ESSEE 4 Pali Serbia 17 22 Sept 2006

ESSEE 4 Palić, Serbia, 17 – 22 Sept. , 2006 Implementation of Advanced Electrodes to the Wastewaters Treatment Achille De Battisti Carlos Alberto Martinez-Huitle Sergio Ferro Laboratory of Electrochemistry University of Ferrara, Italy

Urban legends against the electrochemical way to wastewater remediation ü it’s not useful in case of poorly-conducting electrolytes ü it requires a huge consumption of electricity ü electrodes are expensive ü chemicals are easily available ü reactors are complex and/or difficult to manage ü…
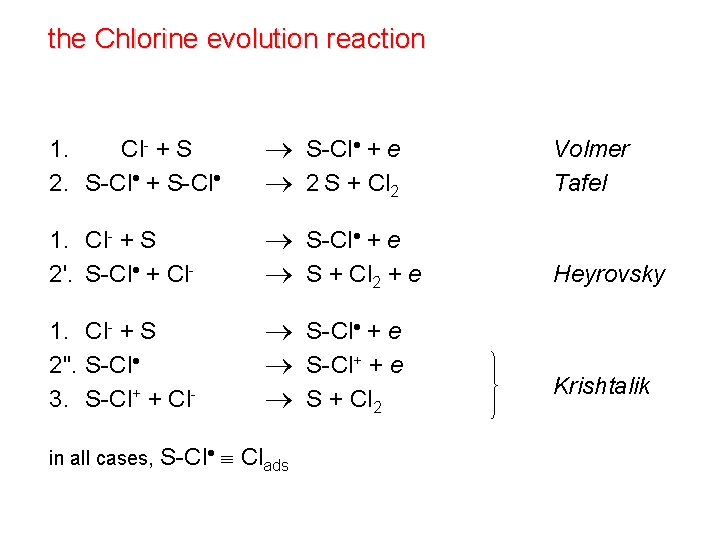
the Chlorine evolution reaction 1. 1. Cl- + S 2. S-Cl + e 2 S + Cl 2 Volmer Tafel 1. Cl- + S 2'. S-Cl + Cl- S-Cl + e S + Cl 2 + e Heyrovsky 1. Cl- + S 2". S-Cl 3. S-Cl+ + Cl- S-Cl + e S-Cl+ + e S + Cl 2 in all cases, S-Cl Clads Krishtalik
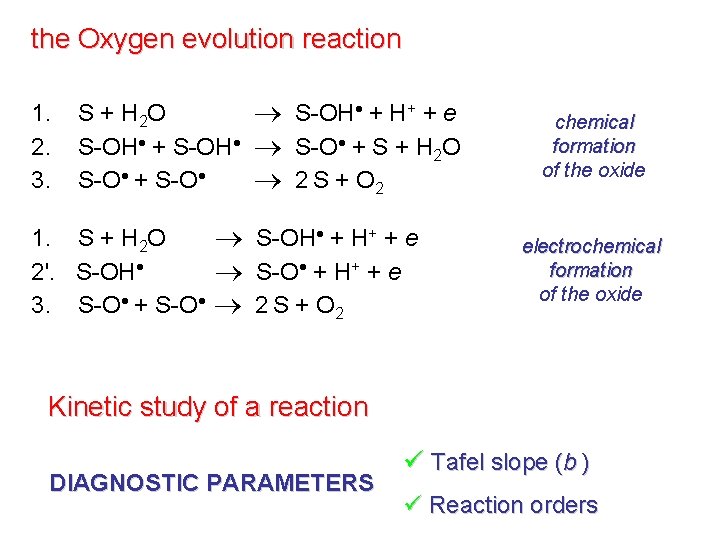
the Oxygen evolution reaction 1. 2. 3. S + H 2 O S-OH + H+ + e S-OH + S-OH S-O + S + H 2 O S-O + S-O 2 S + O 2 1. S + H 2 O S-OH + H+ + e 2'. S-OH S-O + H+ + e 3. S-O + S-O 2 S + O 2 chemical formation of the oxide electrochemical formation of the oxide Kinetic study of a reaction DIAGNOSTIC PARAMETERS ü Tafel slope (b ) ü Reaction orders

O 2 and Cl 2 evolution reactions in electrochemical incineration ü major links with the fundamentals of electrocatalysis ü a solid background for developments from bench to commercial scale Electrochemical incineration: how to follow it? ü Traditional analytical approaches: NMR, IR, UV-Vis. , mass spectrometry, different chromatographies… ü The alternative: v global analytical parameters, like COD and TOC v efficiency parameters (ICE, EOD)
the biological(aerobic/anaerobic) treatment biodegradable effluent toxic or non-biodegradable effluent
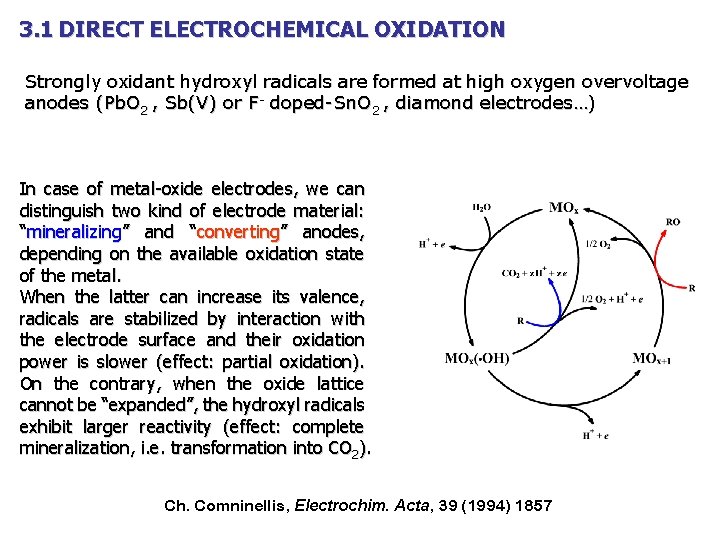
3. 1 DIRECT ELECTROCHEMICAL OXIDATION Strongly oxidant hydroxyl radicals are formed at high oxygen overvoltage anodes (Pb. O 2 , Sb(V) or F- doped-Sn. O 2 , diamond electrodes…) In case of metal-oxide electrodes, we can distinguish two kind of electrode material: “mineralizing” and “converting” anodes, depending on the available oxidation state of the metal. When the latter can increase its valence, radicals are stabilized by interaction with the electrode surface and their oxidation power is slower (effect: partial oxidation). On the contrary, when the oxide lattice cannot be “expanded”, the hydroxyl radicals exhibit larger reactivity (effect: complete mineralization, i. e. transformation into CO 2). Ch. Comninellis, Electrochim. Acta, 39 (1994) 1857
