Essentials of Human Anatomy Physiology Chapter 2 Biochemistry

Essentials of Human Anatomy & Physiology Chapter 2 Biochemistry Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings

Composition of the Body • Elements • 96% of the body is made from six elements • • • Carbon (C) Oxygen (O) Hydrogen (H) Nitrogen (N) Phosphorus (P) Sulfur (S) Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 2

Important Inorganic Chemicals • Water • Most abundant inorganic compound • High heat capacity- does not change temperature easily • Chemical reactivity- serves as a base for reactions • Cushioning • Expands when it freezes • p. H of 7 (neutral) Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 2

Important Inorganic Chemicals • Salts- Vital to many body functions • Include electrolytes which conduct electrical currents • ex. - Na. Cl = sodium chloride, Ca 3(PO 4)2= calcium phosphate Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 2

Acids • Low p. H (below 7), Taste sour • Common Foods- lemon juice, coffee, carbonic acid in soda • In body- Gastric juice, uric acid, vitamin C, amino acids, nucleic acids, lactic acid, fatty acids, vaginal fluids Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 2

Bases • High p. H (above 7), Taste bitter, Slippery • Common Foods- Tums, baking soda, egg whites • Common products: Draino, bleach, oven cleaner • In body- blood, semen Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 2

Important Inorganic Compounds • Neutralization reaction • Acids and bases react to form water and a salt HCl + Na. OH -> H 2 O + Na. Cl Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 24
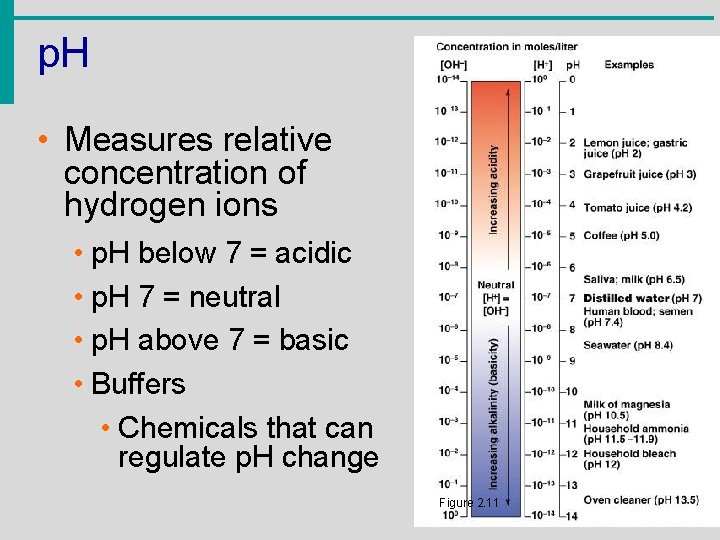
p. H • Measures relative concentration of hydrogen ions • p. H below 7 = acidic • p. H 7 = neutral • p. H above 7 = basic • Buffers • Chemicals that can regulate p. H change Figure 2. 11

Important Organic Compounds • Carbohydrates • Contain carbon, hydrogen, and oxygen in roughly a 1: 2: 1 ratio • Include sugars and starches • Classified according to size Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 26

