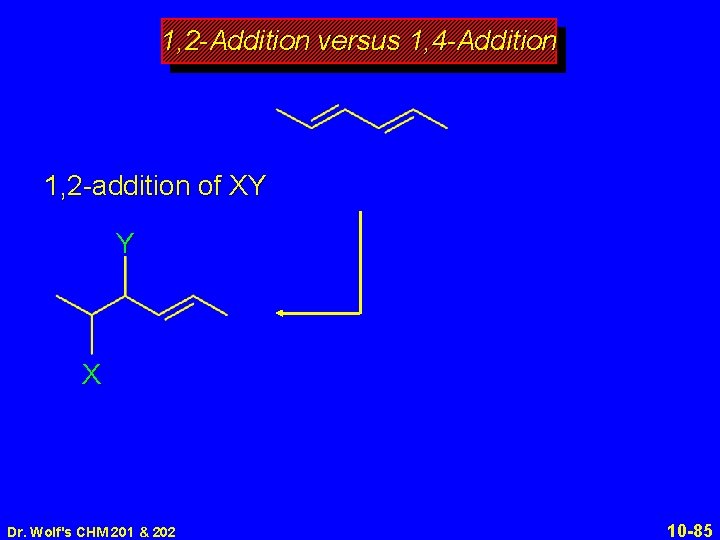
1, 2 -Addition versus 1, 4 -Addition 1, 2 -addition of XY Y X Dr. Wolf's CHM 201 & 202 10 -85
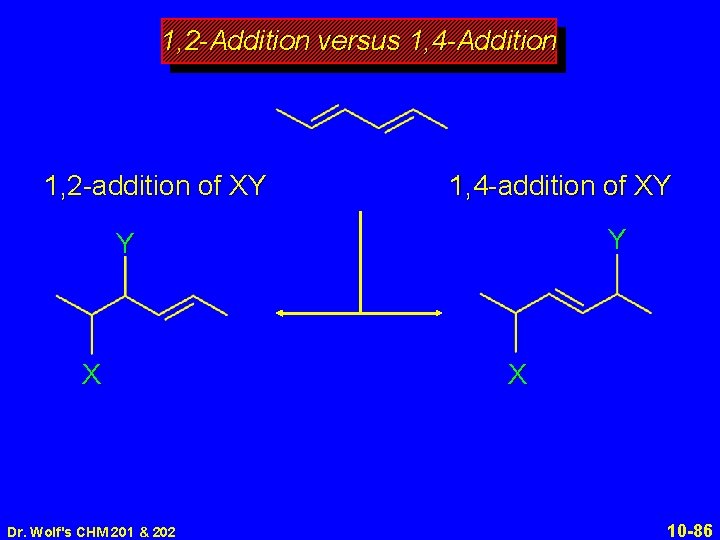
1, 2 -Addition versus 1, 4 -Addition 1, 2 -addition of XY 1, 4 -addition of XY Y Y X Dr. Wolf's CHM 201 & 202 X 10 -86
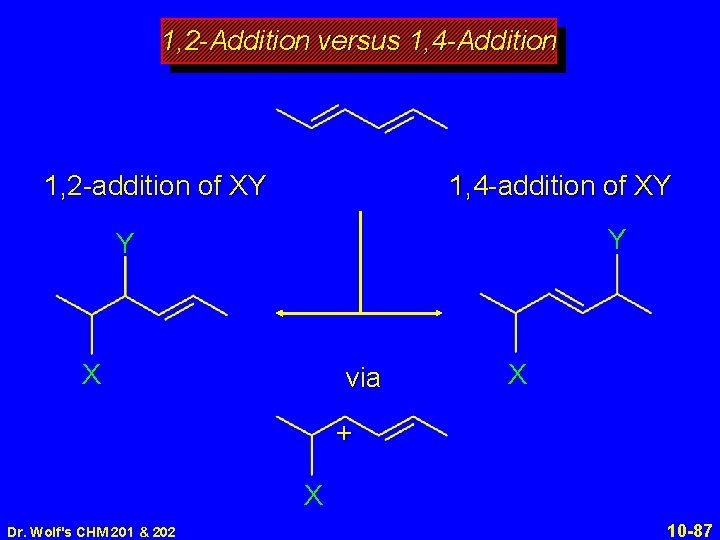
1, 2 -Addition versus 1, 4 -Addition 1, 2 -addition of XY 1, 4 -addition of XY Y Y X via X + X Dr. Wolf's CHM 201 & 202 10 -87
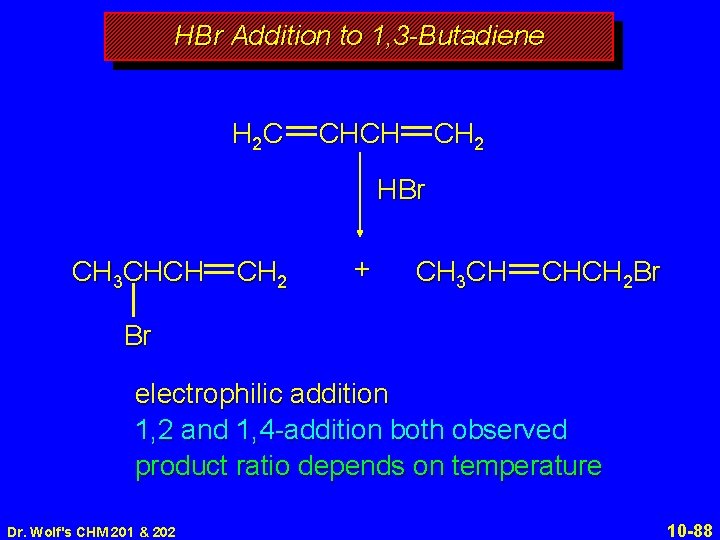
HBr Addition to 1, 3 -Butadiene H 2 C CHCH CH 2 HBr CH 3 CHCH CH 2 + CH 3 CH CHCH 2 Br Br electrophilic addition 1, 2 and 1, 4 -addition both observed product ratio depends on temperature Dr. Wolf's CHM 201 & 202 10 -88
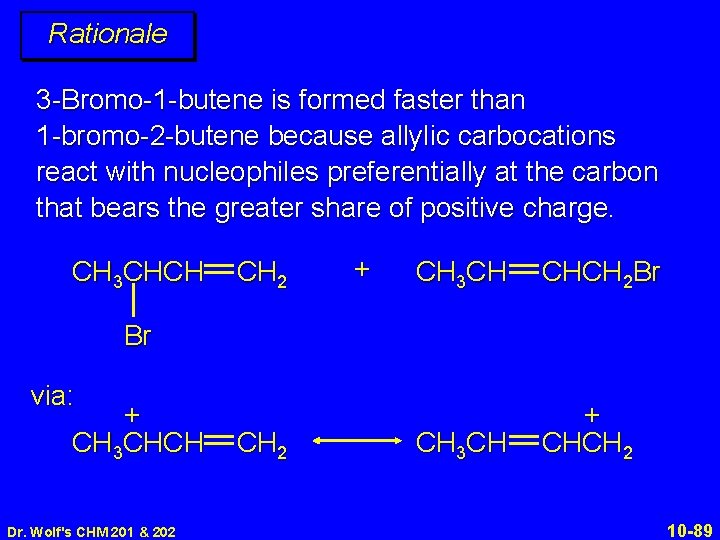
Rationale 3 -Bromo-1 -butene is formed faster than 1 -bromo-2 -butene because allylic carbocations react with nucleophiles preferentially at the carbon that bears the greater share of positive charge. CH 3 CHCH CH 2 + CH 3 CH CHCH 2 Br CH 3 CH + CHCH 2 Br via: + CH 3 CHCH Dr. Wolf's CHM 201 & 202 CH 2 10 -89