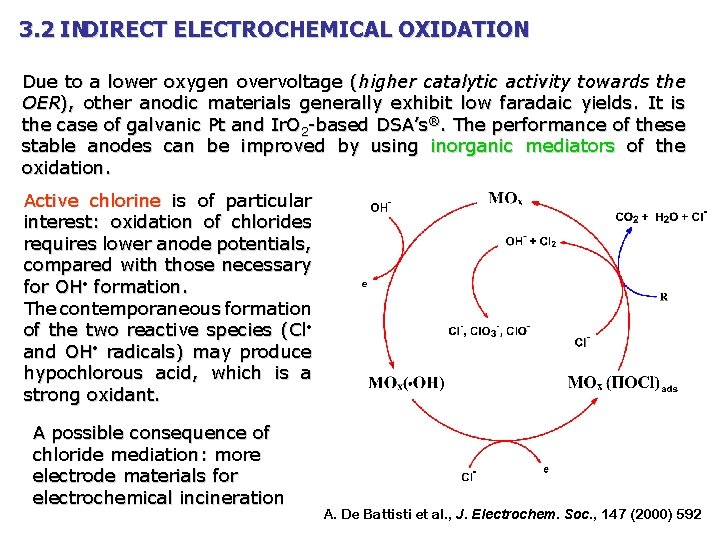
3. 2 INDIRECT ELECTROCHEMICAL OXIDATION Due to a lower oxygen overvoltage (higher catalytic activity towards the OER), other anodic materials generally exhibit low faradaic yields. It is the case of galvanic Pt and Ir. O 2 -based DSA’s®. The performance of these stable anodes can be improved by using inorganic mediators of the oxidation. Active chlorine is of particular interest: oxidation of chlorides requires lower anode potentials, compared with those necessary for OH formation. The contemporaneous formation of the two reactive species (Cl and OH radicals) may produce hypochlorous acid, which is a strong oxidant. A possible consequence of chloride mediation: more electrode materials for electrochemical incineration A. De Battisti et al. , J. Electrochem. Soc. , 147 (2000) 592
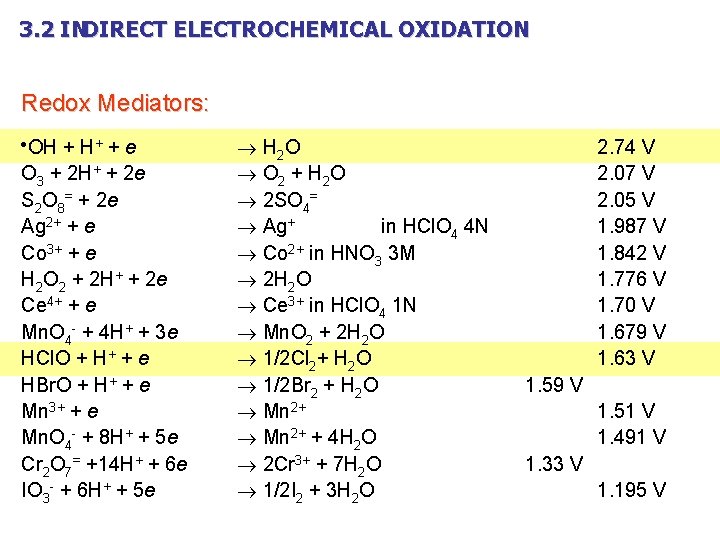
3. 2 INDIRECT ELECTROCHEMICAL OXIDATION Redox Mediators: OH + H+ + e O 3 + 2 H+ + 2 e S 2 O 8= + 2 e Ag 2+ + e Co 3+ + e H 2 O 2 + 2 H+ + 2 e Ce 4+ + e Mn. O 4 - + 4 H+ + 3 e HCl. O + H+ + e HBr. O + H+ + e Mn 3+ + e Mn. O 4 - + 8 H+ + 5 e Cr 2 O 7= +14 H+ + 6 e IO 3 - + 6 H+ + 5 e H 2 O O 2 + H 2 O 2 SO 4= Ag+ in HCl. O 4 4 N Co 2+ in HNO 3 3 M 2 H 2 O Ce 3+ in HCl. O 4 1 N Mn. O 2 + 2 H 2 O 1/2 Cl 2+ H 2 O 1/2 Br 2 + H 2 O Mn 2+ + 4 H 2 O 2 Cr 3+ + 7 H 2 O 1/2 I 2 + 3 H 2 O 2. 74 V 2. 07 V 2. 05 V 1. 987 V 1. 842 V 1. 776 V 1. 70 V 1. 679 V 1. 63 V 1. 59 V 1. 51 V 1. 491 V 1. 33 V 1. 195 V
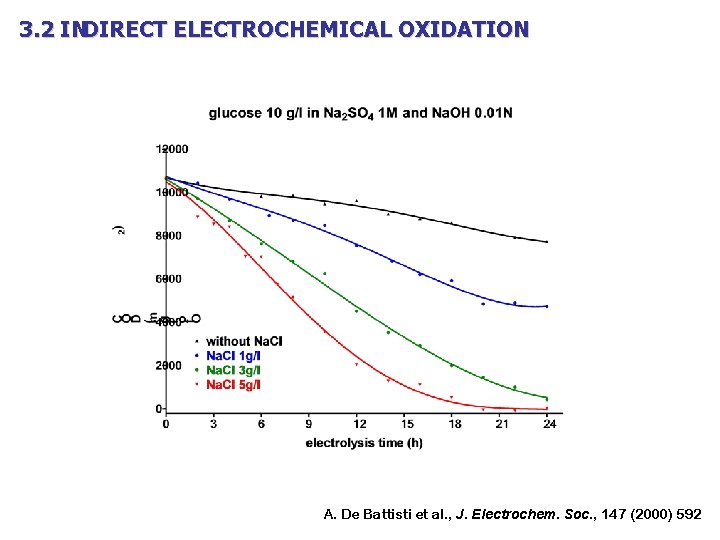
3. 2 INDIRECT ELECTROCHEMICAL OXIDATION A. De Battisti et al. , J. Electrochem. Soc. , 147 (2000) 592
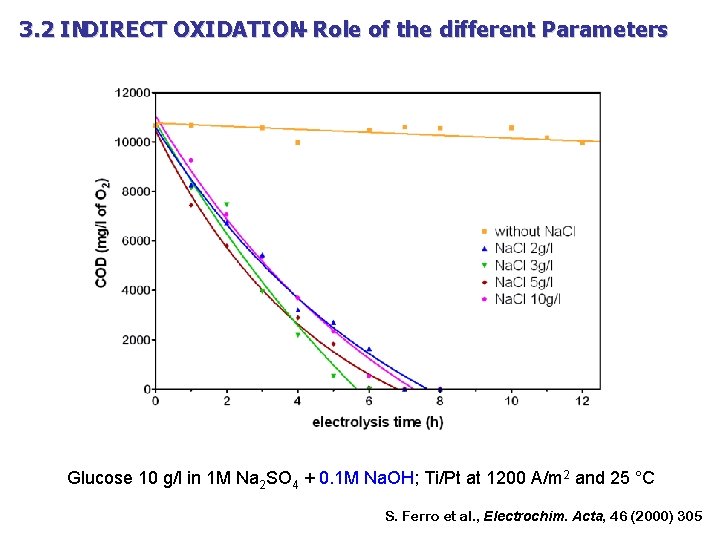
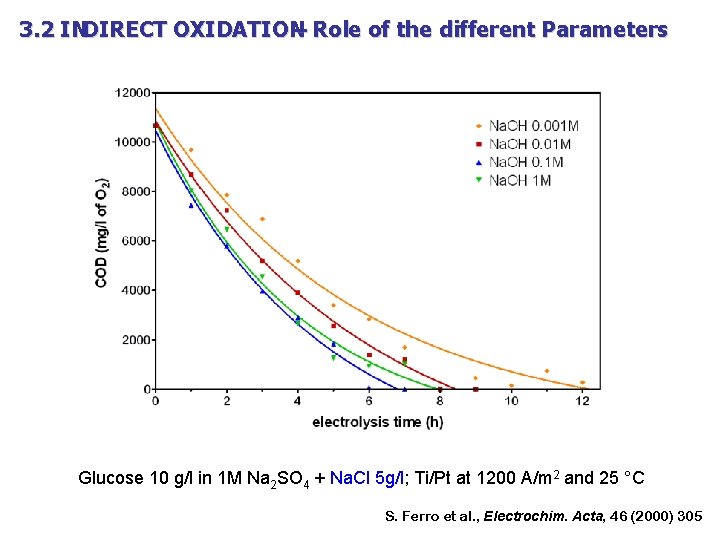
3. 2 INDIRECT OXIDATION – Role of the different Parameters Glucose 10 g/l in 1 M Na 2 SO 4 + 0. 1 M Na. OH; Ti/Pt at 1200 A/m 2 and 25 °C S. Ferro et al. , Electrochim. Acta, 46 (2000) 305
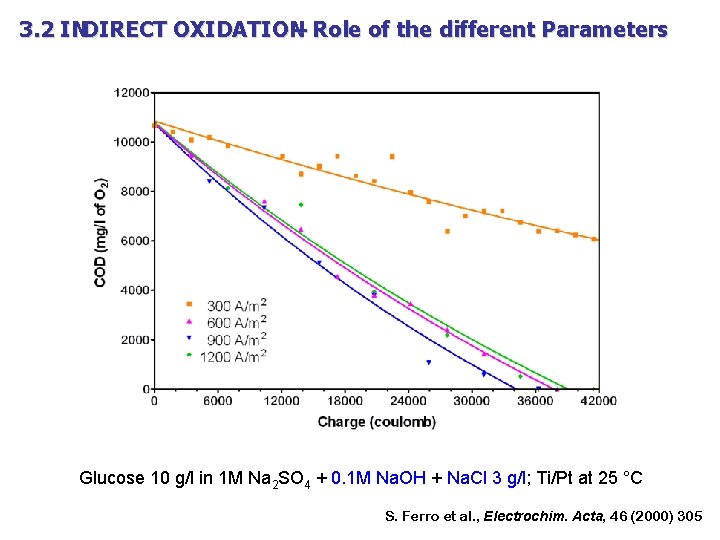
