ESC Basic Science Summer School Sophia Antipolis 2017

ESC Basic Science Summer School, Sophia Antipolis, 2017 CONTROL OF THROMBOSIS AND FIBRINOLYSIS IN VIVO: IMPLICATIONS FOR NEW THERAPIES NICOLA J MUTCH

m o r Th s i s bo Haemo stasis
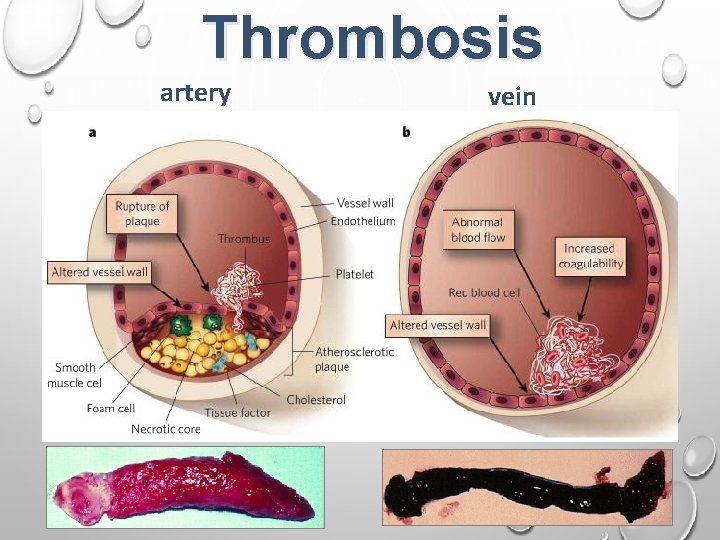
Thrombosis artery vein

Holy grail: An antithrombotic drug that reduces thrombosis with minimal or no bleeding complications
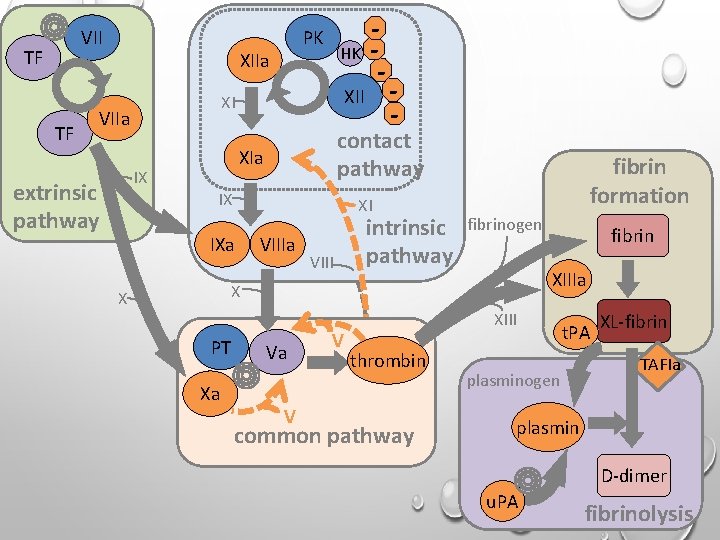
VII TF TF PK XIIa XII XI VIIa - contact pathway XIa IX extrinsic pathway HK - IX XI XI IXa VIII intrinsic pathway fibrin formation fibrinogen XIIIa X X PT Xa Va V V fibrin XIII thrombin common pathway t. PA plasminogen XL-fibrin TAFIa plasmin D-dimer u. PA fibrinolysis
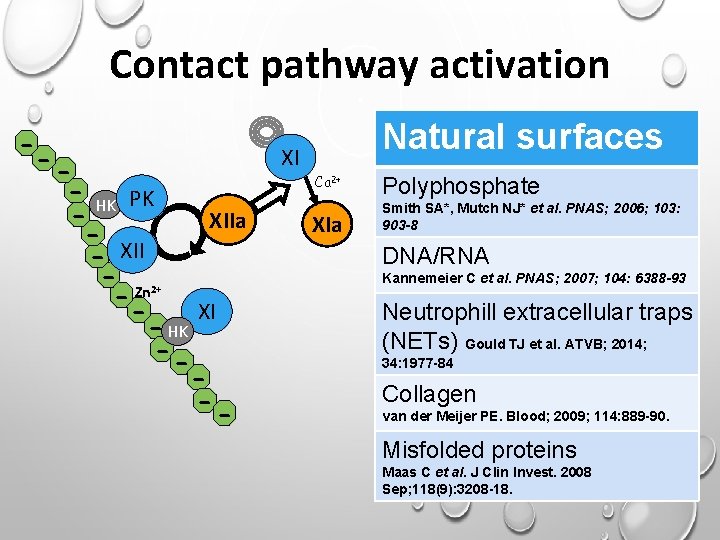
Contact pathway activation - -- HK PK XIIa - XII - Zn XI - HK --2+ XI Natural surfaces Ca 2+ XIa Polyphosphate Smith SA*, Mutch NJ* et al. PNAS; 2006; 103: 903 -8 DNA/RNA Kannemeier C et al. PNAS; 2007; 104: 6388 -93 Neutrophill extracellular traps (NETs) Gould TJ et al. ATVB; 2014; 34: 1977 -84 Collagen van der Meijer PE. Blood; 2009; 114: 889 -90. Misfolded proteins Maas C et al. J Clin Invest. 2008 Sep; 118(9): 3208 -18.