Carbohydrates • Found in plants • Functions: • Used by all cells for energy • Our body’s primary source of energy Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 26
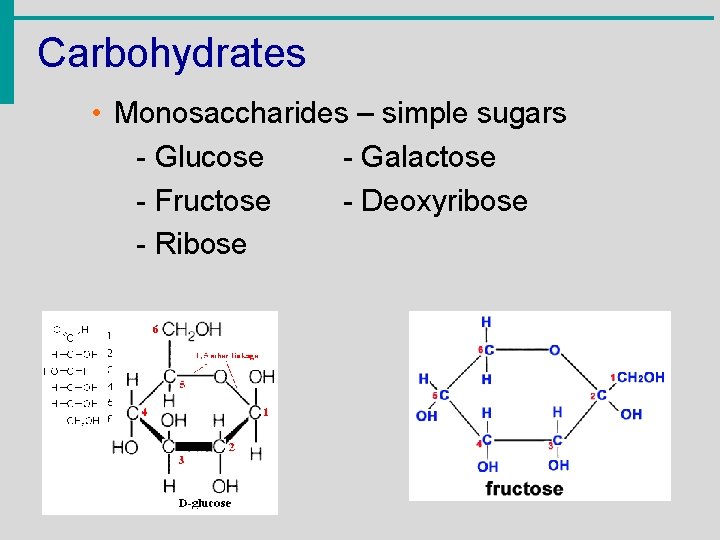
Carbohydrates • Monosaccharides – simple sugars - Glucose - Galactose - Fructose - Deoxyribose - Ribose
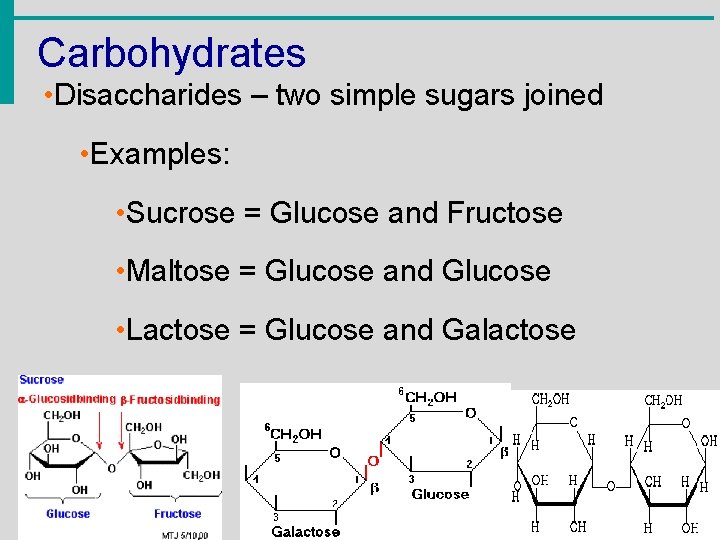
Carbohydrates • Disaccharides – two simple sugars joined • Examples: • Sucrose = Glucose and Fructose • Maltose = Glucose and Glucose • Lactose = Glucose and Galactose
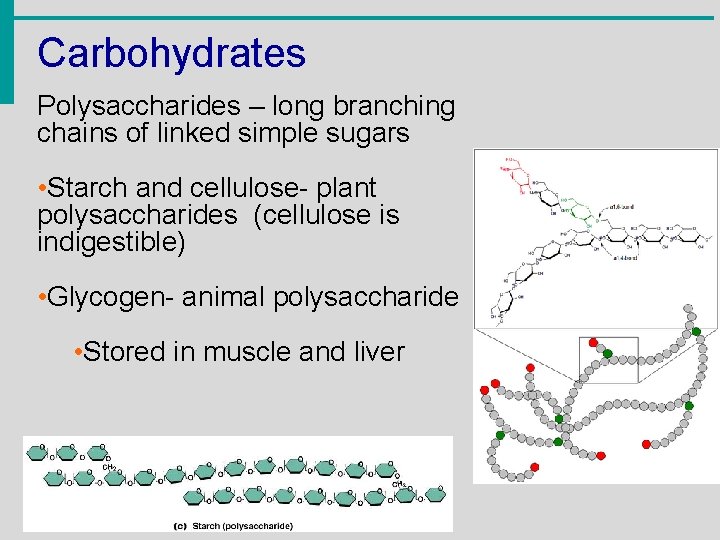
Carbohydrates Polysaccharides – long branching chains of linked simple sugars • Starch and cellulose- plant polysaccharides (cellulose is indigestible) • Glycogen- animal polysaccharide • Stored in muscle and liver

Lipids • Contain carbon, hydrogen, and oxygen • Monomers are fatty acids and glycerol • Most are insoluble in water Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 29

Lipids • Can be found in oils and waxes • Sometimes referred to as “fats” • Functions: • Energy storage • To cushion organs • Insulation • Part of the plasma membrane Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 29
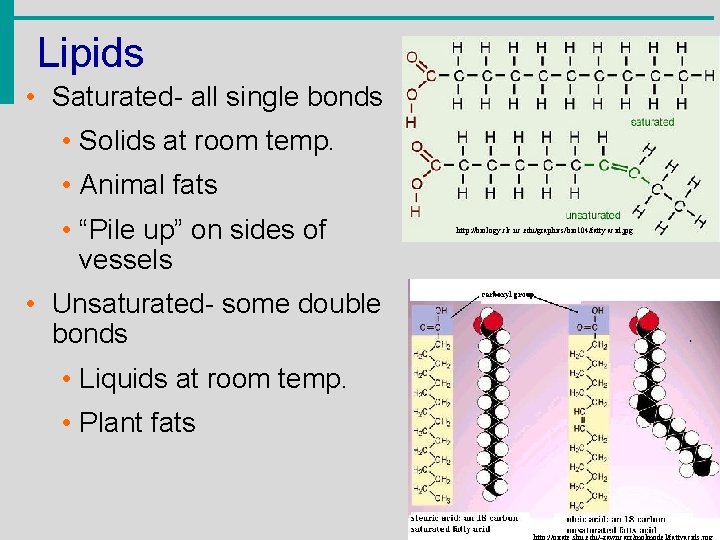
Lipids • Saturated- all single bonds • Solids at room temp. • Animal fats • “Pile up” on sides of vessels • Unsaturated- some double bonds • Liquids at room temp. • Plant fats http: //biology. clc. uc. edu/graphics/bio 104/fatty acid. jpg