3. 2 INDIRECT OXIDATION – Role of the different Parameters Glucose 10 g/l in 1 M Na 2 SO 4 + Na. Cl 5 g/l; Ti/Pt at 1200 A/m 2 and 25 °C S. Ferro et al. , Electrochim. Acta, 46 (2000) 305
3. 2 INDIRECT OXIDATION – Role of the different Parameters Glucose 10 g/l in 1 M Na 2 SO 4 + 0. 1 M Na. OH + Na. Cl 3 g/l; Ti/Pt at 25 °C S. Ferro et al. , Electrochim. Acta, 46 (2000) 305
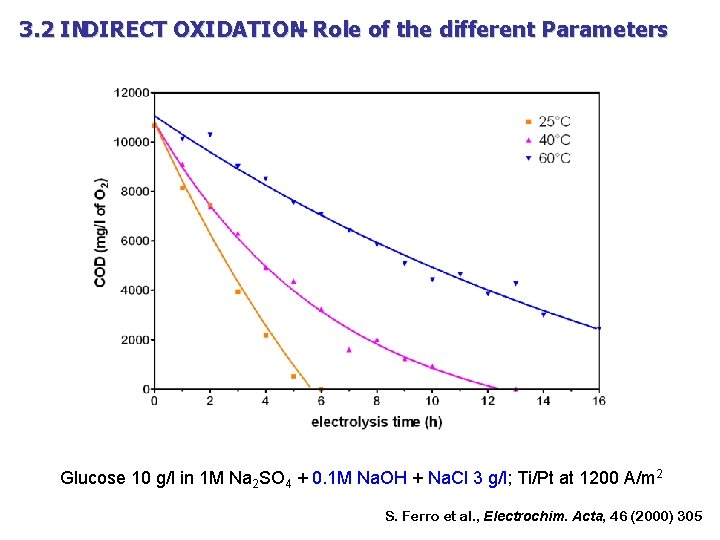
3. 2 INDIRECT OXIDATION – Role of the different Parameters Glucose 10 g/l in 1 M Na 2 SO 4 + 0. 1 M Na. OH + Na. Cl 3 g/l; Ti/Pt at 1200 A/m 2 S. Ferro et al. , Electrochim. Acta, 46 (2000) 305

3. 2 INDIRECT OXIDATION– WHAT HAVE WE LEARNT? ü relatively small amounts of chloride ions may inhibit the OER, causing an increase of the anode potential and therefore a higher reactivity of adsorbed hydroxyl and chloride/oxychloride radicals ü increasing the chloride concentration above a certain critical value would cause a potentiostatic buffering by the chlorine redox system, and consequently a decrease of the anode potential …may inhibit… …would cause… Further investigation is needed! S. Ferro et al. , Electrochim. Acta, 46 (2000) 305

The electrode material is becoming the main character …It means one more “tough” variable We have to simplify the experimental approach Let’ study a simpler substrate: Oxalic Acid
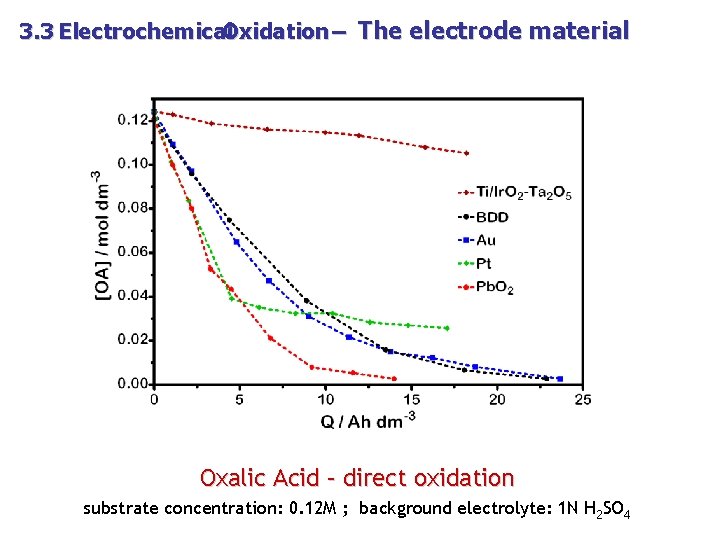
3. 3 Electrochemical Oxidation – The electrode material Oxalic Acid – direct oxidation substrate concentration: 0. 12 M ; background electrolyte: 1 N H 2 SO 4
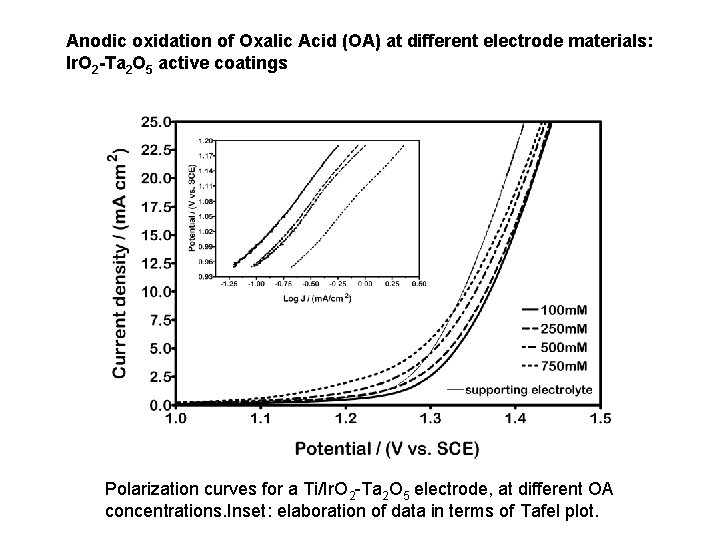
Anodic oxidation of Oxalic Acid (OA) at different electrode materials: Ir. O 2 -Ta 2 O 5 active coatings Polarization curves for a Ti/Ir. O 2 -Ta 2 O 5 electrode, at different OA concentrations. Inset: elaboration of data in terms of Tafel plot.
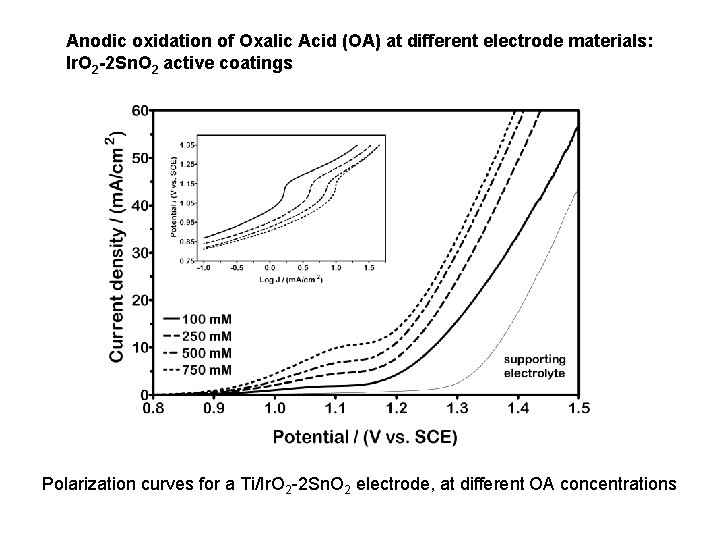
Anodic oxidation of Oxalic Acid (OA) at different electrode materials: Ir. O 2 -2 Sn. O 2 active coatings Polarization curves for a Ti/Ir. O 2 -2 Sn. O 2 electrode, at different OA concentrations
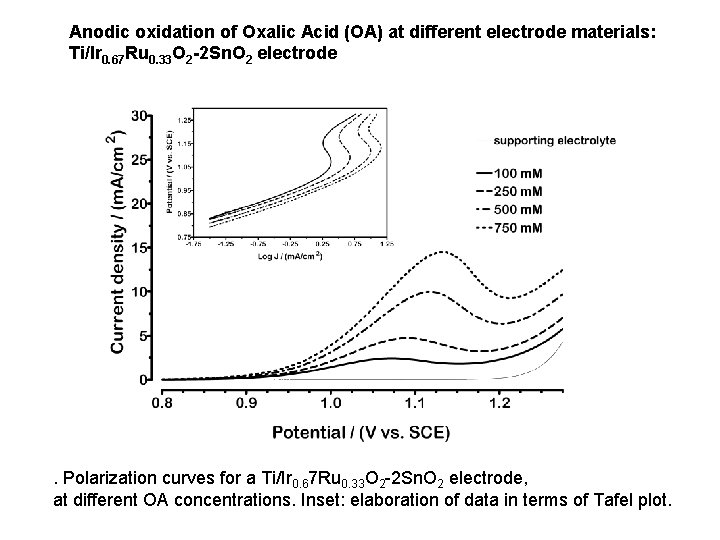
Anodic oxidation of Oxalic Acid (OA) at different electrode materials: Ti/Ir 0. 67 Ru 0. 33 O 2 -2 Sn. O 2 electrode . Polarization curves for a Ti/Ir 0. 67 Ru 0. 33 O 2 -2 Sn. O 2 electrode, at different OA concentrations. Inset: elaboration of data in terms of Tafel plot.