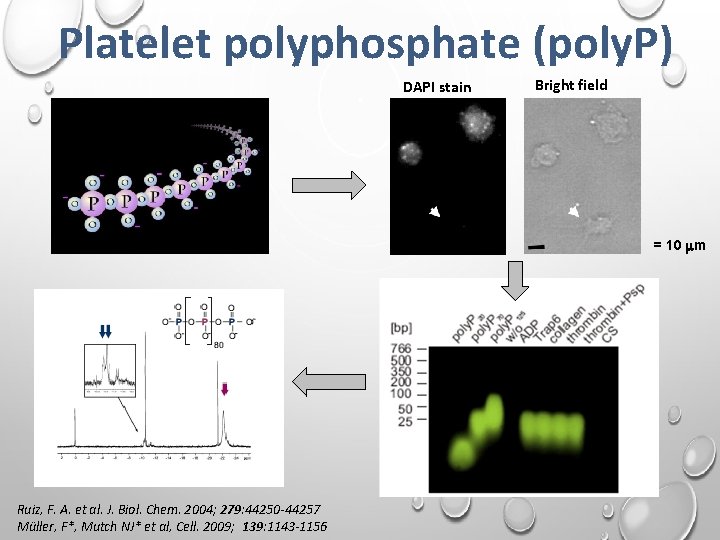
Platelet polyphosphate (poly. P) DAPI stain Bright field = 10 mm Ruiz, F. A. et al. J. Biol. Chem. 2004; 279: 44250 -44257 Müller, F*, Mutch NJ* et al, Cell. 2009; 139: 1143 -1156
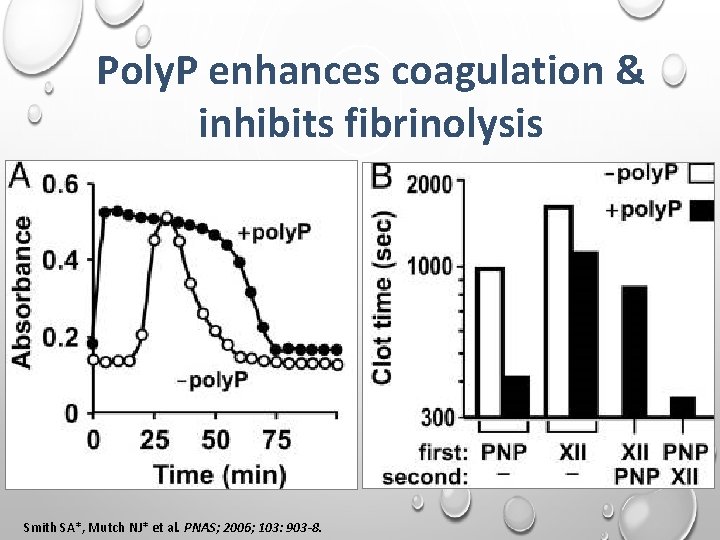
Poly. P enhances coagulation & inhibits fibrinolysis Smith SA*, Mutch NJ* et al. PNAS; 2006; 103: 903 -8.

FXII deficiency attenuates thrombosis but not haemostasis Renné et al, JEM. 2005; 202: 271 -281
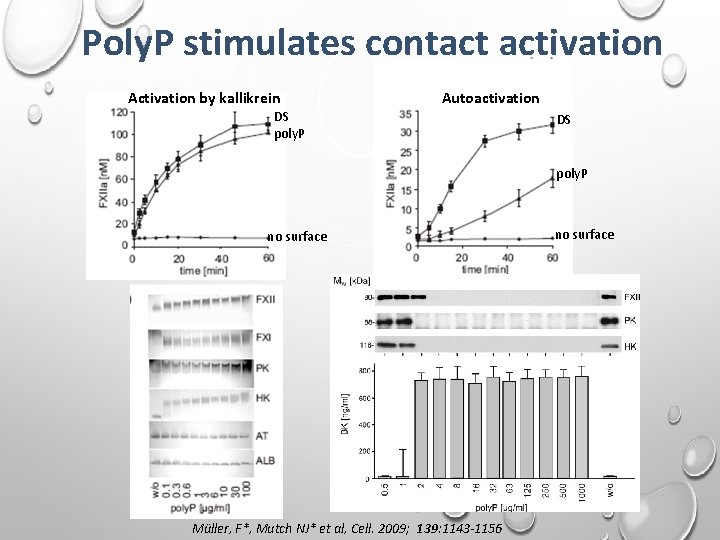
Poly. P stimulates contact activation Activation by kallikrein Autoactivation DS poly. P no surface Müller, F*, Mutch NJ* et al, Cell. 2009; 139: 1143 -1156 no surface
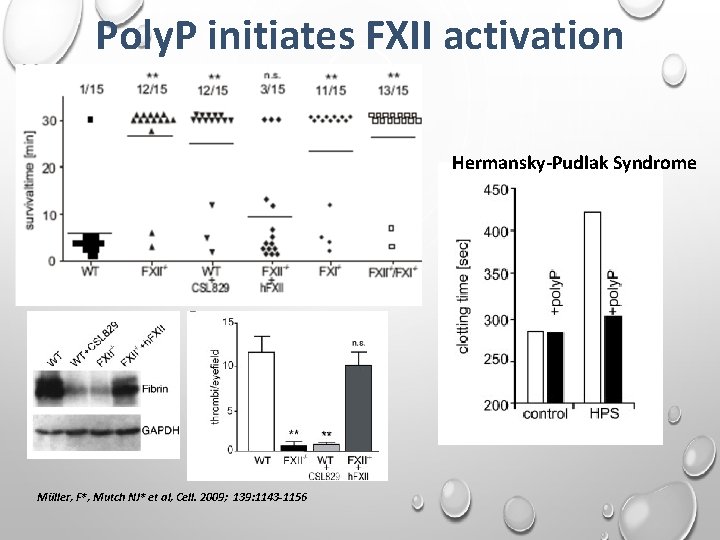
