Essential Medicines Policies WHO Geneva Technical Briefing Seminar

Essential Medicines Policies, WHO, Geneva. Technical Briefing Seminar: 2011 Prequalification Programme: Priority Essential Medicines WHO-PQ INSPECTIONS Presented by Deus K Mubangizi Technical Officer mubangizid@who. int

WHO-PQP Inspections In this presentation: • Procedures and standards used for WHO-PQP inspections • WHO-PQ Inspections: avenues for capacity building and collaboration • Observed deficiencies during Inspection of: – FPP and API manufacturers + QC Labs – Contract Research Organizations (CROs) • Summary and conclusion 2
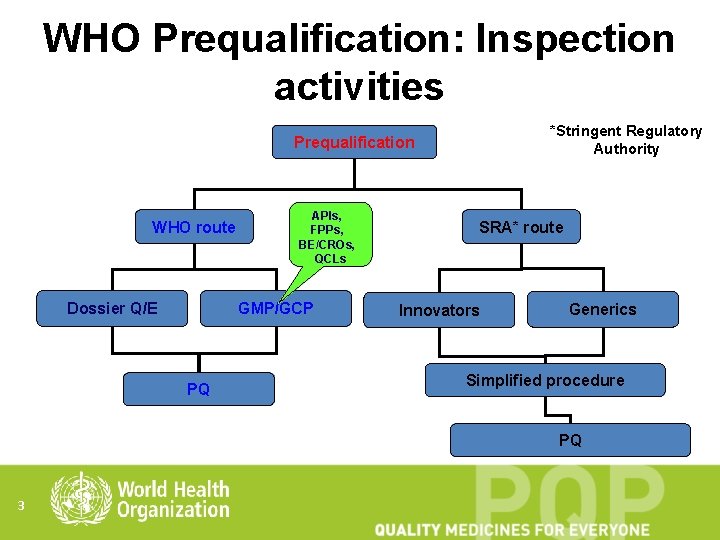
WHO Prequalification: Inspection activities *Stringent Regulatory Authority Prequalification WHO route Dossier Q/E APIs, FPPs, BE/CROs, QCLs GMP/GCP PQ SRA* route Innovators Generics Simplified procedure PQ 3

Prequalification Programme: Use of Inspection reports from other NMRAs è Inspectorates whose reports are recognized: √ PICS member inspectorates √ EU (EDQM + EMA) √ USFDA – new member of PICS è What GMP evidence to submit: – SMF – Up-to-date – Inspection report - conducted NMT 2 years • + CAPAs to deficiencies + final conclusion – Product Quality Review – not more than 1 year old è Review of the report: ü scope covered the specific API ü Is comprehensive and supports the final outcome. è PQP reserves the right to inspect the API manufacturer – as long as product is active in WHO-PQP. è on-going GMP compliance will be confirmed by WHO √ Desk review may only be use once in every 5 years. 4

Prequalification: Inspection Processes q By a team of qualified and experienced inspectors üWHO representative (qualified inspector) üInspector from well-established inspectorate (Pharmaceutical Inspection Cooperation Scheme countries – PIC/S) üNational inspector/s invited to be part and observe the inspection üObserver from recipient/developing countries (nominated by DRA of the country) q Scope: q Compliance with guidelines: q. GMP for API and FPP sites, q. GCP for CROs, q. GLP for FPP/API factory QCL, CRO-BAL, NQCL, IQCL èData verification – data manipulation, falsification, (validation, stability, clinical, bioanalytical) 5

Risk-based approach in: definition and classification of deficiencies • Deficiencies are descriptions of non-compliance with GMP requirements. • A distinction is made between deficiencies as a result of: – a defective system or, – failure to comply with the system. • Deficiencies may be classified as: – Critical Observation – potential risk harm to the user – Major Observation – major deviation from GMP/GCP – Minor or Other Observation – departure from good practice 6