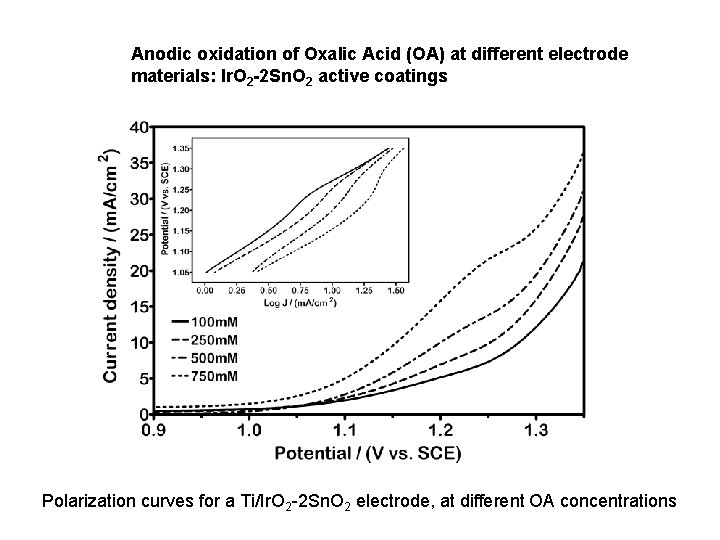
Anodic oxidation of Oxalic Acid (OA) at different electrode materials: Ir. O 2 -2 Sn. O 2 active coatings Polarization curves for a Ti/Ir. O 2 -2 Sn. O 2 electrode, at different OA concentrations
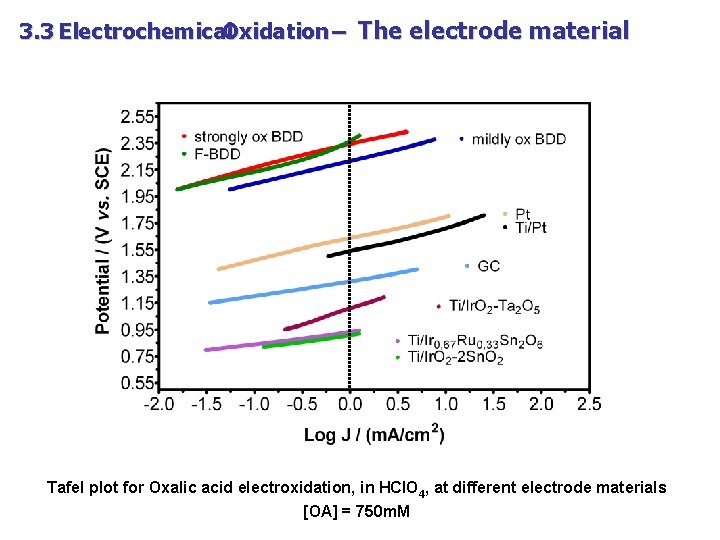
3. 3 Electrochemical Oxidation – The electrode material Tafel plot for Oxalic acid electroxidation, in HCl. O 4, at different electrode materials [OA] = 750 m. M
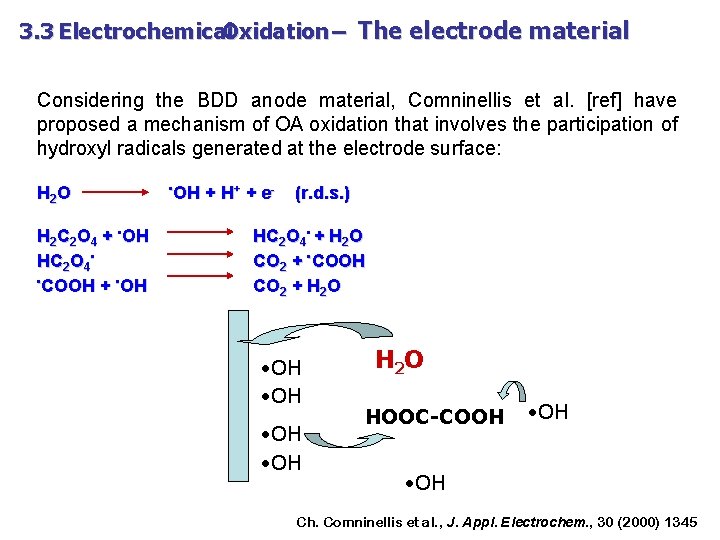
3. 3 Electrochemical Oxidation – The electrode material Considering the BDD anode material, Comninellis et al. [ref] have proposed a mechanism of OA oxidation that involves the participation of hydroxyl radicals generated at the electrode surface: H 2 O H 2 C 2 O 4 + • OH HC 2 O 4 • • COOH + • OH + H + + e- (r. d. s. ) HC 2 O 4 • + H 2 O CO 2 + • COOH CO 2 + H 2 O • OH H 2 O HOOC-COOH • OH Ch. Comninellis et al. , J. Appl. Electrochem. , 30 (2000) 1345
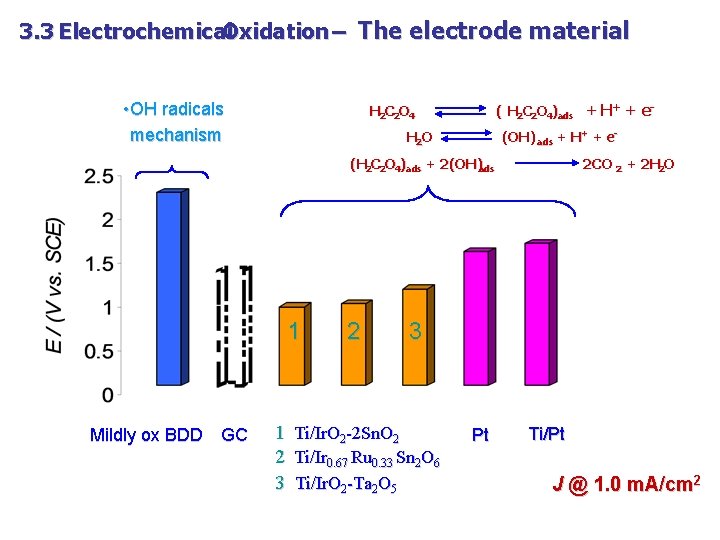
3. 3 Electrochemical Oxidation – The electrode material • OH radicals mechanism ( H 2 C 2 O 4)ads + H+ + e- H 2 C 2 O 4 H 2 O (OH) ads + H+ + e- (H 2 C 2 O 4)ads + 2(OH)ads 1 Mildly ox BDD GC 2 2 CO 2 + 2 H 2 O 3 1 Ti/Ir. O 2 -2 Sn. O 2 2 Ti/Ir 0. 67 Ru 0. 33 Sn 2 O 6 3 Ti/Ir. O 2 -Ta 2 O 5 Pt Ti/Pt J @ 1. 0 m. A/cm 2
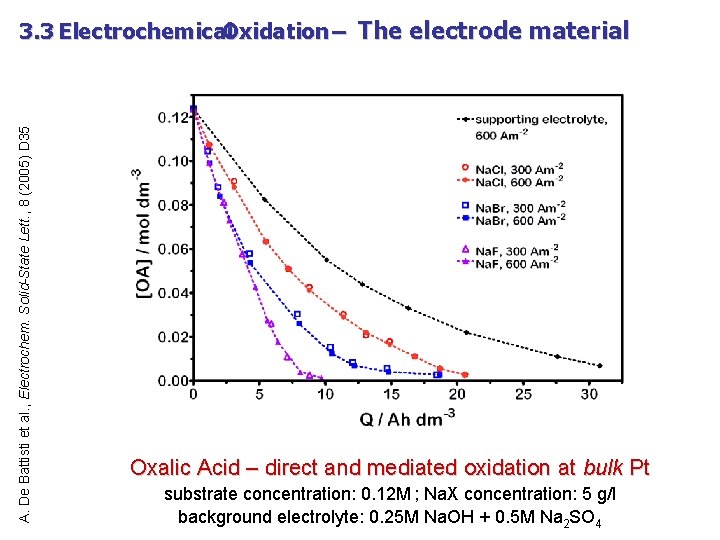
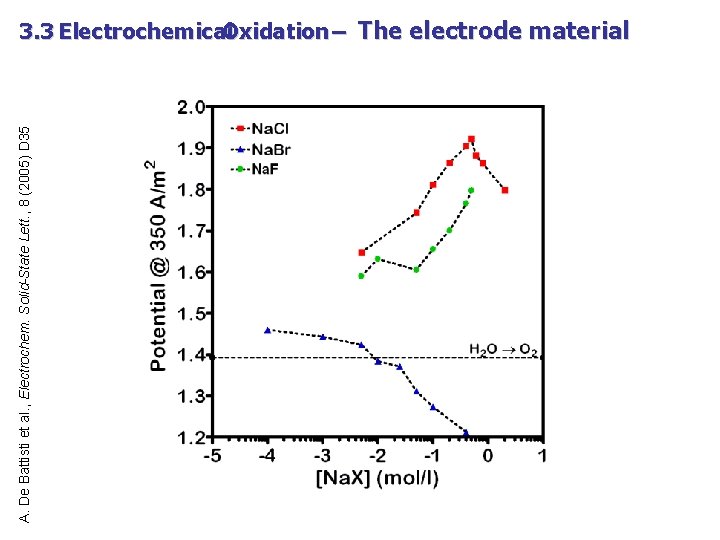
A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35 3. 3 Electrochemical Oxidation – The electrode material Oxalic Acid – direct and mediated oxidation at bulk Pt substrate concentration: 0. 12 M ; Na. X concentration: 5 g/l background electrolyte: 0. 25 M Na. OH + 0. 5 M Na 2 SO 4
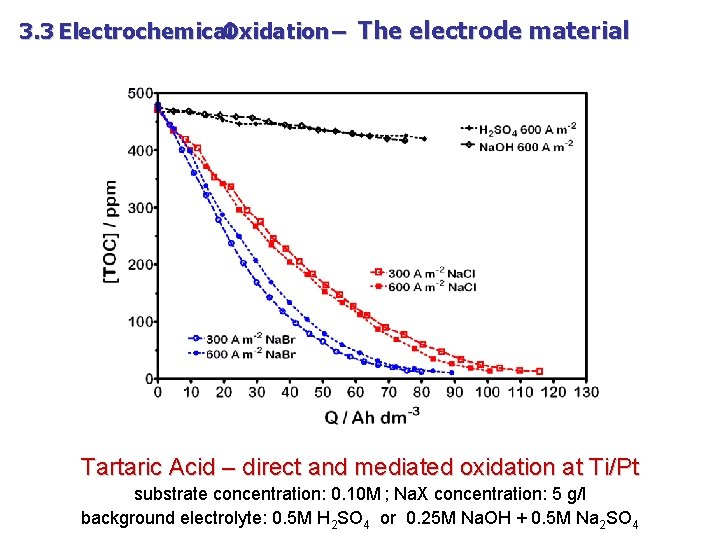
3. 3 Electrochemical Oxidation – The electrode material Tartaric Acid – direct and mediated oxidation at Ti/Pt substrate concentration: 0. 10 M ; Na. X concentration: 5 g/l background electrolyte: 0. 5 M H 2 SO 4 or 0. 25 M Na. OH + 0. 5 M Na 2 SO 4
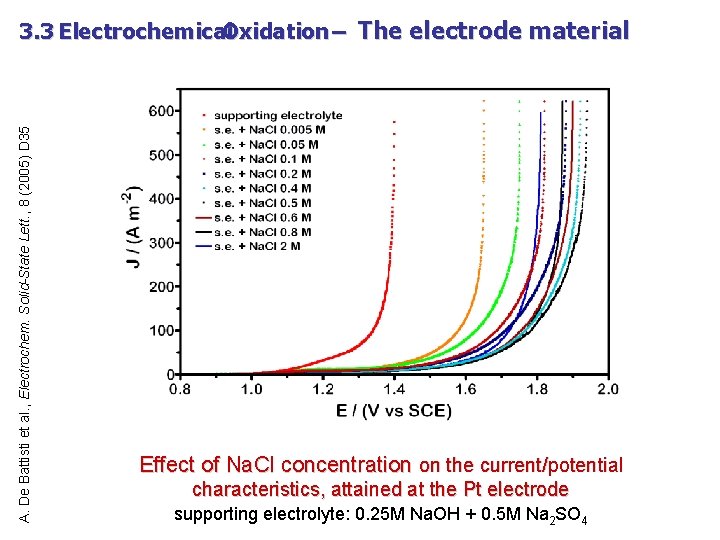
A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35 3. 3 Electrochemical Oxidation – The electrode material Effect of Na. Cl concentration on the current/potential characteristics, attained at the Pt electrode supporting electrolyte: 0. 25 M Na. OH + 0. 5 M Na 2 SO 4