Poly. P initiates FXII activation Hermansky-Pudlak Syndrome Müller, F*, Mutch NJ* et al, Cell. 2009; 139: 1143 -1156
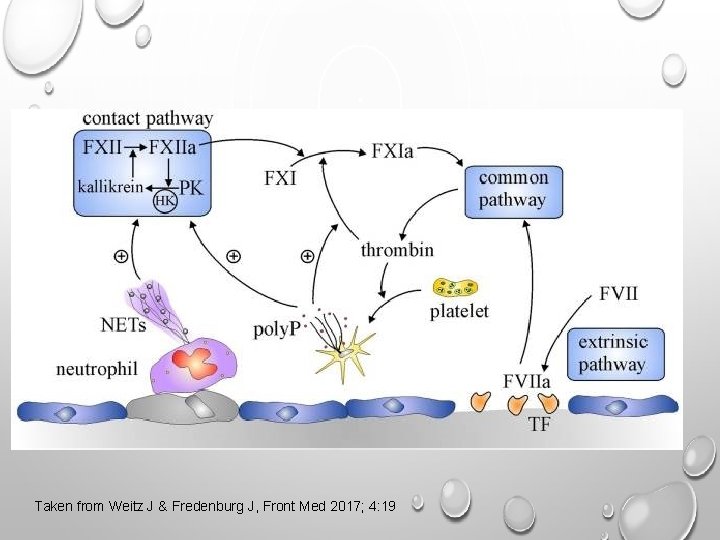
Taken from Weitz J & Fredenburg J, Front Med 2017; 4: 19
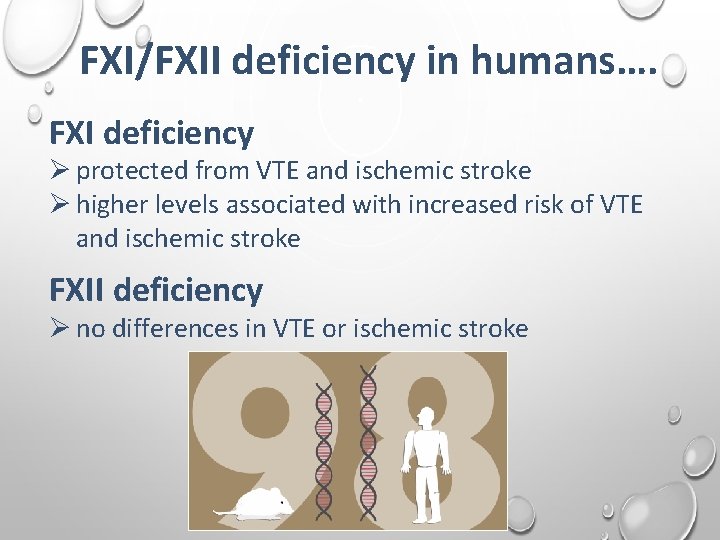
FXI/FXII deficiency in humans…. FXI deficiency Ø protected from VTE and ischemic stroke Ø higher levels associated with increased risk of VTE and ischemic stroke FXII deficiency Ø no differences in VTE or ischemic stroke
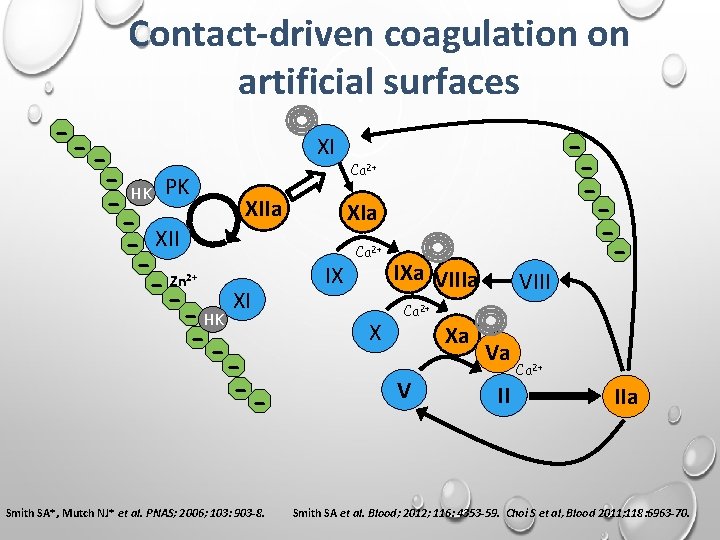
Contact-driven coagulation on artificial surfaces - -- HK PK XIIa - XII - Zn XI - HK --2+ Smith SA*, Mutch NJ* et al. PNAS; 2006; 103: 903 -8. XI Ca 2+ XIa IX Ca 2+ X IXa VIII - Ca 2+ Xa V Va II Ca 2+ IIa Smith SA et al. Blood; 2012; 116: 4353 -59. Choi S et al, Blood 2011; 118: 6963 -70.