Risk-based approach in: Conclusion following an inspection • When there are "other" observations only: – considered to be operating at an acceptable level of compliance with WHO GMP. – The manufacturer is expected to provide CAPAs. – CAPAs are evaluation and followed up during the next routine inspection. • When the are "other" and a few "major" observations: – compliance with WHO GMP/ICHQ 7 is made after the CAPAs have been assessed. – CAPAs for majors to include documented evidence of completion. – CAPAs paper evaluated ± an on-site follow up inspection. • When there are "critical" or several "major" observations: – considered to be operating at an unacceptable level of compliance with WHO GMP/ICHQ 7 guidelines. – Another inspection will be required 7

Information put in public domain - available for use by NMRAs: WHOPIRs and NOCs • These are published in response to the WHA Resolution WHA 57. 14 of 22 May 2004, which requested WHO, among other actions: – "3. (4) to ensure that the prequalification review process and the results of inspection and assessment reports of the listed products, aside from proprietary and confidential information, are made publicly available; " • A WHO Public Inspection Report (WHOPIR) reflects a positive outcome after an inspection • A Notice of Concern (NOC) is a letter reflecting areas of concern where the non-compliances require urgent attention and corrective action by the manufacturer or research organization. 8

Prequalification Programme: International norms, standards and guidelines used in inspection activities to ensure wide applicability USP BP Ph. Eur. Ph. Int. Other guidelines e. g. ICH, ISO http: //apps. who. int/prequal/assessment_inspect/info_inspection. htm#2 9

WHO-PQ offers new avenues for collaboration in inspection q WHO-PQ Collaborative Procedure in Inspections – nominated inspectors from NMRAs of selected member states are invited to participate in WHO-PQ organized inspections and in turn, the NMRAs is given appropriate access to outcomes of these inspections. • Capacity building of NMRAs inspectors. • Facilitating use of WHO-PQ inspection results in national regulatory environment for information and decision making. • Facilitation of harmonization through joint inspections and sharing of outcomes. • Share the workload and promote avoiding duplicative inspections. http: //apps. who. int/prequal/info_general/documents/inspection/NMRAs/GUIDANCE_WHOPQM_NMRAs_Collaborative. Procedure. pdf 10

Inspection observers have been from: �AFRO Region - External Observers �WPRO Region – Host country Observers
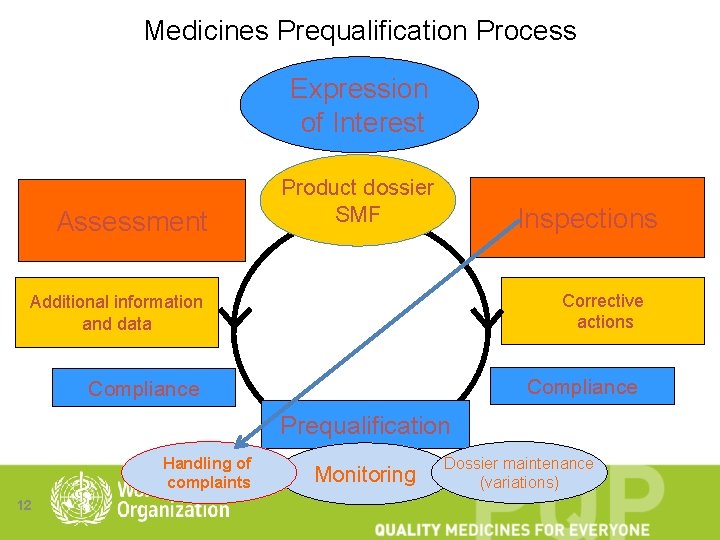
Medicines Prequalification Process Expression of Interest Assessment Product dossier SMF Inspections Corrective actions Additional information and data Compliance Prequalification Handling of complaints 12 Monitoring Dossier maintenance (variations)

