ESSENTIAL TRACE ELEMENTS By Dr Rajesh Kumar Malik

ESSENTIAL TRACE ELEMENTS By Dr. Rajesh Kumar Malik Dept. of Chemistry MDU

ROLE OF ESSENTIAL AND TRACE METAL IS BIOLOGICAL PROCESS • The Role of essential in biological process this is all related to bio inorganic chemistry. This branch of chemistry explain the physiological and biochemical process of the metals and trace elements. • We know that our living body are made up of fifty many compounds such as protein, amino acids and they are possessing various elements. • About fifty to sixty occurring elements has been detected in living system. i. e. Animals, Human as well as Plants also Toxic elements

Role of Bulk or Structural Elements • H, C, N, O, P, S are the structural elements. • These elements are used by biological system beyond the trace levels. • Hydrogen and oxygen are the constituents of water. • Carbon and hydrogen are used in biosynthesis of all organic molecules and biogenetic carbonates. Nitrogen is essential for the biosynthesis of proteins, nuclei acids, etc. • Phosphorus is an important constituent of DNA, RNA, bones, teeth, some shells, membranes phospholipids, ADP and ATP and metabolic intermediates. • Sulphur is essential element in most proteins. It plays an important role in the tertiary structure (S – S linkage) of proteins. • Sulphur is present in vitamins and involved in fat metabolism and detoxification processes. H 2 SO 4 is known digestive fluid in ascidians (sea squirts).
![The Classification of Bio Inorganic elements [1] Essential elements: Na, K, Ca, Mg, P, The Classification of Bio Inorganic elements [1] Essential elements: Na, K, Ca, Mg, P,](http://slidetodoc.com/presentation_image_h2/5dc475cacf6155be586cdf6f29ec6844/image-4.jpg)
The Classification of Bio Inorganic elements [1] Essential elements: Na, K, Ca, Mg, P, S, Fe, Cl, Si They are also called macro elements. [2] Trace Elements: Mo, Mn, Fluorine, Iodine, Cu, Zn They are known as possible trace element. [3] Trance Ultra elements: Ni, Cr, Se, B, Sb, W (presence in very low quantity) Toxic elements : The presence of these elements arises bad effect on body and some time they caused typical diseases. Excess of these elements cause diseases: • As (18 mg), Sn (6. 0 mg), Bi (0. 20 mg) & Te (0. 1 mg), Cr+6(1, 5 mg) excess causes Cancer. • Pb (121 mg) causes Plambism & Cd (5. 0 mg) causes High B. P. • Sb (8. 0 mg) causes Poisoning Be 0. 04 mg Causes Hearts diseases

Essential trace elements : • These elements are universally required for the growth and survival of organisms. There is an impressive number of trace elements which normally exist and work in cells at very-very low concentration, generally far less than 1 µM and as low as 10 -8 – 10 -9 M; e. g. 50µg/d (for selenium). • There is considerable number of trace elements, mainly among the transition metal ions, which have been represented to serve as required growth factors at extremely low concentration. In most of the functions of trace elements focus on their role as metalloenzymes. • The most important fact about the trace elements to be essential is that they serve as required prosthetic groups in active site and/or as coenzymes. In the metalloenzymes, a fixed number of specific metal atoms (usually Fe, Zn, Cu, Mn, Co, Ni, etc. ) are firmly associated with protein. The combination of protein with essential trace element produces a unique catalytic function.

Copper (Cu) : Sources : Nuts, Fish, Cow, Milk, whole grains, beans, organ meats( liver, kidney) etc. Requirement: Adult 2. 5 mg/daily, Infant and children – 0. 5 mg. Function: • It is essential constituents of many proteins metallo-enzyme and some natural occurring pigments (skin of fruits & vegetables). • It is essential for maintaining the strength of skin, blood vessels , epithelial and connective tissues in the body. • Helps in Formation of pigment melanin in the skin. • Copper helps in synthesis of phospholipids. • It is present in cytochrome oxidase which is the final agent of electron transport from one component to other component

Deficiency : – 1 • Deficiency effect of copper has never been positively demonstrated but it has been suspected in case of spores (single cell species) or in nephrosis (kidney problem). • When copper level are low, the body may absorb less iron this can cause iron deficiency anemia , condition in which body is unable to carry enough oxygen to its tissues.