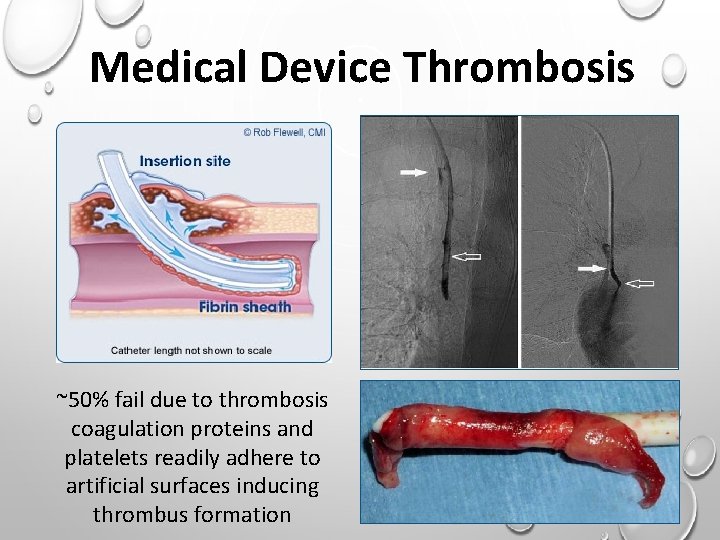
Medical Device Thrombosis ~50% fail due to thrombosis coagulation proteins and platelets readily adhere to artificial surfaces inducing thrombus formation
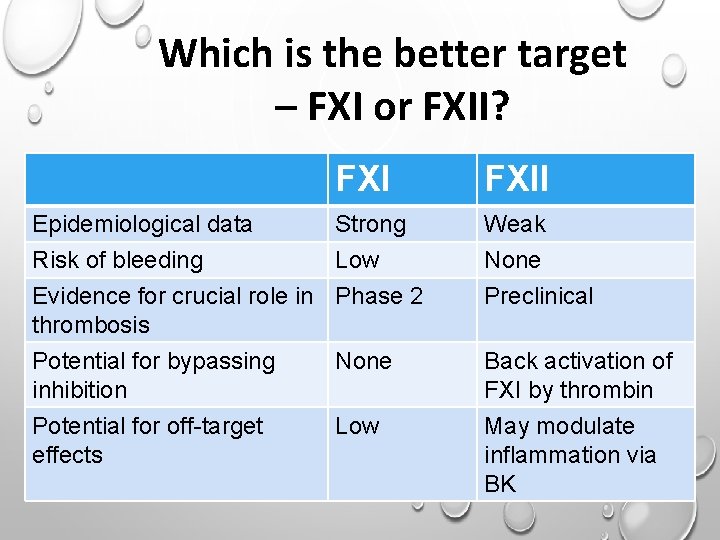
Which is the better target – FXI or FXII? Epidemiological data Risk of bleeding FXII Strong Low Weak None Evidence for crucial role in Phase 2 thrombosis Potential for bypassing None inhibition Potential for off-target Low effects Preclinical Back activation of FXI by thrombin May modulate inflammation via BK

Indicators for FXI & FXII targeted therapies Indicator Rationale Primary VTE prophylaxis Simple and safe single-dose regimens Extended VTE treatment Safer than current therapies for extended VTE treatment Prevention of ischemia Safer anticoagulant platform on top after ACS of antiplatelet therapy Medical devices Prevent clotting on heart valves,

Summary FXII- and FXI-directed anticoagulant strategies are a new era in anticoagulation that will minimise thrombosis with minimal risk of bleeding risk.