Zidolam-N Zidovudine 300 mg, Lamivudine 150 mg and Nevirapine 200 mg tablets • Prequalified on 23 May 2006 with reference number HA 275 • Manufactured by Hetero at its Unit III, Andhra Pradesh, India 13
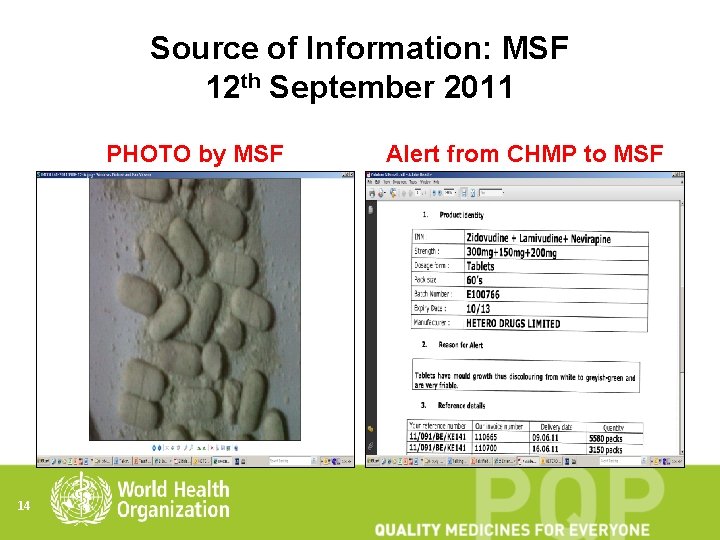
Source of Information: MSF 12 th September 2011 PHOTO by MSF 14 Alert from CHMP to MSF

Immediate Investigation 13 th September 2011 • Alerted Kenya Pharmacy and Poisons Board – Had not been notified by CHMP (Kenya) or MSF (Kenya) • Special Inspection of Hetero Unit 3: – – – 15 Retention samples Manufacturing records Analysis records Distribution records Observed re-analysis of retention samples
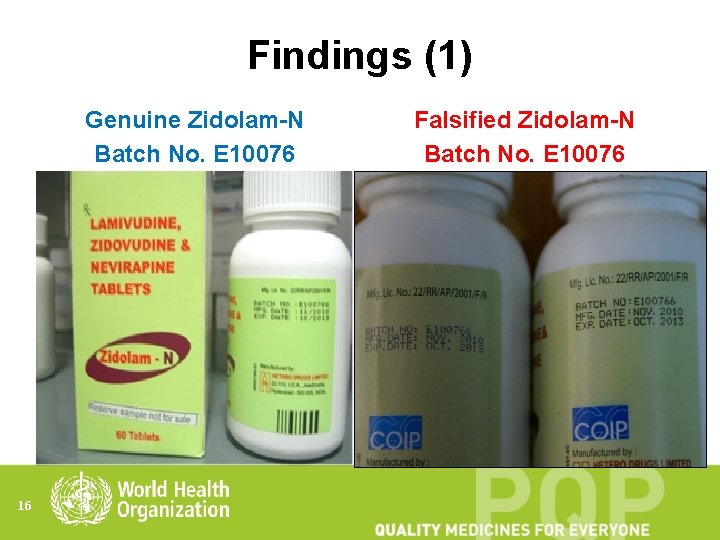
Findings (1) Genuine Zidolam-N Batch No. E 10076 16 Falsified Zidolam-N Batch No. E 10076

Findings (2) ZIDOLAM-N Batch No. E 100766 GENUINE FALSEFIED Font style of zero the batch number; is printed as 0. number; is printed as Ø. The spacing between licence number and the batch number on the bottle label is on the bottle label varies from constant in all samples. one bottle to another. The samples of the genuine Hetero batch do not carry a COIP logo. 17 Falsified samples bear the COIP logo. (The logo is that of a Canadian nongovernmental organization. )

Findings (3) • Affected batches; – E 100766, E 110467, A 9351, A 9357, A 9366 • the genuine batches E 100766 and E 110467 were never supplied to the Kenyan market • the quantities of Zidolam-N with a reference to “batch number A 9351, A 9357 or A 9366”, found in Kenya, exceed the quantities manufactured, packed and dispatched by Hetero as batches A 9351, A 9357 and A 9366 to Kenya. 18

Findings (4) • Results of analysis by Kenya’s NQCL and Hetero: – Intact samples comply with the manufacturers’ and with international pharmacopoeia specifications. – Open samples discoloured with high friability, low assay, fungal growth: Possible poor handling by patient. • Nature of falsification: – Relabeling and repackaging of donated batches (whose expiry dates are unknown) in order to divert them to the commercial market. – Extent and conditions under which the falsification (relabelling) was undertaken is unknown and thus the quality of the products cannot be fully ascertained 19