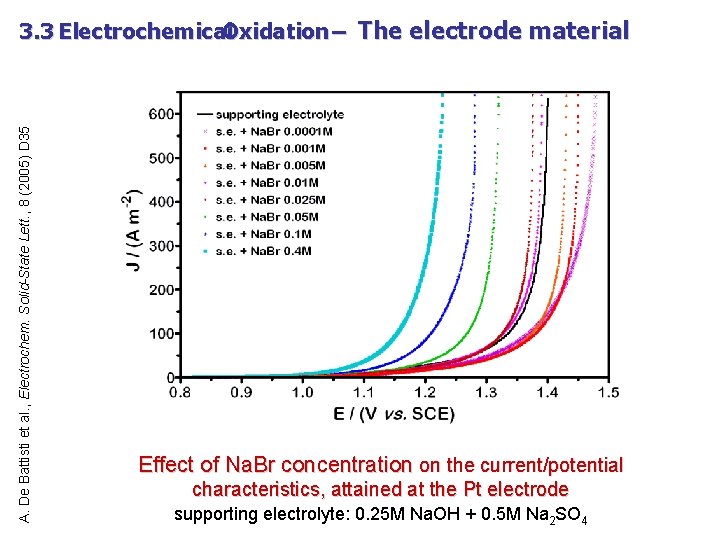
A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35 3. 3 Electrochemical Oxidation – The electrode material Effect of Na. Br concentration on the current/potential characteristics, attained at the Pt electrode supporting electrolyte: 0. 25 M Na. OH + 0. 5 M Na 2 SO 4
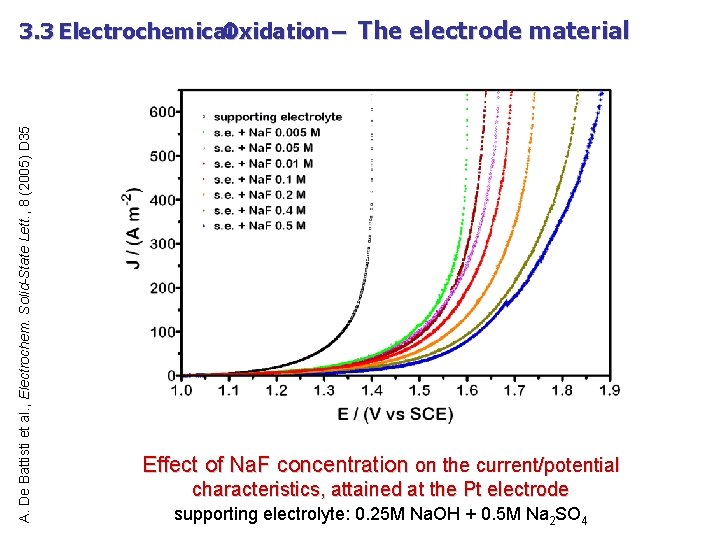
A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35 3. 3 Electrochemical Oxidation – The electrode material Effect of Na. F concentration on the current/potential characteristics, attained at the Pt electrode supporting electrolyte: 0. 25 M Na. OH + 0. 5 M Na 2 SO 4
A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35 3. 3 Electrochemical Oxidation – The electrode material

Halogenide-mediated (indirect) electrochemical incineration (alkaline media): ü Volume reaction of the substrate with electrogenerated strong oxidants (Cl. O 2, HCl. O, Cl. O-, Br. O 3 -); ü Surface reaction of the adsorbed substrate with electrosorbed species (e. g. : oxy-chloro radicals); ü Inhibition of the oxygen evolution reaction. “Direct” electrochemical incineration: ü Concomitant with oxygen evolution reaction; ü Good faradaic yields at high-oxygen overvoltage anodes; ü Weakly adsorbed hydroxyl radicals are the main factor leading to electrochemical incineration; ü As an extreme case, hydroxyl radicals may act within a reaction cage nearby the electrode surface. A. De Battisti et al. , Electrochem. Solid-State Lett. , 8 (2005) D 35
a real approach… for the potabilization of water objectives sterilization of solutions for medical purposes low capacity municipal plants final treatment
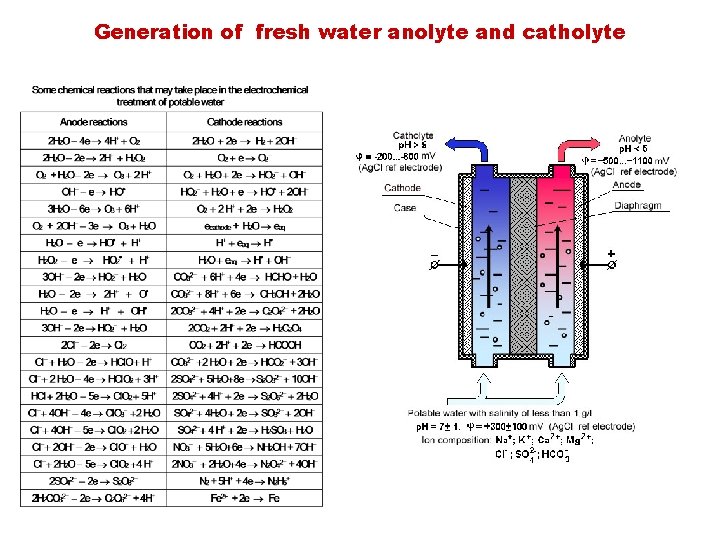
Generation of fresh water anolyte and catholyte
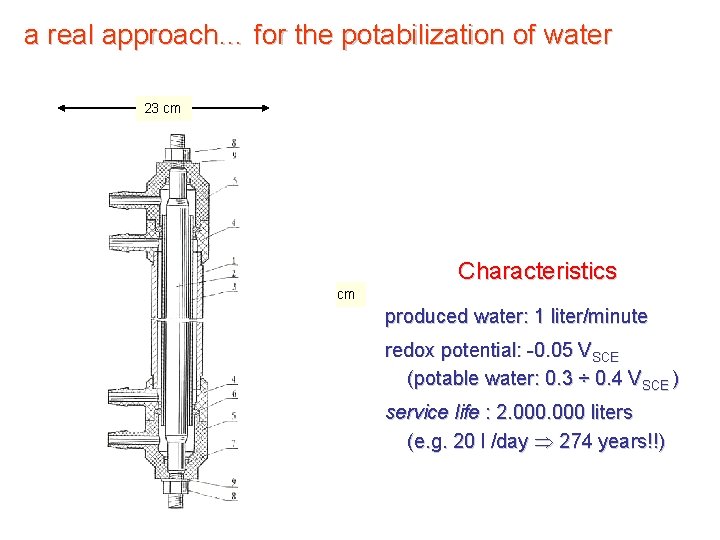
a real approach… for the potabilization of water 23 cm Characteristics 29 cm produced water: 1 liter/minute redox potential: -0. 05 VSCE (potable water: 0. 3 ÷ 0. 4 VSCE ) service life : 2. 000 liters (e. g. 20 l /day 274 years!!)