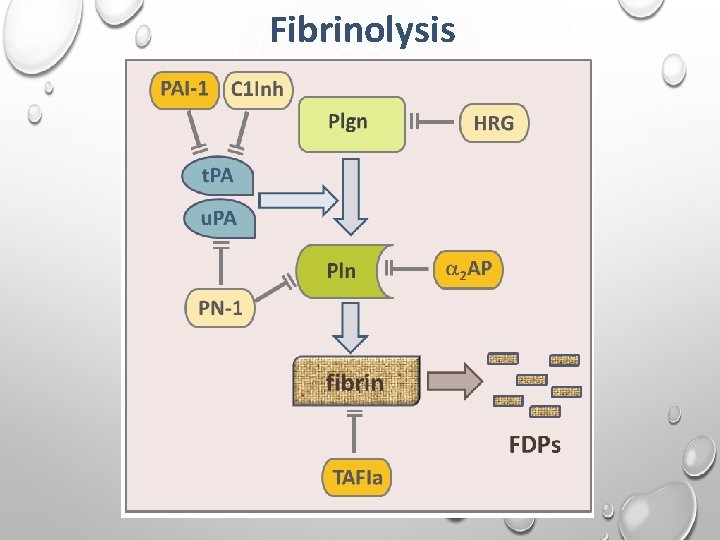
Fibrinolysis
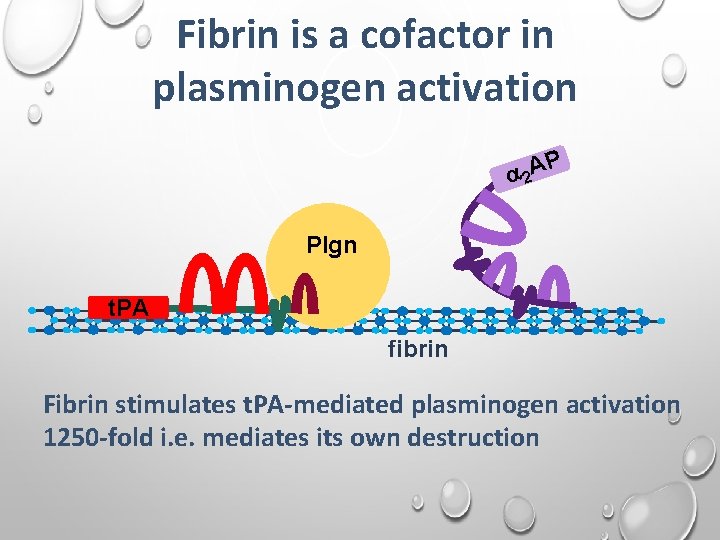
Fibrin is a cofactor in plasminogen activation P 2 A Plgn t. PA fibrin Fibrin stimulates t. PA-mediated plasminogen activation 1250 -fold i. e. mediates its own destruction
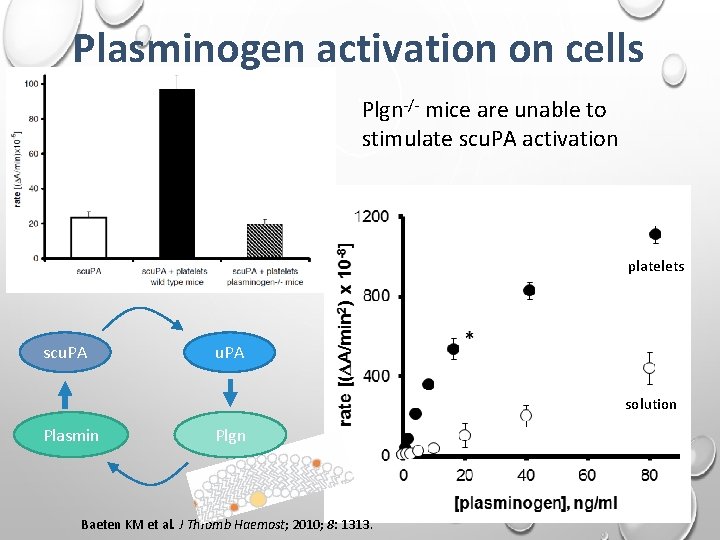
Plasminogen activation on cells Plgn-/- mice are unable to stimulate scu. PA activation platelets scu. PA solution Plasmin Plgn Baeten KM et al. J Thromb Haemost; 2010; 8: 1313.
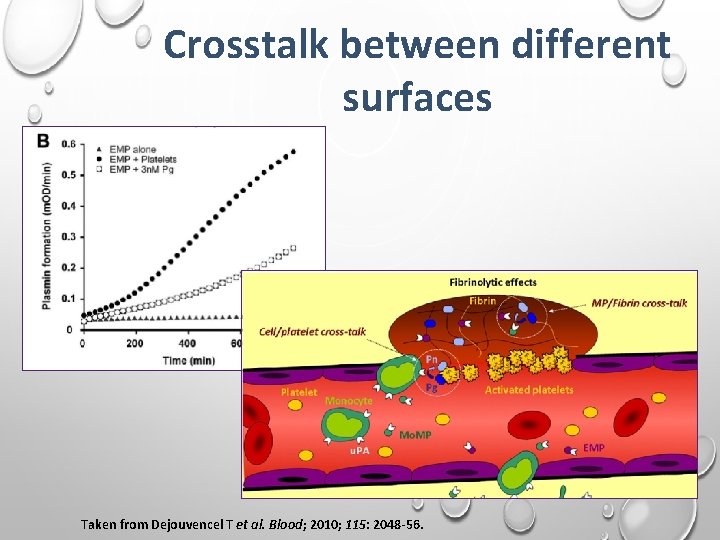
Crosstalk between different surfaces Taken from Dejouvencel T et al. Blood; 2010; 115: 2048 -56.
