Essential Question How are elements compounds and mixtures













![Additional Review Activities [see resources] Vocabulary Matching Pairs Types of Matter Cube Review Additional Review Activities [see resources] Vocabulary Matching Pairs Types of Matter Cube Review](https://slidetodoc.com/presentation_image_h/291b788bc8f30eac69006e168bf22191/image-18.jpg)
- Slides: 18

Essential Question: How are elements, compounds, and mixtures related? S 8 P 1 b. Describe the difference between pure substances (elements and compounds) and mixtures

Matter is anything that has mass and takes up space (volume) There are different types of Matter: Pure Substances (elements and compounds) and Mixtures The composition (structure) of a substance determines its Matter type.

Characteristics of Pure Substances s i h t s e o d t a h W mean? • Fixed composition • Distinct properties • Cannot be separated into simpler substances by physical methods • Can only be changed in identity and properties by chemical methods • Properties do not vary one sample to another sample

Types of Pure Substances: Elements And Compounds

Elements • Made up of one type of atom • Cannot be broken down by physical and chemical methods • Examples: Oxygen, Nitrogen, Carbon Sample of the Element Lead Sample of the Element Chlorine

Compounds • Form when two or more different elements join (bond) together chemically • Composition is identical in each sample • Can be separated only by chemical methods • Properties of a compound are totally different than the properties of the elements that form them • Examples: Water, Carbon dioxide, Sugar Animated images and notes from http: //www. chem. purdue. edu/gchelp/atoms/elements. html

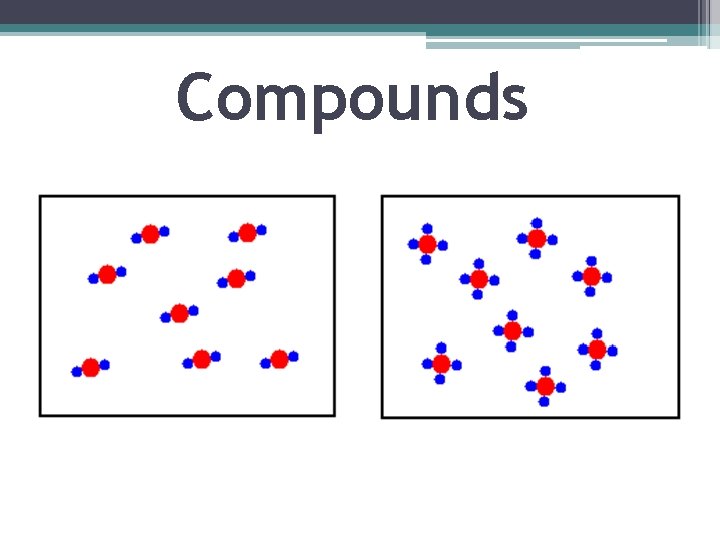
Compounds

Elements and Compounds Study Jams Video http: //studyjams. scholastic. com/studyjams/sc ience/matter/elements-and-compounds. htm

Mixtures • Form when elements and/or compounds are combined physically NOT chemically (no reactions between substances)

Mixtures are often referred to as homogeneous or heterogeneous.

Mixtures • Homogeneous mixtures (called Solutions) have a uniform distribution. • For example: Tea, Perfume, Air

Mixtures • Heterogeneous mixtures do not have a uniform distribution. • Parts are often visible • For example: Salad, Beach Sand, Oil and Vinegar dressing

Mixtures • Properties of a mixture are related to its components • Composition varies from sample to sample • Can be separated by physical methods • Examples of Mixtures: Tea, Perfume, Air, Salad, Beach sand, oil and vinegar salad dressing, etc.