Rationale 3 -Bromo-1 -butene is formed faster than 1 -bromo-2 -butene because allylic carbocations react with nucleophiles preferentially at the carbon that bears the greater share of positive charge. CH 3 CHCH CH 2 + CH 3 CH CHCH 2 Br Br formed faster Dr. Wolf's CHM 201 & 202 10 -90
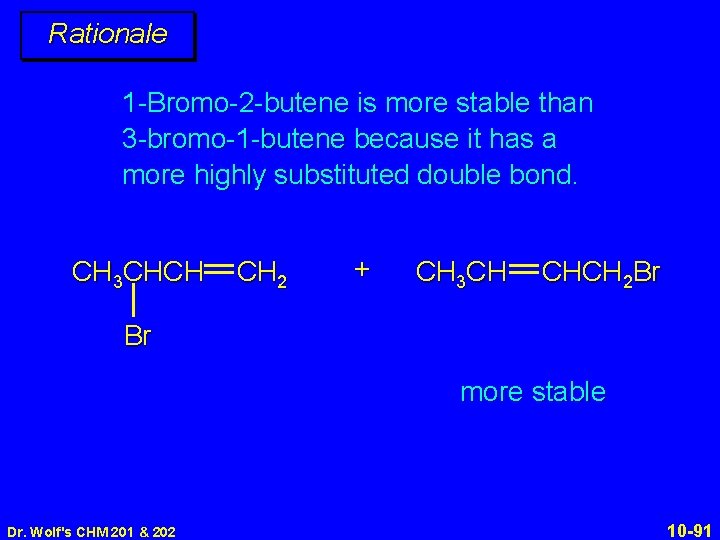
Rationale 1 -Bromo-2 -butene is more stable than 3 -bromo-1 -butene because it has a more highly substituted double bond. CH 3 CHCH CH 2 + CH 3 CH CHCH 2 Br Br more stable Dr. Wolf's CHM 201 & 202 10 -91

Rationale The two products equilibrate at 25°C. Once equilibrium is established, the more stable isomer predominates. CH 3 CHCH CH 2 Br major product at -80°C (formed faster) Dr. Wolf's CHM 201 & 202 CH 3 CH CHCH 2 Br major product at 25°C (more stable) 10 -92
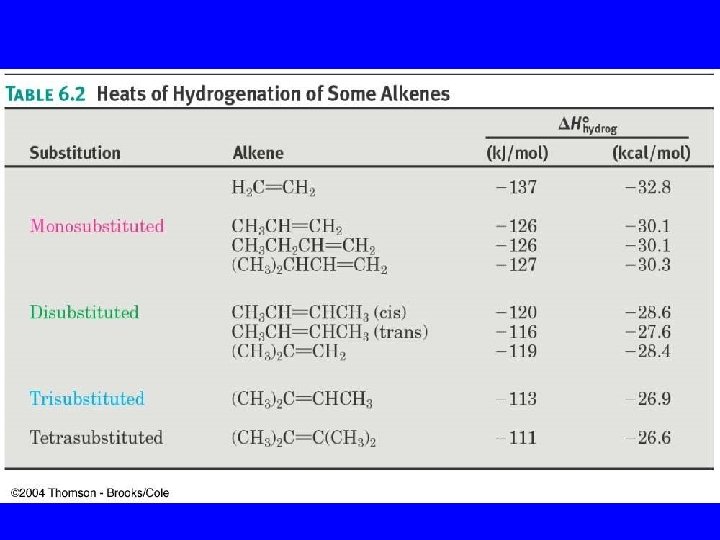