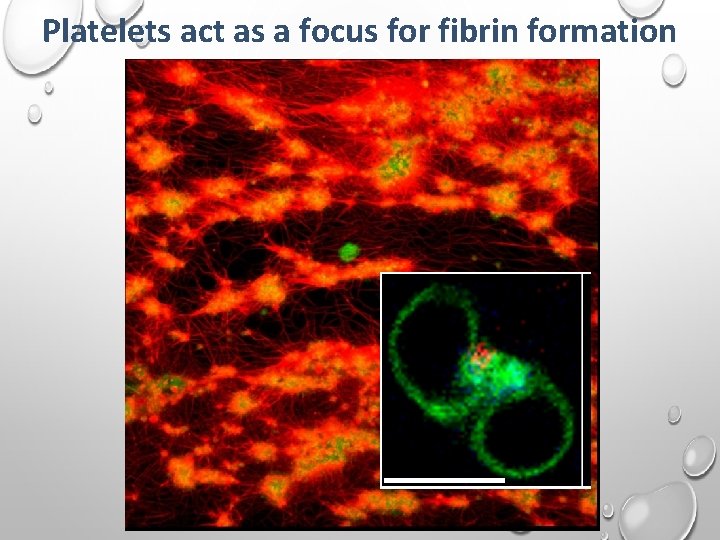
Platelets act as a focus for fibrin formation
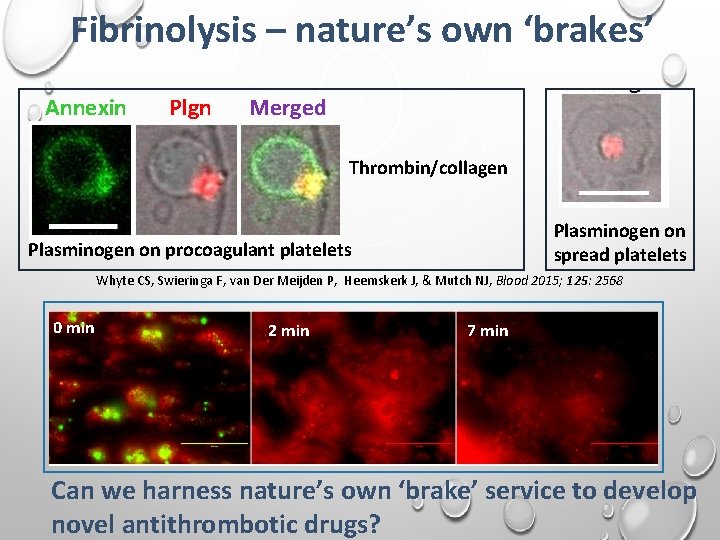
Fibrinolysis – nature’s own ‘brakes’ Annexin Plgn Merged Thrombin/collagen Plasminogen on spread platelets Plasminogen on procoagulant platelets Whyte CS, Swieringa F, van Der Meijden P, Heemskerk J, & Mutch NJ, Blood 2015; 125: 2568 0 min 2 min 7 min Can we harness nature’s own ‘brake’ service to develop novel antithrombotic drugs?