Lipids • Common lipids in the human body • Neutral fats (triglycerides) • Found in fat deposits • Composed of 3 fatty acids and glycerol • Source of stored energy Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 30 a
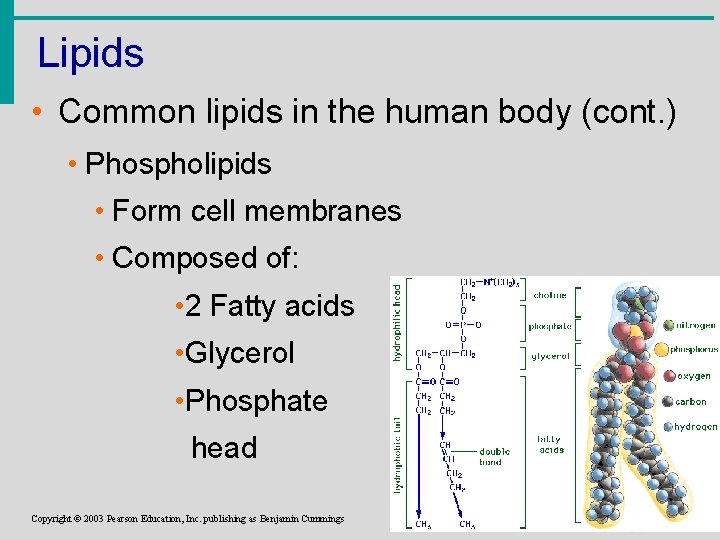
Lipids • Common lipids in the human body (cont. ) • Phospholipids • Form cell membranes • Composed of: • 2 Fatty acids • Glycerol • Phosphate head Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 30 b
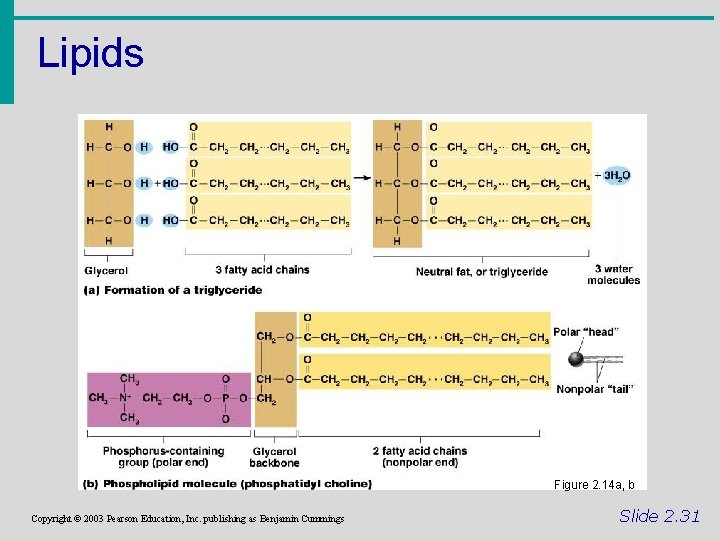
Lipids Figure 2. 14 a, b Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 31
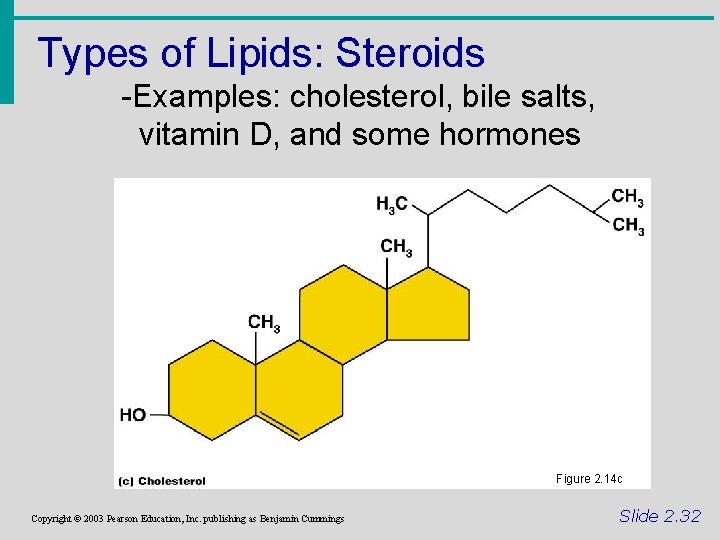
Types of Lipids: Steroids -Examples: cholesterol, bile salts, vitamin D, and some hormones Figure 2. 14 c Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 32