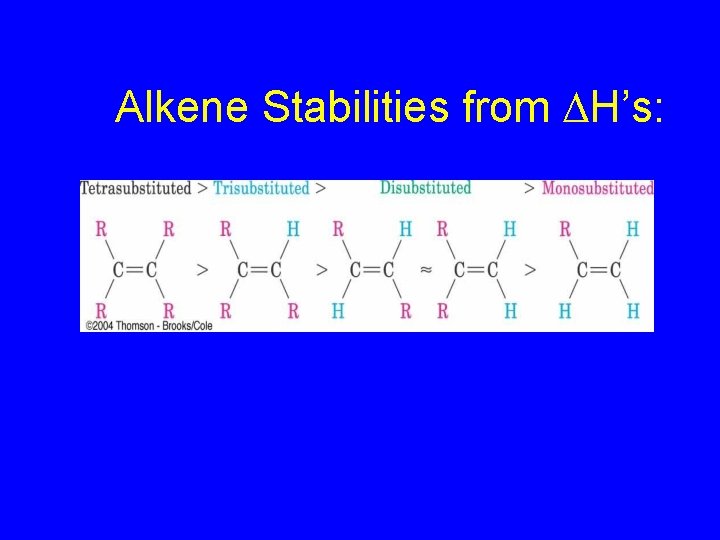
Alkene Stabilities from DH’s:
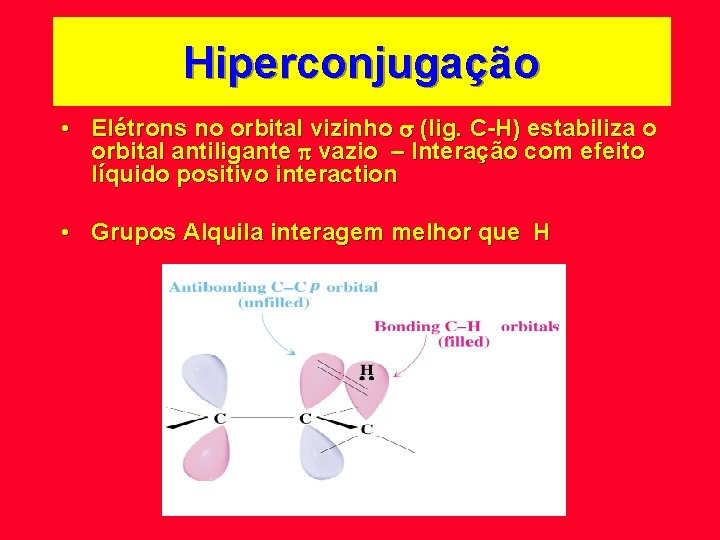
Hiperconjugação • Elétrons no orbital vizinho (lig. C-H) estabiliza o orbital antiligante vazio – Interação com efeito líquido positivo interaction • Grupos Alquila interagem melhor que H

Kinetic Control versus Thermodynamic Control • Kinetic control: major product is the one formed at the fastest rate • Thermodynamic control: major product is the one that is the most stable Dr. Wolf's CHM 201 & 202 10 -93
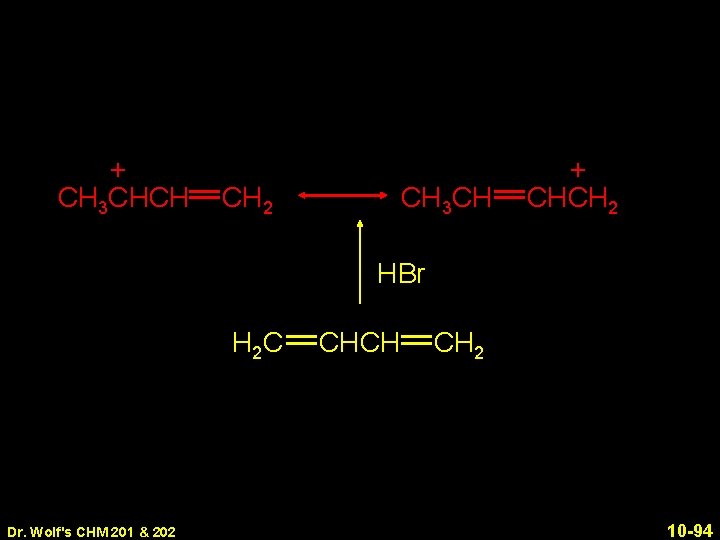
+ CH 3 CHCH CH 2 CH 3 CH + CHCH 2 HBr H 2 C Dr. Wolf's CHM 201 & 202 CHCH CH 2 10 -94
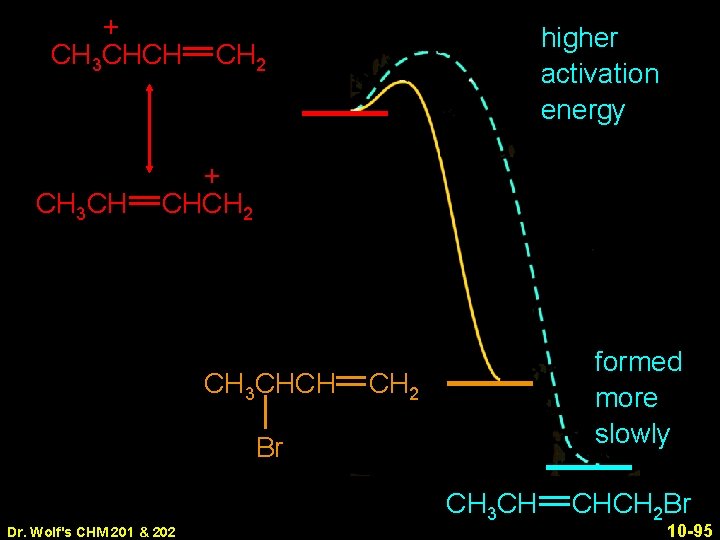
+ CH 3 CHCH CH 3 CH higher activation energy CH 2 + CHCH 2 CH 3 CHCH formed more slowly CH 2 Br Dr. Wolf's CHM 201 & 202 CH 3 CH CHCH 2 Br 10 -95
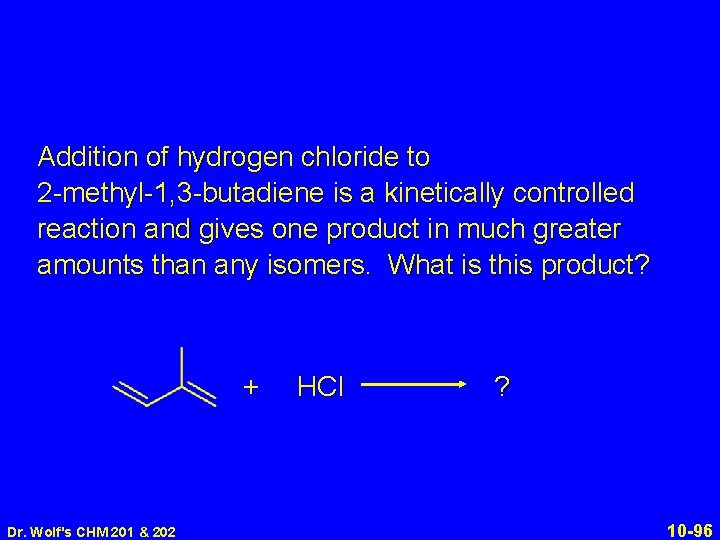