Mixtures Study Jams Video http: //studyjams. scholastic. com/studyjam s/jams/science/matter/mixtures. htm

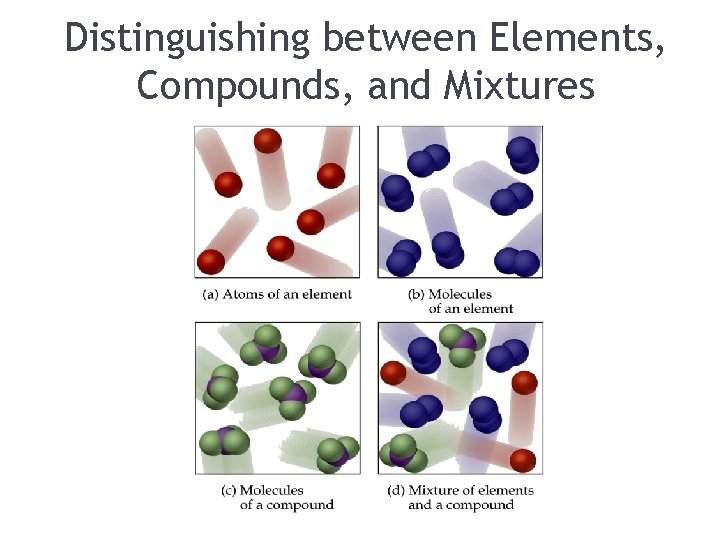
Distinguishing between Elements, Compounds, and Mixtures

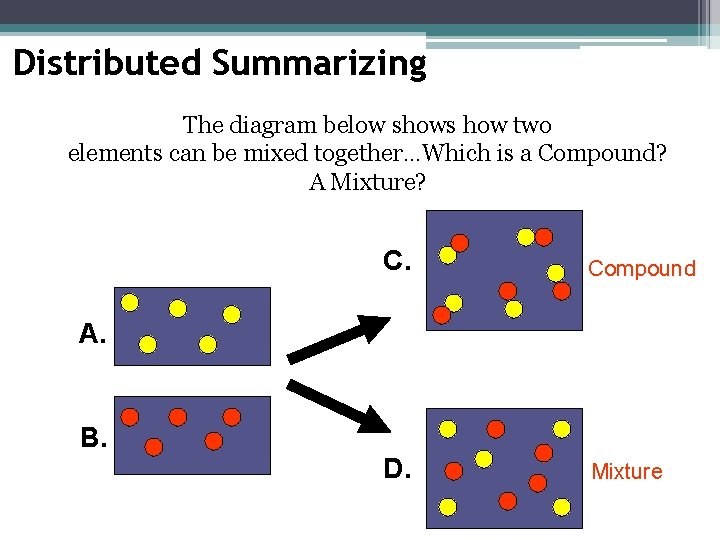
Distributed Summarizing The diagram below shows how two elements can be mixed together…Which is a Compound? A Mixture? C. Compound D. Mixture A. B.

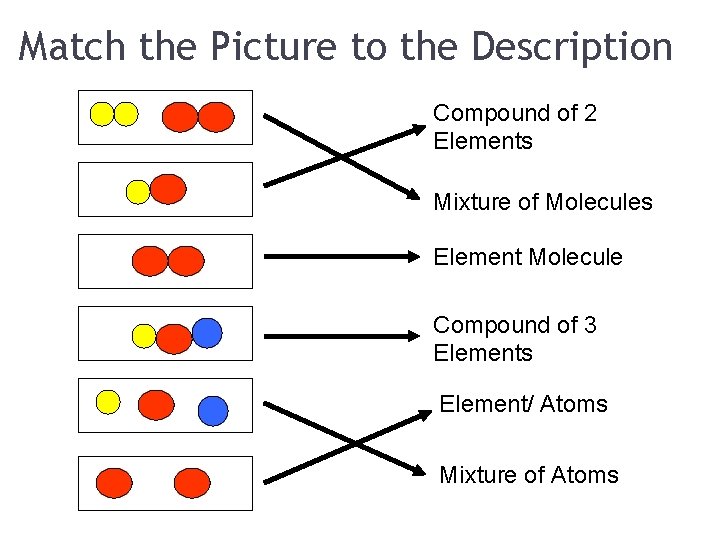
Match the Picture to the Description Compound of 2 Elements Mixture of Molecules Element Molecule Compound of 3 Elements Element/ Atoms Mixture of Atoms
![Additional Review Activities see resources Vocabulary Matching Pairs Types of Matter Cube Review Additional Review Activities [see resources] Vocabulary Matching Pairs Types of Matter Cube Review](https://slidetodoc.com/presentation_image_h/291b788bc8f30eac69006e168bf22191/image-18.jpg)
Additional Review Activities [see resources] Vocabulary Matching Pairs Types of Matter Cube Review