Actions taken • Collaborated with Kenya authorities in conducting investigation and taking necessary actions. • Special inspection of the manufacturing site. • Analysis of complaint and retention samples. • KPPB ordered a recall of the batches. • Issued public notices which were updated as more information became available – careful not to cause anxiety and fear. 20
21

Lessons for the future • Inadequate market controls and procurement procedures: – Diversion of donations, relabeling and repackaging. – Purchase from middlemen at very low prices without question. – NMRA not informed promptly. • PQ prompt reaction and coordination facilitated quick investigation. • Collaboration between PQ, MSF, KPPB, KNQCL and Hetero was crucial for the success of investigations. 22

Recommendation • Supply chain should be fully known and actions should be taken to shorten it wherever possible • A supply chain is no stronger than its weakest link 23

Inspection of FPP manufacturers 24

Deficiencies observed during PQ Inspections PREMISES – Inadequate segregation. – Illogical process flow. – Inadequate provision for Utilities: HVAC, water, compressed gases • Poor design and management of the HVAC system: – Multipurpose plant used re-circulated air but had no HEPA filters. – Adequate pressure differentials: reversal of air flow. – No sequence of switching on and off of AHUs of adjacent areas. 25 Mi x -u p s C o n ta m i n a ti o n C r o s s c o n ta m i n a ti o n • Poor design and construction of premises:

Deficiencies observed during PQ Inspections MATERIALS • Inadequate goods and materials management – – – – 26 Starting materials: sourcing and sampling – ID per container. Packaging materials: inadequate sampling – ISO 2859 or BS 6001. Intermediate and bulk products – holding time not set, or justified, or respected. Finished products: Release procedures – no adequate review by QA or QP. Rejected materials and products: not adequate segregation or disposal. Reagents and culture media: no GPT, positive and negative control Reference Standards: inadequate standardisation, storage and use.
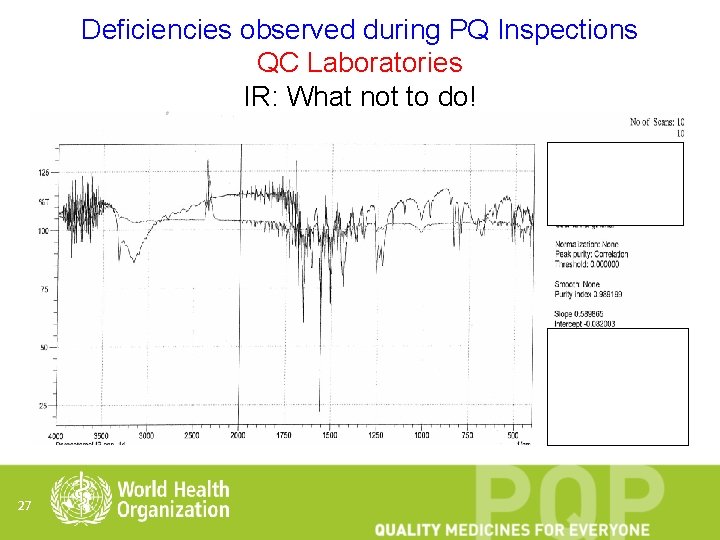
Deficiencies observed during PQ Inspections QC Laboratories IR: What not to do! 27

Deficiencies observed during PQ Inspections QUALITY CONTROL: Microbiology • Media Preparation: – No positive and negative control – No separate room for media preparation • Equipment: – No separate autoclave for sterilization of media and decontamination of used media. • Environmental Monitoring: – Inadequate exposure of plate method, adequate air sampling or swabbing. • Validation of Sanitising Agents: no challenge tests using the standard stock cultures (103 -104/0. 1 ml) in order to ascertain the minimum dilutions to effect a kill. 28

Deficiencies observed during PQ Inspections FPP manufacturers: – sterile products Poor aseptic techniques: • Extensive movement of operators in Grade A close to the open vials. • Vials that were not stoppered by the machine taken from the conveyor belt into class B area and manually placed back into class A to be manually stoppered. • Inadequate Media Fill Tests 29 Environmental monitoring: • The viable particle continuous monitoring for Grade A zone does not cover the whole time period of setting up of the equipment and the vial fillingcapping process. • Personnel garments and gloves were not monitored after manufacturing operations in grade A/B areas