Wilsons disease (brown circle in eyes) • Autosomal recessive disorder • Mutation in ATP 7 b gene( this protein transport excess copper to bile where it is excreted in waste products) • Causes excessive accumulation of Cu Clinical features: symptoms are typically related to liver and brain • Diarrhea, vomiting, Cardiac & renal failure. • Hepatic necrosis( death of liver cells). • encephalopathy( term related to brain disease that alters brain functions. Treatment: • life long treatment, Avoidance of high Cu diet. • In early stages Zn may be effective as it competes with Cu for absorption • Penicillamine

Zinc (Zn): Sources : eggs, sea foods, milk, whole grains , oysters , red meat , poultry , baked beans , chickpeas , nuts, etc. Requirement: RDA: Adult – 15 mg/daily , Female: During pregnancy and lactation 10 -12 mg. Function: – • • zinc is present in cells throughout the body. It is essential for normal growth, sexual maturity & reproduction ability. It is needed for the body’s defensive ( immune ) system to properly work It plays a role in cell division , cell growth, and the breakdown of carbohydrates. It is essential for tissue repairing and wound healing & maintaining conc. of Vitamin-A It is essential components of enzymes present (about 70) in human body. Such as alcohol dehydrogenase alkaline, phosphatase carbonic anhydrase. The function of Insulin is also regulated with zinc.

• It behaves as Lewis acid in biochemical system. It is soft acceptor as compared to other divalent cations like Mg 2+, Ca 2+, Mn 2+. • Carbonic anhydrase is zinc containing enzyme which has vital role in respiration in animals. Effect of deficiency: It causes dwarfism (less height of person), hypogonadism (lac of sex hormones in males), loss of appetite (hungry) and taste, smell sensitivity and many metabolic disorders. 2

Cobalt (Co) : Source : fish , green leafy vegetables, liver, kidney , etc. Requirement : The average adult intake of cobalt is 5– 8 mcg per day. A safe Recommended Dietary Allowance (RDA) for cobalt hasn't been set yet. Foods high in vitamin B-12 are the only source of cobalt used by the body. Function : – • It is main constituent of vitamin B-12, which is necessary for normal RBC formation. • It is required for activation of various enzyme activity included methyl malonyl enzyme. • Co-enzyme A maltose oxido- reductase, homocysteine ribo-nucleotide reductase.

• Toxicity: It is very toxic to plants and moderately toxic to mammals when injected intravenously. • Effect of deficiency: The deficiency of cobalt in the soil adversely affects the health of grazing animals. So, it is required to add cobalt salts to soil (in case of Cobalt deficiency). • Effect of excessive intake: It damages kidney Polycythemia (increased RBC), malfunction of thyroid. • Toxic dose: > 500 mg per day 1