Proteins • Made of amino acids • 20 different amino acids • Contain carbon, oxygen, hydrogen, nitrogen, and sometimes sulfur Slide 2. 33 a

Proteins • Can be found in meat, animal products, and beans • Functions: • Maintains cell growth/formation of new body tissue • Helps build and repair body tissue • Aids in the contraction of muscle tissue • Aids in the formation of enzymes, some hormones, and antibodies • Provides energy if sufficient carbohydrates and fats are not supplied by diet Slide Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings 2. 33 b

Proteins • Essential amino acids- needed in diet • Non-essential amino acids- made in body • Account for over half of the body’s organic matter • Provides for construction materials for body tissues • Plays a vital role in cell function • Act as enzymes, hormones, and antibodies Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 33 b
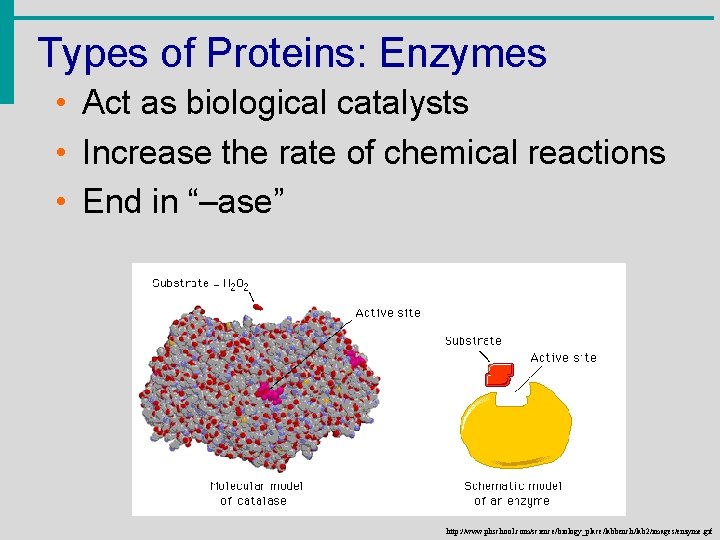
Types of Proteins: Enzymes • Act as biological catalysts • Increase the rate of chemical reactions • End in “–ase” http: //www. phschool. com/science/biology_place/labbench/lab 2/images/enzyme. gif
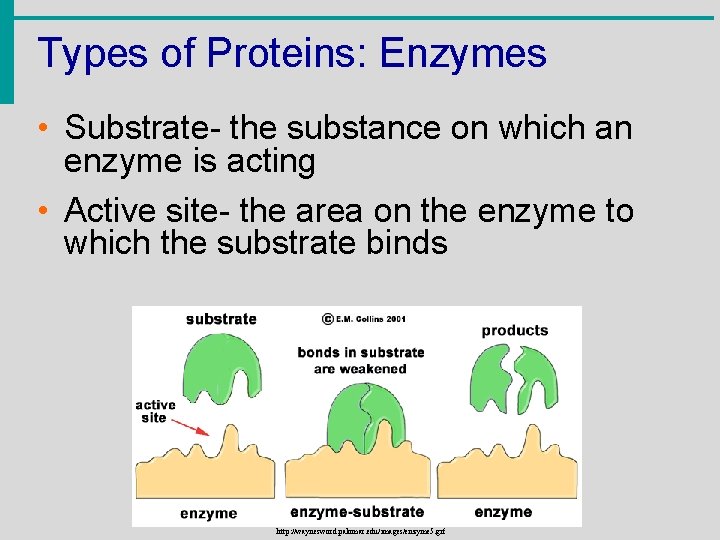
Types of Proteins: Enzymes • Substrate- the substance on which an enzyme is acting • Active site- the area on the enzyme to which the substrate binds http: //waynesword. palomar. edu/images/enzyme 5. gif

Types of Proteins: Enzymes • Things that denature (deactivate) enzymes: • Heat • p. H change • Addition of ionic substances