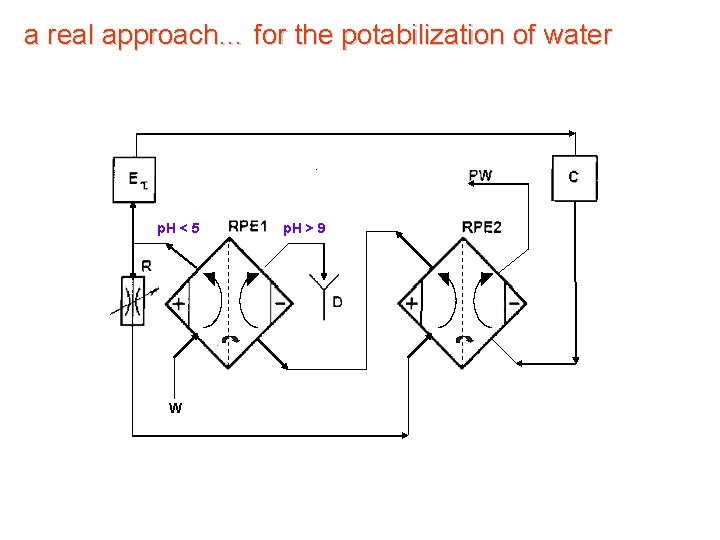
a real approach… for the potabilization of water p. H < 5 W p. H > 9
Potable water Food Processing Agricultural Applications Cooling Towers Disinfection applications Swimming Pools, Spas, Hot tubs Wastewater
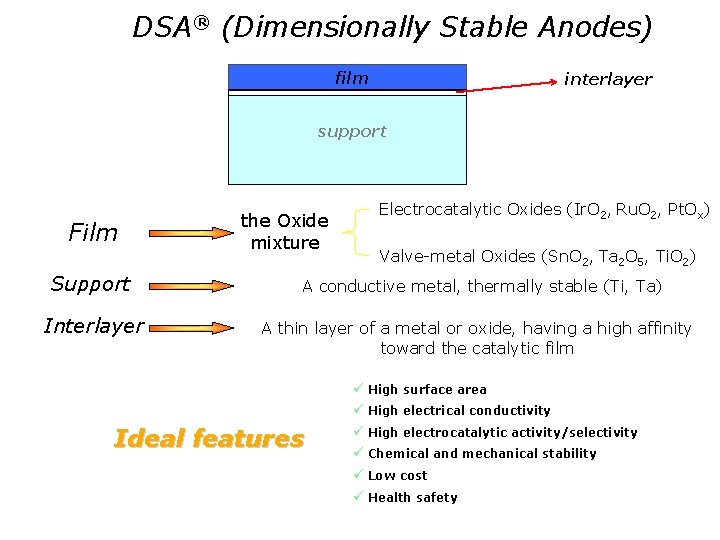
DSA® (Dimensionally Stable Anodes) film interlayer support Film Support Interlayer the Oxide mixture Electrocatalytic Oxides (Ir. O 2, Ru. O 2, Pt. Ox) Valve-metal Oxides (Sn. O 2, Ta 2 O 5, Ti. O 2) A conductive metal, thermally stable (Ti, Ta) A thin layer of a metal or oxide, having a high affinity toward the catalytic film ü High surface area ü High electrical conductivity Ideal features ü High electrocatalytic activity/selectivity ü Chemical and mechanical stability ü Low cost ü Health safety

The accelerated service-life test for oer DSA Need for quicker diagnostics, e. g. : 1 -4 months Typical test example : Solution: 3 M H 2 SO 4 Temperature: 60°C Galvanostatic conditions, j = 10 – 50 k. A-2 m Test end upon 1 V increase in cell potential (polarization curves and CV’s recorded during the experiments)

The accelerated service-life test for oer DSA. A possible way toanalizethe results Ø The passivation(deactivation) time can be misleading (the catalyst loading (film thickness) is not properly considered). Ø Charge consumption per unit electrode-surface-area (e. g. k. Ah m-2) is more meaningful than time; Ø Normalization to film thickness (catalyst loading) is mandatory. Ø At fixed coating composition the amount of noblemetal (e. g. : g. Ir m-2) can be used as normalizing factor
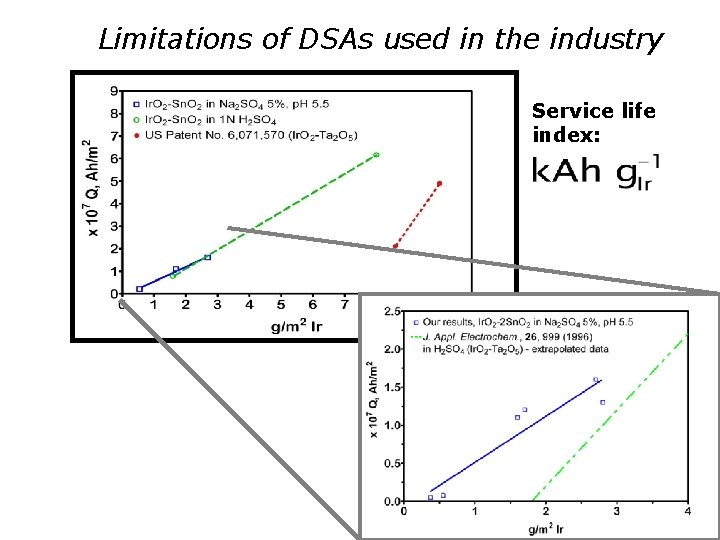
Limitations of DSAs used in the industry Service life index:
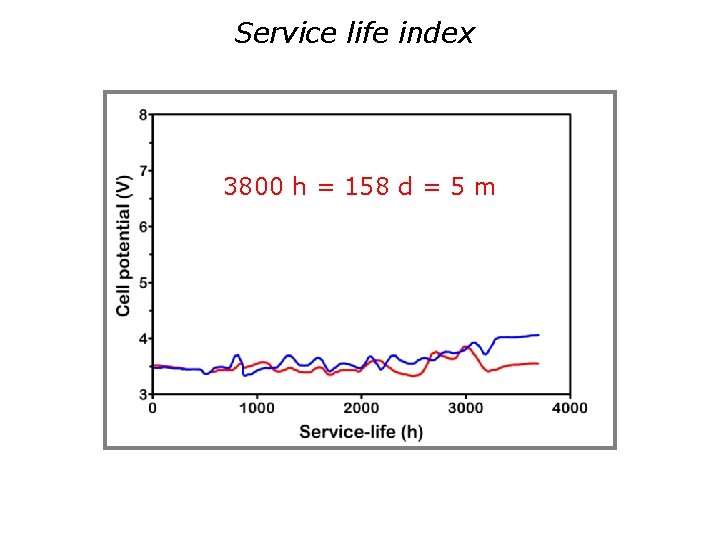
Service life index 3800 h = 158 d = 5 m

“Building Blocks” § Preparation (0 100% Mol Ir. O 2 ) § Characterization • Microstructure(XRD) (SEM) (EDX) (AFM) • Electrochemical Activity • Service life

“Building Blocks” üPreparation (0 100% Ir, constant-mass deposits) • Ti-support etching by conc. Na. OH • (acidic treatments lead to shorter s. l. ) • Interlayer deposition (thermal methods) • precursor deposition (Ir(IV) and Sn(IV) chloro-aceto • • complexes, colloidal suspensions) Thermal decomposition: 450 °C)

Scanning Electron Microscopy Images 1200 X “cracked mud” Increase of the Ir % 1200 X 35% Ir 30000 X 50% Ir 100% Ir Complete absence of organisation
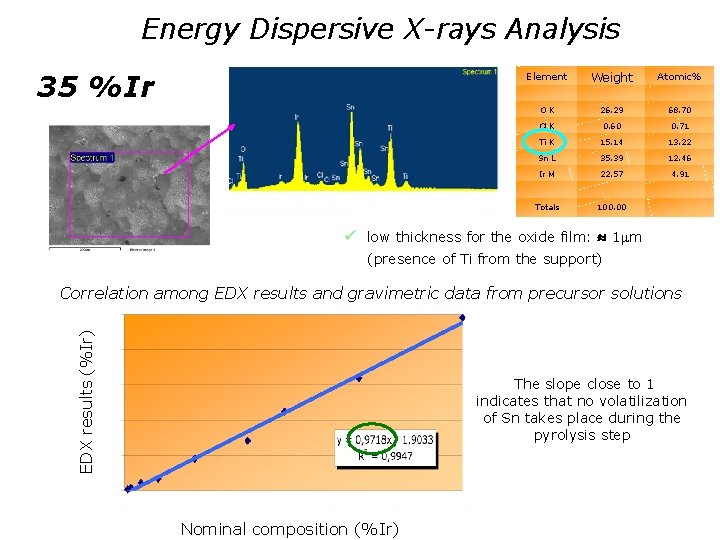
Energy Dispersive X-rays Analysis 35 % Ir Weight % Atomic% OK 26. 29 68. 70 Cl K 0. 60 0. 71 Ti K 15. 14 13. 22 Sn L 35. 39 12. 46 Ir M 22. 57 4. 91 Totals 100. 00 Element ü low thickness for the oxide film: 1 m (presence of Ti from the support) EDX results (%Ir) Correlation among EDX results and gravimetric data from precursor solutions The slope close to 1 indicates that no volatilization of Sn takes place during the pyrolysis step Nominal composition (%Ir)
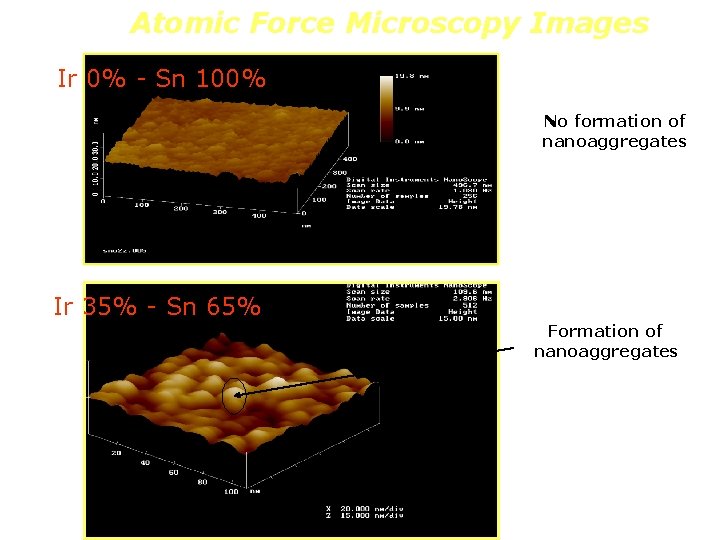
Atomic Force Microscopy Images Ir 0% - Sn 100% No formation of nanoaggregates Ir 35% - Sn 65% Formation of nanoaggregates
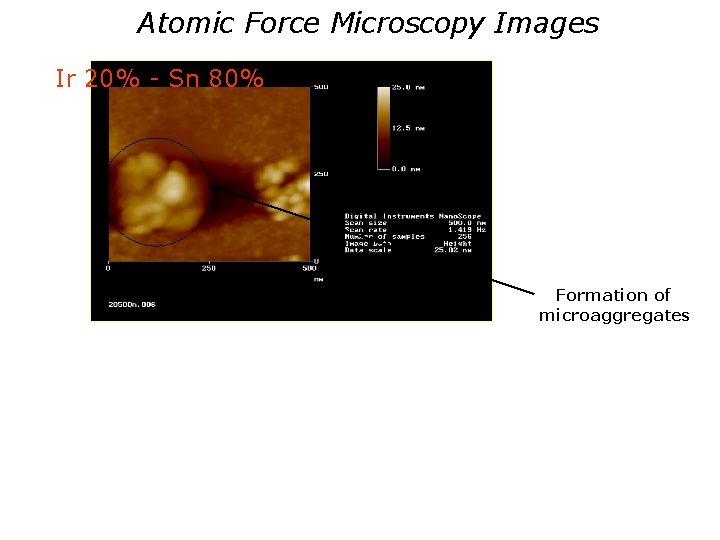
Atomic Force Microscopy Images Ir 20% - Sn 80% Formation of microaggregates
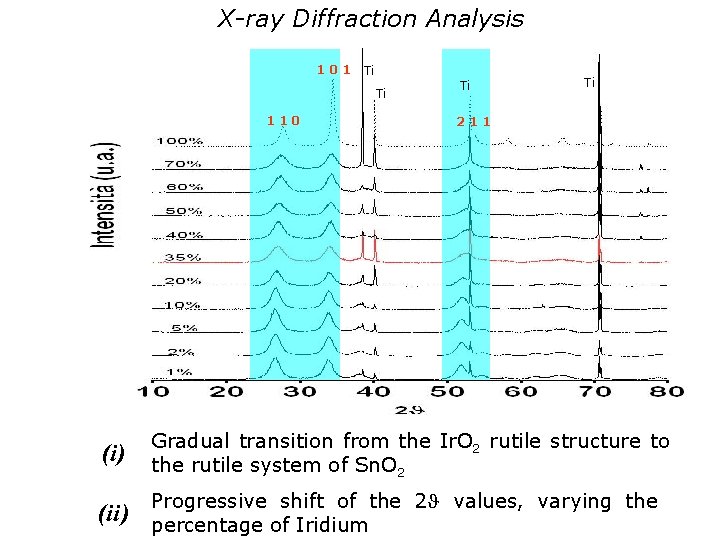
X-ray Diffraction Analysis 101 Ti Ti 110 Ti Ti 211 (i ) Gradual transition from the Ir. O 2 rutile structure to the rutile system of Sn. O 2 (ii ) Progressive shift of the 2 values, varying the percentage of Iridium