Iodine (I): • Sources : Iodine is found in traces in foods, common salt, sea food, iodized salt, fish (cod and tuna), milk , yogurt, etc. • RDA: Adult – 100 -150 µg/day. • Function : – Thyroid hormone synthesis and also have broad spectrum germicidal action • It is well require for the biosynthesis of thyroid Or Thyroxine Hormones such as T-3 (Tri-Iodothyronine, it is natural thyroid supplement) , T-4 = Tetra & T-5 = Penta Iodo-thyronine, which influence large number of metabolic functions.
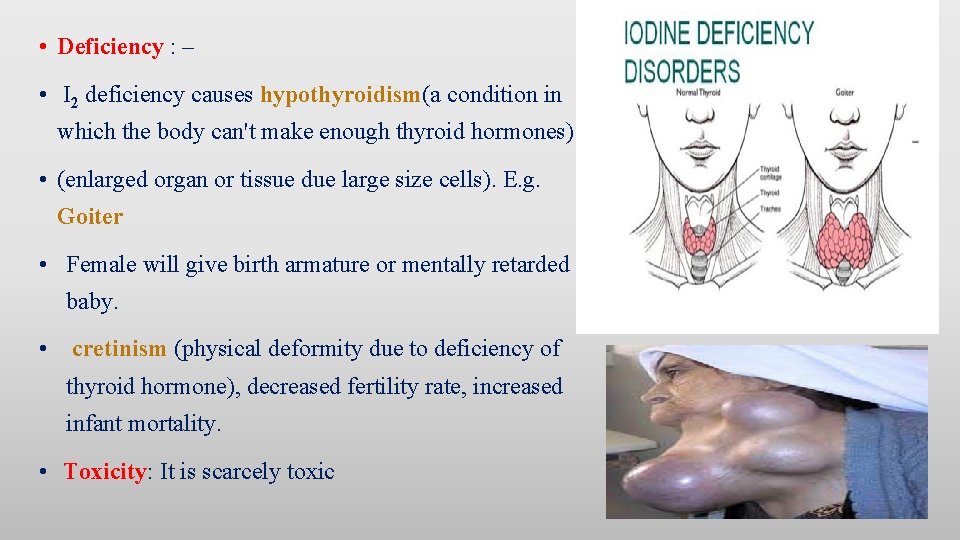
• Deficiency : – • I 2 deficiency causes hypothyroidism(a condition in which the body can't make enough thyroid hormones) • (enlarged organ or tissue due large size cells). E. g. Goiter • Female will give birth armature or mentally retarded baby. • cretinism (physical deformity due to deficiency of thyroid hormone), decreased fertility rate, increased infant mortality. • Toxicity: It is scarcely toxic

Fluorine (F) : • Sources : Fluoride tends to get concentrated in tea, coffee, shellfish, grapes (raisins, wine, grape juice), artificial sweeteners, sodas, potatoes, baby foods, and hot cereals made with tap water. • Requirements: It is Ultra-trace element. RDA: 2. 5 ppm • Functions : • Very important in preventing dental caries. • essential for the maintenance and solidification of our bones. • Promotes remineralization of decalcified enamel.
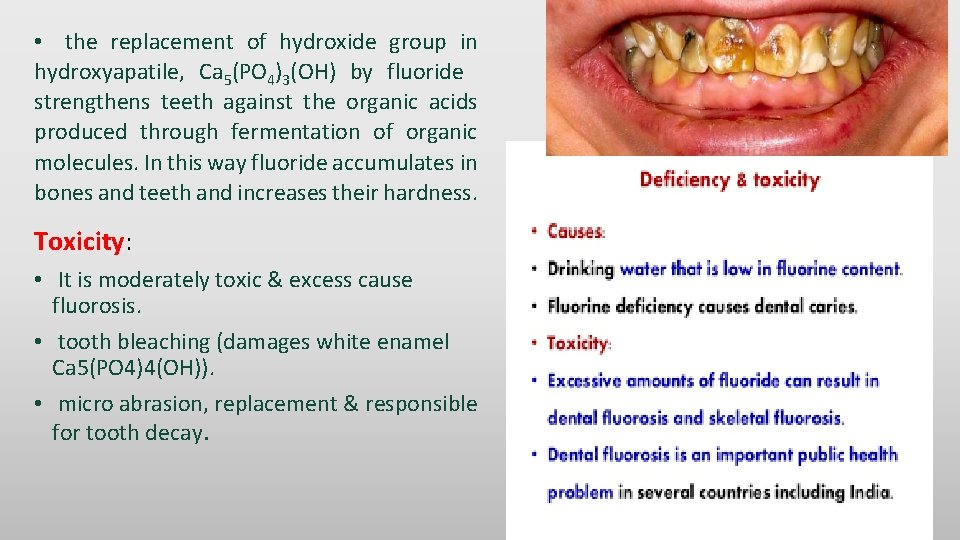
• the replacement of hydroxide group in hydroxyapatile, Ca 5(PO 4)3(OH) by fluoride strengthens teeth against the organic acids produced through fermentation of organic molecules. In this way fluoride accumulates in bones and teeth and increases their hardness. Toxicity: • It is moderately toxic & excess cause fluorosis. • tooth bleaching (damages white enamel Ca 5(PO 4)4(OH)). • micro abrasion, replacement & responsible for tooth decay.