Nucleic Acids • Composed of nucleotides • Sugar (ribose or deoxyribose) • Phosphate • Nucleotide bases • A = Adenine • G = Guanine • C = Cytosine • T = Thymine (Only in DNA) • U = Uracil (Only in RNA) • A bonds to T (U), G to C
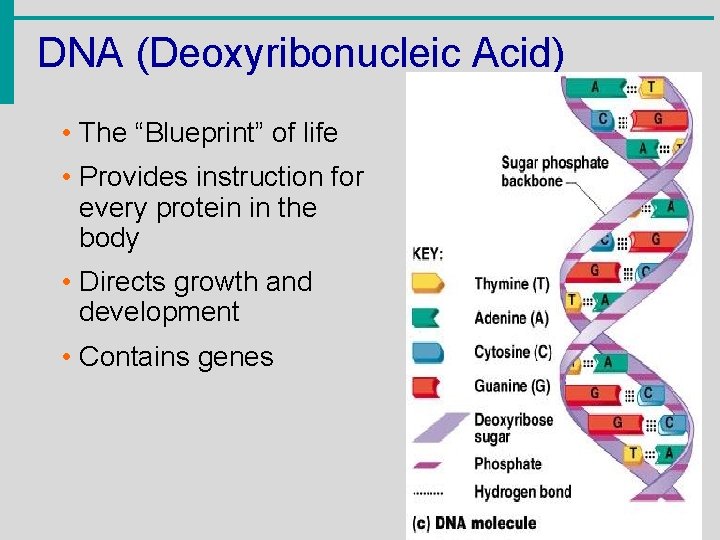
DNA (Deoxyribonucleic Acid) • The “Blueprint” of life • Provides instruction for every protein in the body • Directs growth and development • Contains genes
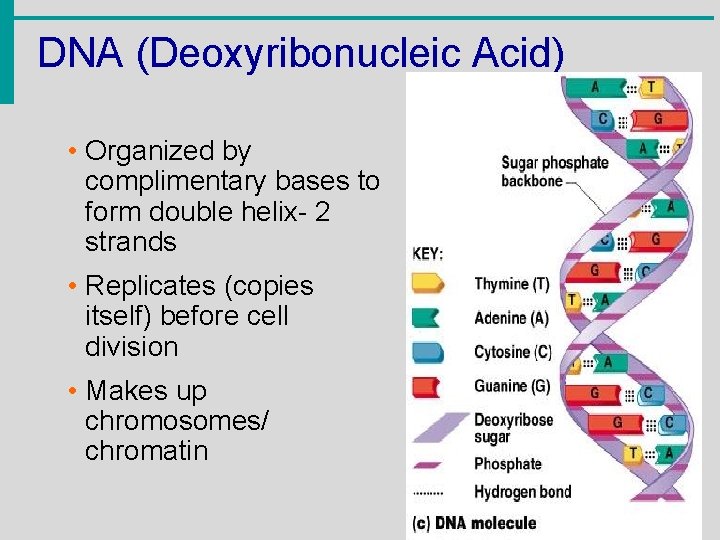
DNA (Deoxyribonucleic Acid) • Organized by complimentary bases to form double helix- 2 strands • Replicates (copies itself) before cell division • Makes up chromosomes/ chromatin

Adenosine Triphosphate (ATP) • Chemical energy used by all cells • Energy is released by breaking high energy phosphate bond • ATP is replenished by oxidation of food fuels Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Slide 2. 37
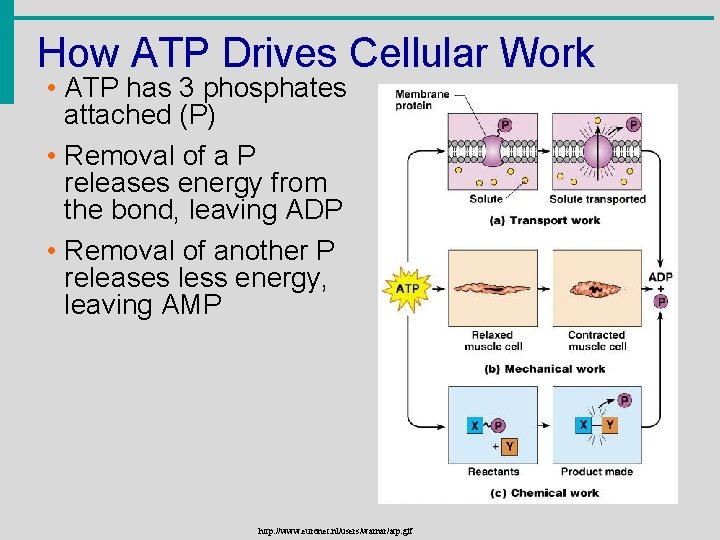
How ATP Drives Cellular Work • ATP has 3 phosphates attached (P) • Removal of a P releases energy from the bond, leaving ADP • Removal of another P releases less energy, leaving AMP http: //www. euronet. nl/users/warnar/atp. gif
- Slides: 31