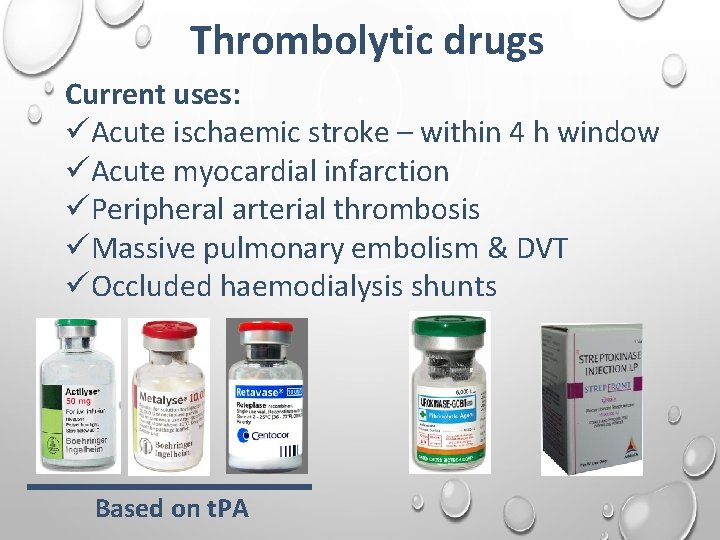
Thrombolytic drugs Current uses: üAcute ischaemic stroke – within 4 h window üAcute myocardial infarction üPeripheral arterial thrombosis üMassive pulmonary embolism & DVT üOccluded haemodialysis shunts Based on t. PA
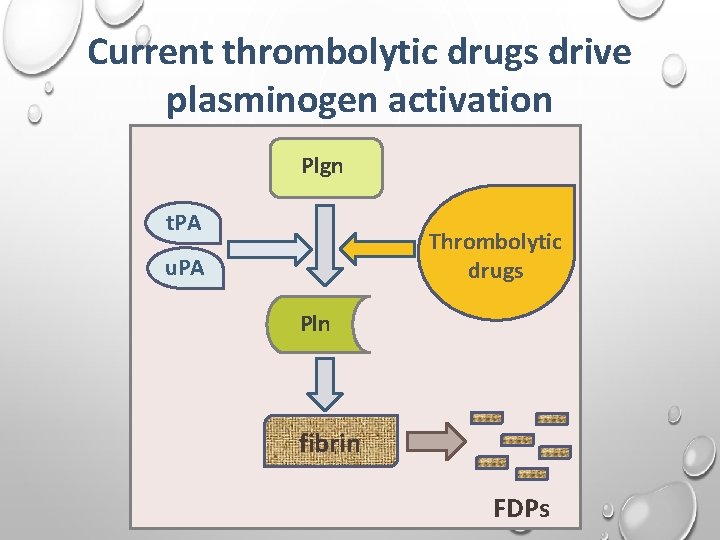
Current thrombolytic drugs drive plasminogen activation Plgn t. PA Thrombolytic drugs u. PA Pln fibrin FDPs
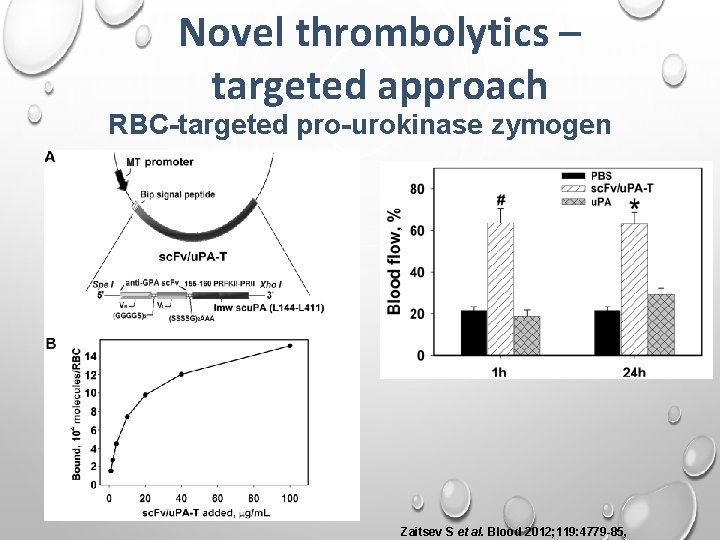
Novel thrombolytics – targeted approach RBC-targeted pro-urokinase zymogen Zaitsev S et al. Blood 2012; 119: 4779 -85,
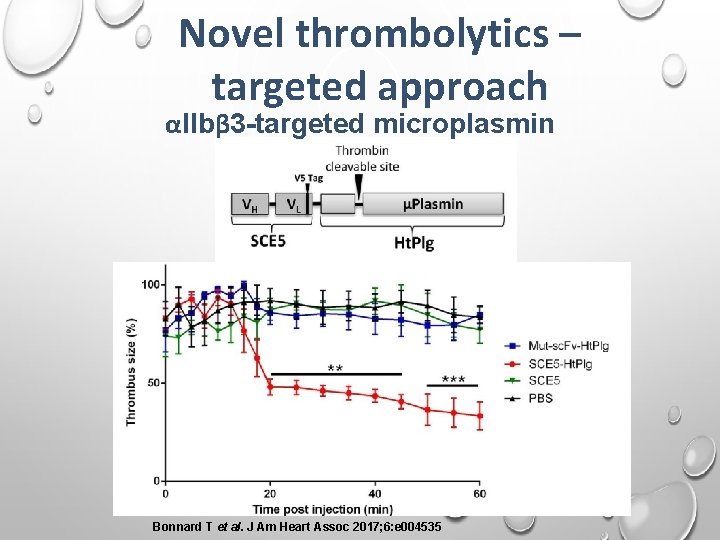
Novel thrombolytics – targeted approach αIIbβ 3 -targeted microplasmin Bonnard T et al. J Am Heart Assoc 2017; 6: e 004535
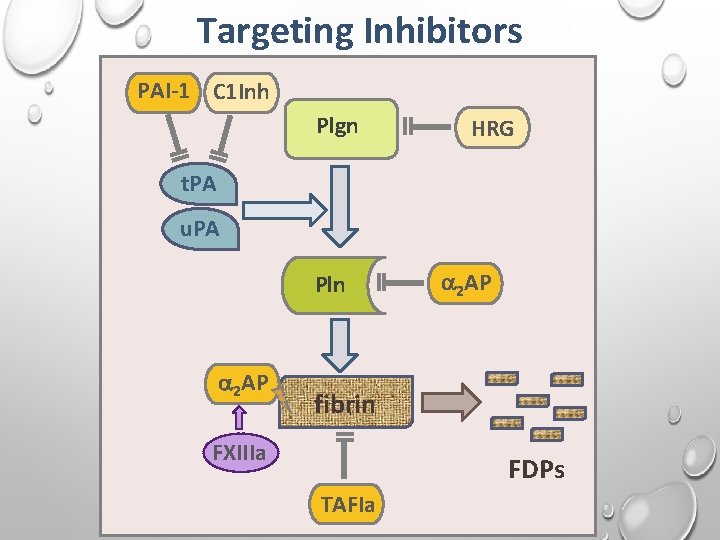
Targeting Inhibitors PAI-1 C 1 Inh Plgn HRG t. PA u. PA Pln 2 AP fibrin FXIIIa FDPs TAFIa

2 AP & FXIII deficient plasma lyse at strikingly similar rates TG inhibitor has no impact on lysis of 2 AP-depleted thrombi 2 AP ± TG inhibitor, P>0. 5 PNP + TG inhibitor, P<0. 005 PNP Fraser et al, 2011 Blood Lysis of FXIII and α 2 AP-depleted thrombi is normalized by PNP 2 AP depleted plasma FXIII depleted plasma