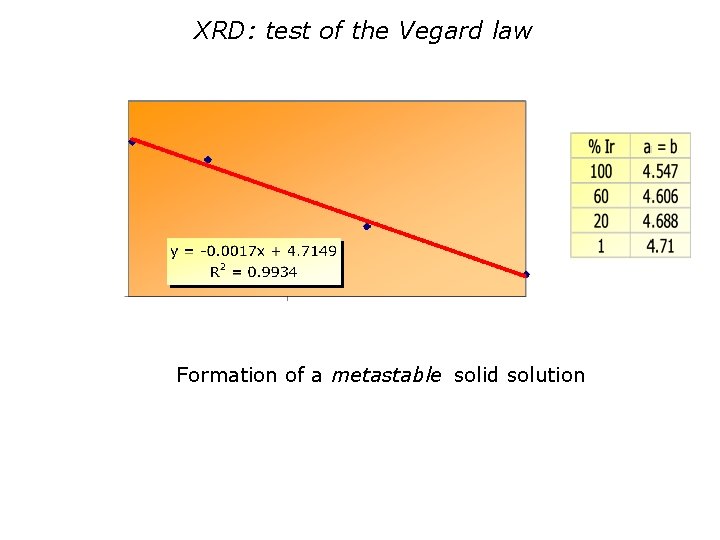
XRD: test of the Vegard law Formation of a metastable solid solution
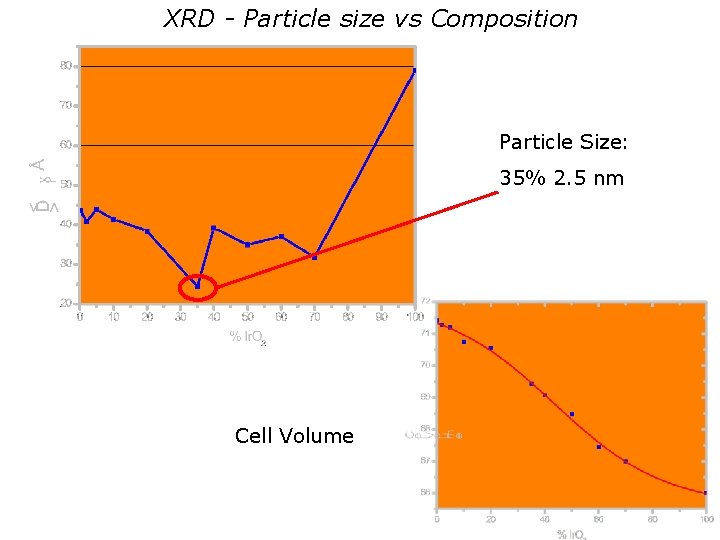
XRD - Particle size vs Composition Particle Size: 35% 2. 5 nm Cell Volume
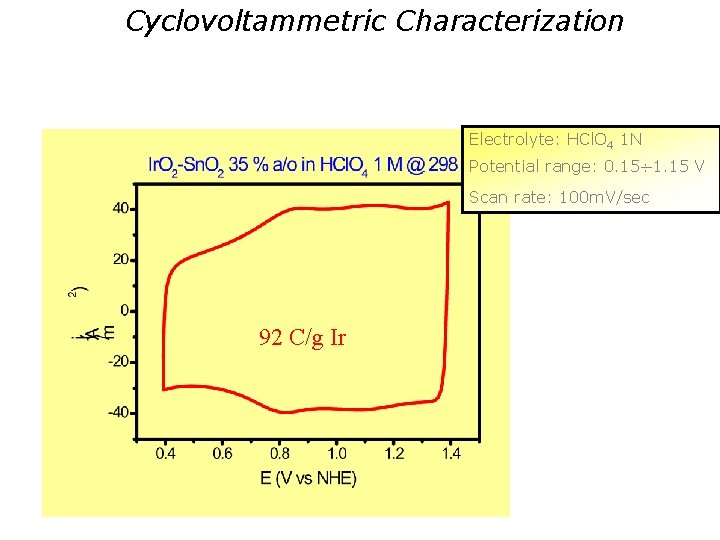
Cyclovoltammetric Characterization Electrolyte: HCl. O 4 1 N Potential range: 0. 15 1. 15 V Scan rate: 100 m. V/sec 92 C/g Ir
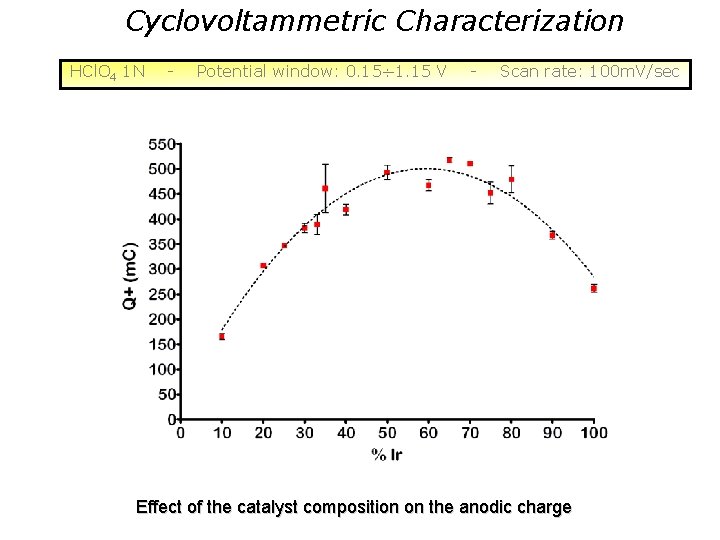
Cyclovoltammetric Characterization HCl. O 4 1 N - Potential window: 0. 15 1. 15 V - Scan rate: 100 m. V/sec Effect of the catalyst composition on the anodic charge
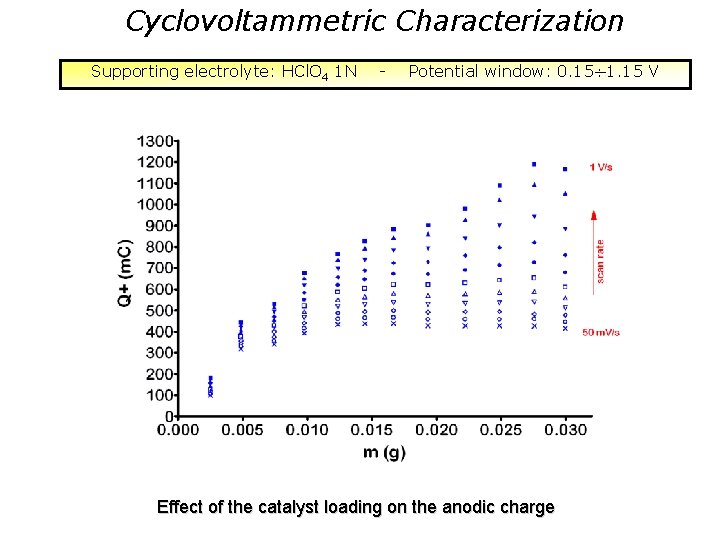
Cyclovoltammetric Characterization Supporting electrolyte: HCl. O 4 1 N - Potential window: 0. 15 1. 15 V Effect of the catalyst loading on the anodic charge
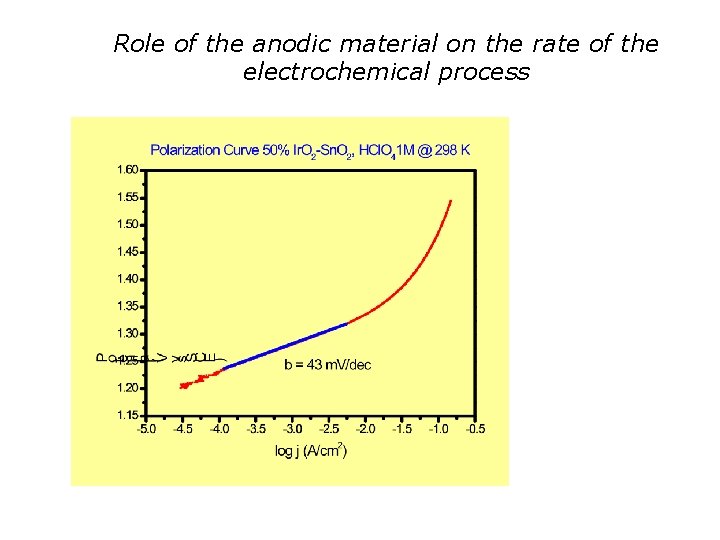
Role of the anodic material on the rate of the electrochemical process

Kinetic study: considerations % Ir bexp (m. V/dec) 30 46 35 46 40 47 50 43 bexp beff S-OH « 1 Optimal catalytic activity (high Turn. Over number)

Kinetic study of the Oxygen Evolution Reaction Electrochemical Oxide formation Hypothesis • H 2 O (H 2 O)ads • first step in equilibrium • second step rate determining • low overpotentials • s-OH 0 K 1 (reql constant)
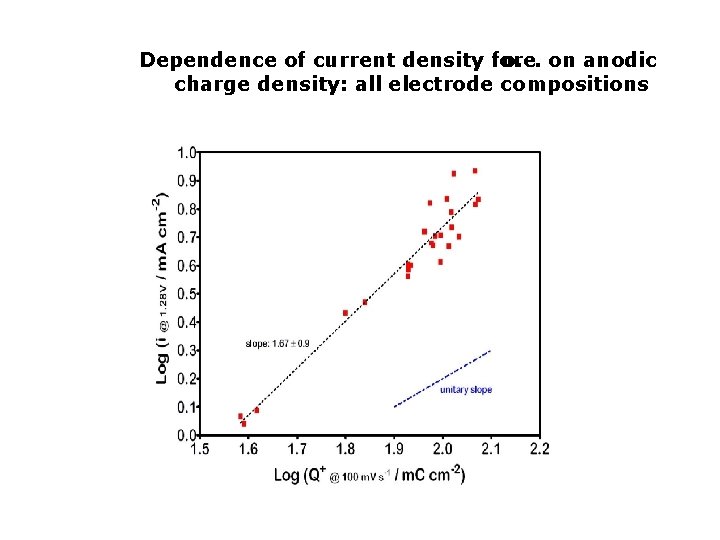
Dependence of current density for o. e. on anodic charge density: all electrode compositions
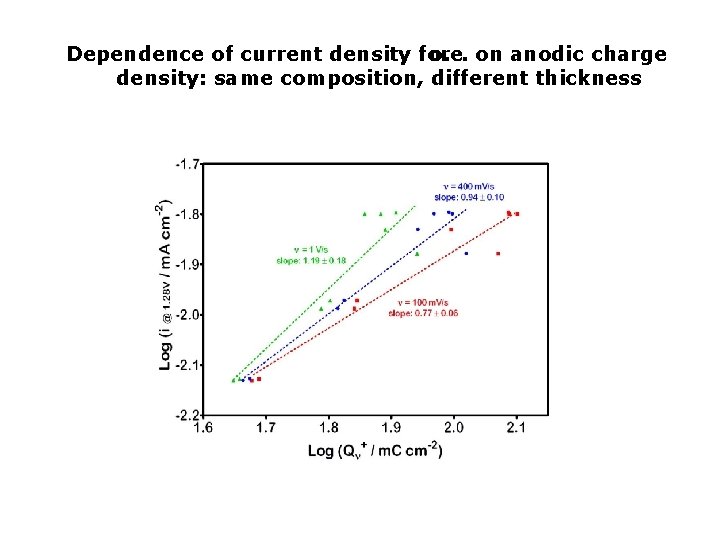
Dependence of current density for o. e. on anodic charge density: same composition, different thickness

The Group!: Martina. Donatoni Sergio Ferro Fabio Galli Carlos Alberto Martinez. Huitle Davide Perelli Lourdes Vazquez-Gomez
- Slides: 59