Molybdenum (Mo) : • Sources : Legumes are the richest sources of molybdenum. Other foods high in molybdenum include whole grains, nuts, and beef liver, etc. • RDA: 45 mcg/day in pregnancy and lactation 50 mcg/day • Functions : • Act as a catalyst for enzymes and helps facilitate breakdown of certain amino acids. • Molybdenum in human tooth enamel may have a role in lowering the risk of tooth decay.

• It activates enzymes that help break down harmful sulfites and prevent toxins from building up in the body. • It is constituent of several enzymes which are used by nature for nitrogen fixation and nitrate reduction. Deficiency – esophageal carcinoma (cancer in the food pipe in body). Toxicity: It is moderately toxic. Excess of molybdenum in biological system may cause copper deficiency and is associated with gout like syndrome. Excess intake of Mo also causes impaired growth, anemia, diarrhea and de-pigmentation of hair.

Manganese (Mn) : • Sources : whole grains, clams, oysters, nuts, soybeans and other legumes, rice, leafy vegetables, coffee, tea, and many spices, such as black pepper. Drinking water also contains small amounts of manganese at concentrations of 1 to 100 mcg/L. • RDA: For male 3. 3 mg per day and for female 1. 8 mg per day. • Biological role: • It activates a number of enzymes & essential to all organisms. • It is required in the synthesis of cholesterol, mucopolysaccharides (long chain sugar molecules, found through body) and glycoproteins. • It is also essential for growth and reproduction. • Mn 2+ enzyme produced in the liver converts nitrogenous waste products into urea which is carried out by blood to the kidneys which excrete it out into the urine.
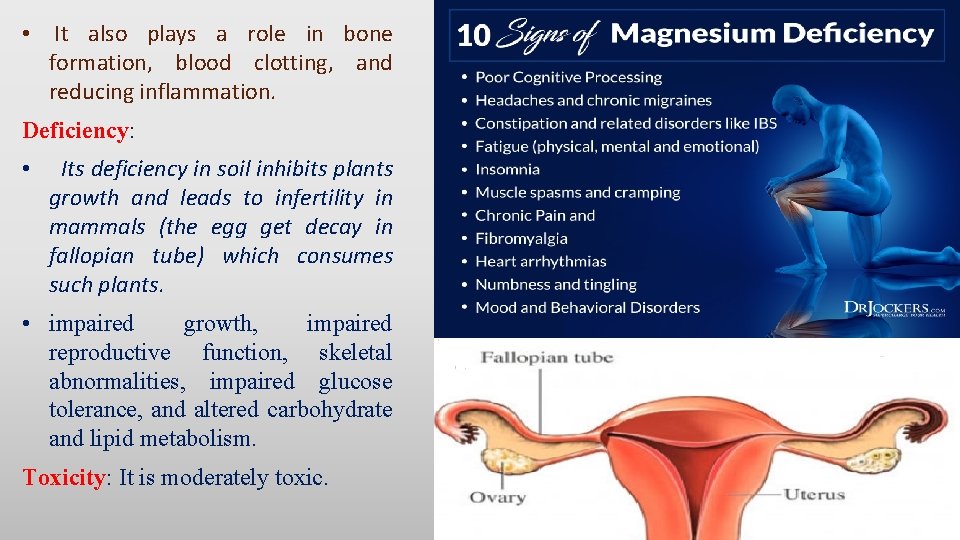
• It also plays a role in bone formation, blood clotting, and reducing inflammation. Deficiency: • Its deficiency in soil inhibits plants growth and leads to infertility in mammals (the egg get decay in fallopian tube) which consumes such plants. • impaired growth, impaired reproductive function, skeletal abnormalities, impaired glucose tolerance, and altered carbohydrate and lipid metabolism. Toxicity: It is moderately toxic.

Here are ten factors that affect and metabolism: Muscle mass. The amount of muscle tissue on your body. . Age. As you get older, your metabolic rate generally slows. . Body size. . Gender & Sexual factors. . . Genetics. . Physical activity. . Hormonal factors. . . Environmental factors… Contaminated water We will discuss it in another lecture

THANKS • References: • Bio-Inorganic By Dr. Neelam Kumari & Dr. R. K. Malik • Internet & Wikipedia • Bio-Inorganic By Dr. W. Kaim • Bio-Inorganic By Stephen’s J. Lippard
- Slides: 22