Addition of hydrogen chloride to 2 -methyl-1, 3 -butadiene is a kinetically controlled reaction and gives one product in much greater amounts than any isomers. What is this product? + Dr. Wolf's CHM 201 & 202 HCl ? 10 -96

Think mechanistically. + Protonation occurs: at end of diene system in direction that gives most stable carbocation HCl Kinetically controlled product corresponds to attack by chloride ion at carbon that has the greatest share of positive charge in the carbocation Dr. Wolf's CHM 201 & 202 10 -97
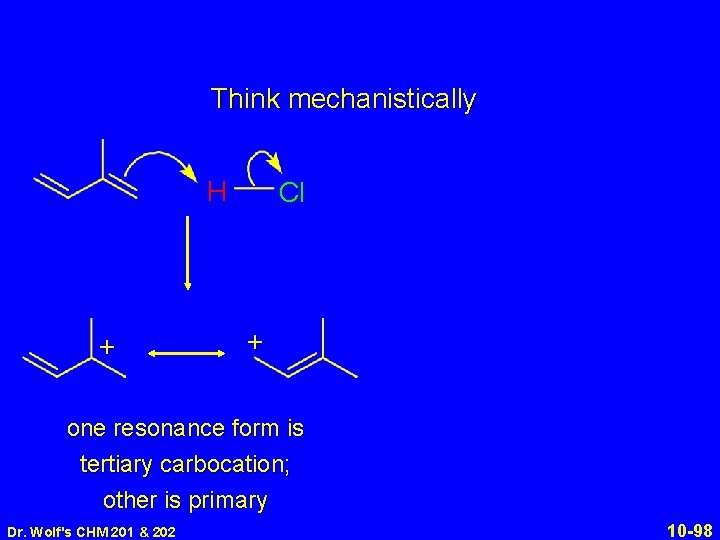
Think mechanistically H + Cl + one resonance form is tertiary carbocation; other is primary Dr. Wolf's CHM 201 & 202 10 -98
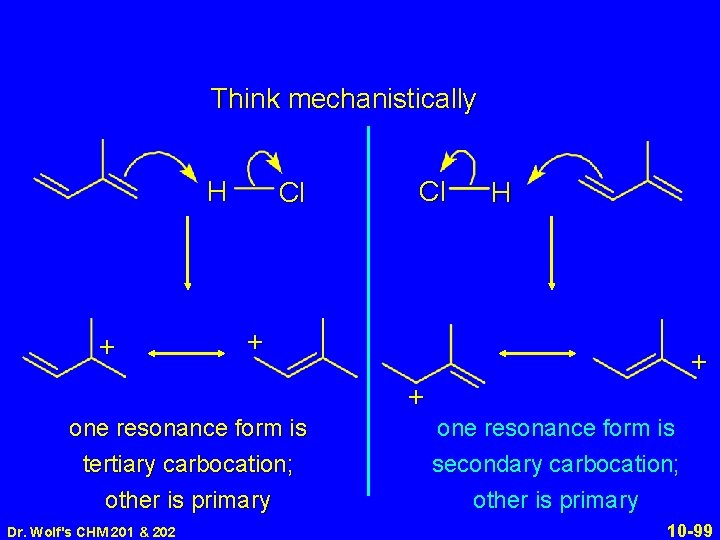
Think mechanistically H + Cl Cl H + + + one resonance form is tertiary carbocation; other is primary Dr. Wolf's CHM 201 & 202 one resonance form is secondary carbocation; other is primary 10 -99
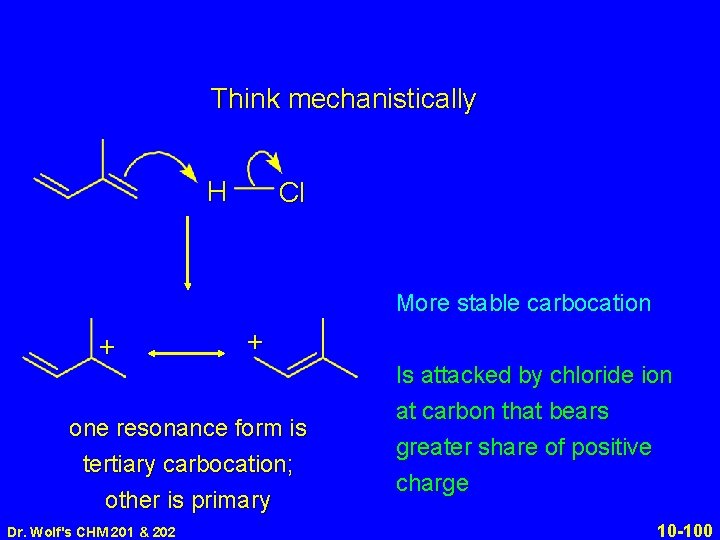