Inspection of API manufacturers 30

Out of 126 API sites participating in PQ activities, 49 were accepted based on approval by PICS inspectorates and/or ICH countries while 31 were inspected. 31

The sites inspected were the ones producing APIs used in most FPPs (average each API site representing 21 FPPs). Thus maximizing use of available inspection resources. 32
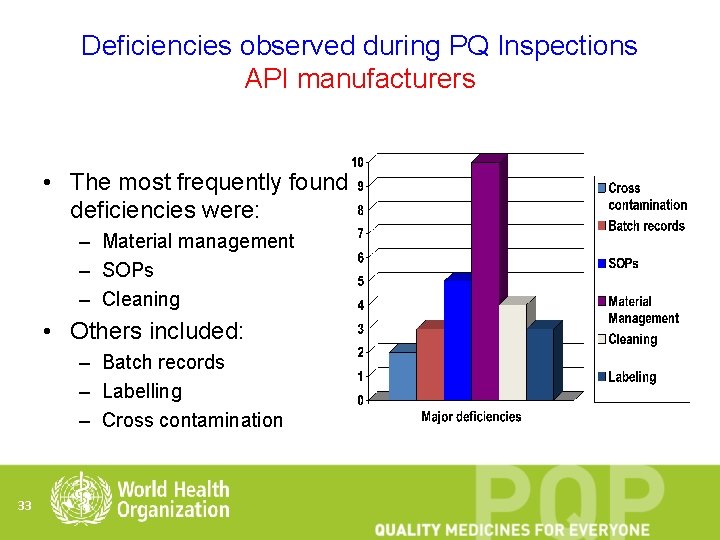
Deficiencies observed during PQ Inspections API manufacturers • The most frequently found deficiencies were: – Material management – SOPs – Cleaning • Others included: – Batch records – Labelling – Cross contamination 33

Inspections of Contract Research Organizations (CROs) 34
http: //apps. who. int/prequal/ 35

Prequalification Programme: International norms, standards and guidelines used in inspection activities to ensure wide applicability • HANDBOOK FOR GOOD CLINICAL RESEARCH PRACTICE (GCP) Guidance for implementation http: //whqlibdoc. who. int/publications/2005/924159392 X_eng. pdf • Guidelines for good clinical practice (GCP) for trials on pharmaceutical products. World Health Organization, 1995 (WHO Technical Report Series, No. 850), Annex 3. http: //apps. who. int/prequal/info_general/documents/TRS 850/WHO_TRS_850 Annex 3. pdf • Additional guidance for organizations performing in vivo bioequivalence studies. WHO Technical Report Series, No. 937, 2006, Annex 9 http: //apps. who. int/prequal/info_general/documents/TRS 937/WHO_TRS_937__an nex 9_eng. pdf • 36 Guidelines for the preparation of a contract research organization master file. World Health Organization, WHO Technical Report Series, No. 957, 2010 Annex 7, Page 271. http: //www. who. int/medicines/publications/TRS 957_2010. pdf Other guidelines e. g. ICH

CRO/BE Inspections: Problems with integrity, archiving and retrieval of documents 37