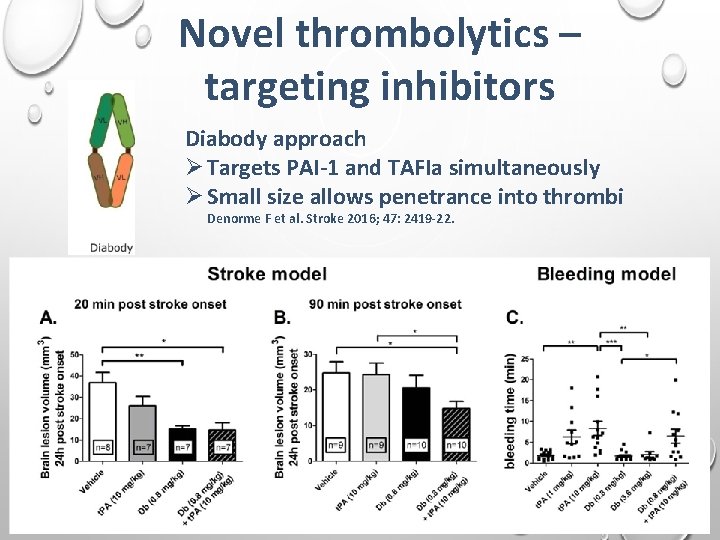
Novel thrombolytics – targeting inhibitors Diabody approach Ø Targets PAI-1 and TAFIa simultaneously Ø Small size allows penetrance into thrombi Denorme F et al. Stroke 2016; 47: 2419 -22.
Summary Current thrombolytics exhibit limited efficacy and are associated with bleeding complications. Novel therapies strive to target thrombi directly to facilitate clearance without bleeding.
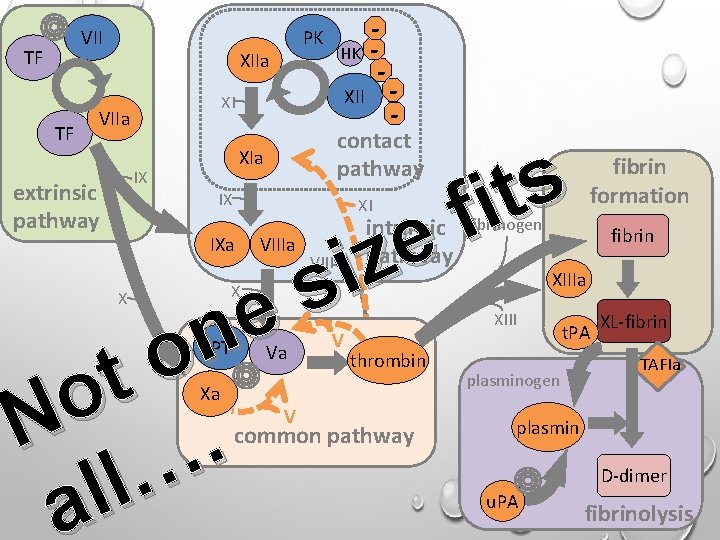
VII TF TF PK XIIa XII XI VIIa IX VIIIa e n o t o N. … l l a X PT Xa s t i f e z i s XI XI IXa X - contact pathway XIa IX extrinsic pathway HK - Va V VIII V intrinsic pathway fibrinogen common pathway fibrin XIIIa XIII thrombin fibrin formation t. PA plasminogen XL-fibrin TAFIa plasmin D-dimer u. PA fibrinolysis

Acknowledgements • • • Microscopy and Histology • Core Facility The Iain Fraser Cytometry • • Centre Prof Johan Heemskerk Frauke Swieringa Dr Paola van Der Meijden Tom Mastenbroek
- Slides: 34