Think mechanistically H Cl More stable carbocation + + one resonance form is tertiary carbocation; other is primary Dr. Wolf's CHM 201 & 202 Is attacked by chloride ion at carbon that bears greater share of positive charge 10 -100
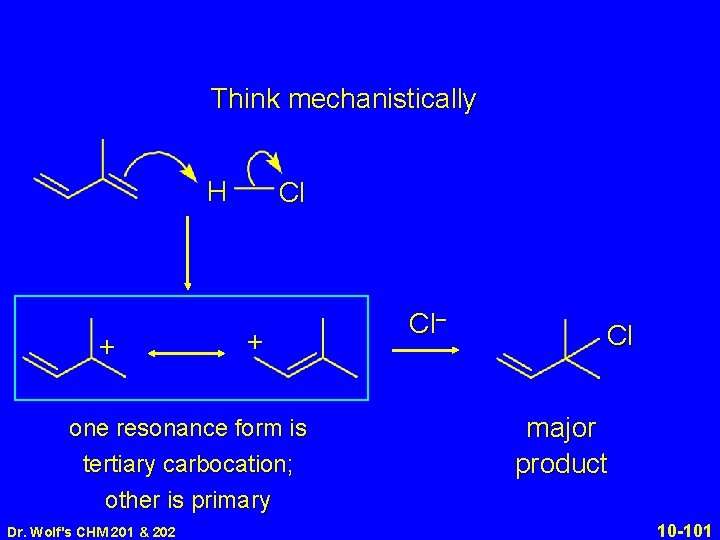
Think mechanistically H + Cl + one resonance form is tertiary carbocation; other is primary Dr. Wolf's CHM 201 & 202 Cl– Cl major product 10 -101

Terpenos e Terpenóides Regra do isopreno - Wallach (1887) O nome terpeno deriva da terebentina (turpentine) de onde foram isolados a cânfora e o a-pineno. As estruturas foram elucidadas em torno de 1894.

Borracha Natural

Borracha Natural • Gutta percha – History • Palaquium gutta trees from the Malay peninsula • (1843) Gutta percha was used to make knife handles – Properties • • trans-1, 4 -polyisoprene Softened in hot water and then pressed into desired shape Solid at room temperature and is ductile and strong At higher temperature it can be drawn out into strips with no recoil like rubber • Highly inert and resists vulcanization – Applications • Excellent insulator for Transatlantic Cables (Used until the 1930 s)

Natural rubber • Natural rubber consists of 1, 4 -cispolyisoprene, extracted from the rubber tree (Hevea brasiliensis). • It is produced in the tree by the biocatalyst hydroxynitrilelyase (2 hydroxyisobutyronitrile acetonelyase). • Synthetic rubber accounts for 75 % of rubber usage. However, natural rubber has advantages of elasticity, resilience and thermal properties. • Natural rubber is easily broken down in the environment, however vulcanisation (treatment with sulfur) renders it resistant to biodegradation.

Tomates Licopeno §Pigmento vermelho dos tomates §Polieno Conjugado §Antioxidante, neutraliza radicais Livres

Sunscreen Octyl methoxycinnamate §Conjugated cinnamate ester §Found in Coppertone Sport, Bullfrog Sunblock, Hawaiian Tropic Water Sport §Absorbs radiation, preventing skin damage
- Slides: 67