CRO/BE Inspections: Inadequate data integrity Source data either not available or authenticity questionable: • Source data could not be located to verify entries in VRFs – – destroyed accidently by fire or rain Sponsor claims the data were kept by the CRO, and the CRO claims the data were kept by the sponsor • Two of the ECGs shown to the inspectors, bearing different subject numbers and initials, were found to be identical. • Other ECGs bearing different subject numbers and initials appear to have been recorded from a single subject. Out of 95 ECGs copied by the inspectors, 43 appear to have been recorded from the same and single subject during a single session 38
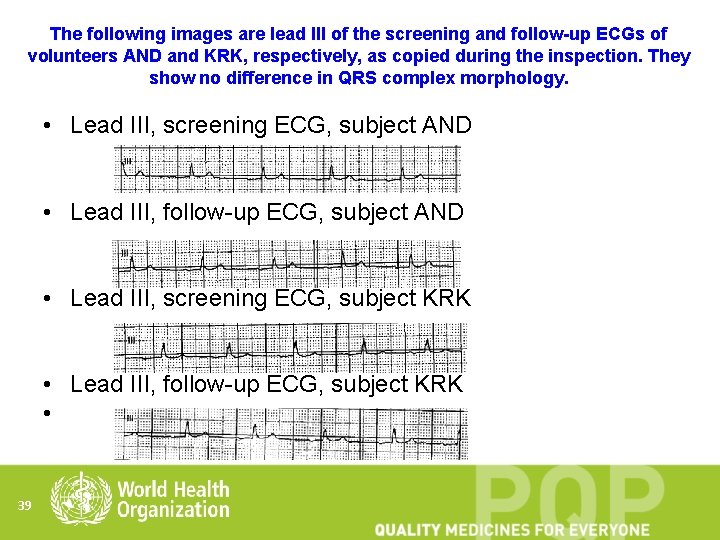
The following images are lead III of the screening and follow-up ECGs of volunteers AND and KRK, respectively, as copied during the inspection. They show no difference in QRS complex morphology. • Lead III, screening ECG, subject AND • Lead III, follow-up ECG, subject AND • Lead III, screening ECG, subject KRK • Lead III, follow-up ECG, subject KRK • 39

CRO/BE Inspections: Data manipulation – inappropriate manual integration of peaks • Manual reintegration of peak was done inappropriately and inconsistently for all peaks inclusive internal standard – For some samples checked, especially QCs or standards close or outside the 15% of their nominal concentration, the baseline of the chromatograms were modified manually. This was not done appropriately and consistently for all peaks inclusive internal standard. For modified integration, initial integration was not available. • No paper or electronic audit trail of manual integration available. • Each analytical run did not include calibration and quality control samples. 40
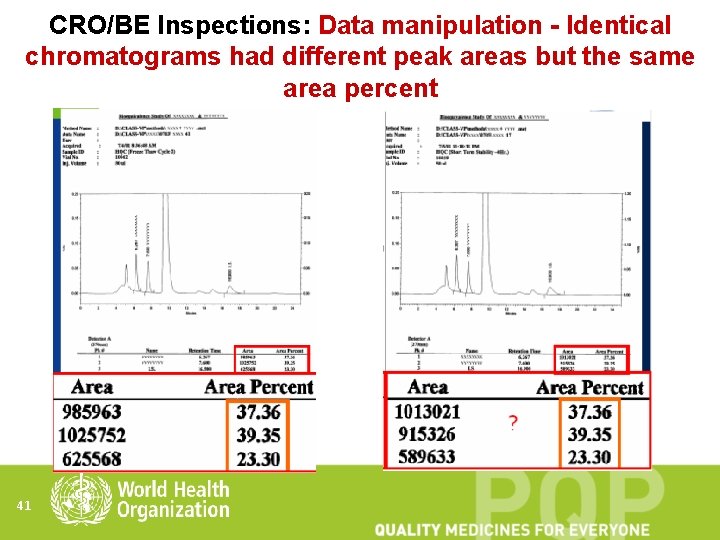
CRO/BE Inspections: Data manipulation - Identical chromatograms had different peak areas but the same area percent 41

Deficiencies observed during PQ Inspections QC Laboratories: Conclusions • Data manipulation and misrepresentation is not acceptable to the WHO and according to the law of most national regulatory authorities (could result in the publication of NOC or NOS on behalf of the WHO). • The honest way is always the "right way". • Good ethics are key to reliable data. • Always prioritize data integrity – not quick batch release at any cost. 42

WHO-PQ Inspections Summary and Conclusions • • • API, FPP and CRO/BE Inspections are an important part of the WHO-PQP evaluation and continuous monitoring process International norms, standards and guidelines are used in inspection activities to ensure wide applicability Collaborative and Risk management principles are applied to ensure efficient use of available resources Information put in public domain - available for use by NMRAs: WHOPIRs and NOCs Inspection results show that there are still a lot of poor manufacturing practices out there. Collaborative effort and skills are needed to ensure access to medicines of assured quality. Results show that WHO-PQP has made tremendous contribution in this respect. The support of NRAs in providing co-inspectors and observers is appreciated. This is good for: – – 43 Tapping into international skills Ensuring transparency Facilitating ownership Contributing to capacity building �����
- Slides: 43