ERT 313 BIOSEPARATION ENGINEERING Introduction to Bio Separation

ERT 313 : BIOSEPARATION ENGINEERING Introduction to Bio. Separation Process & Some Mechanical-Physical By; Mrs Hafiza Binti Shukor Separation Process ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Students should be able to; DEFINE and EXPLAIN the meaning, nature and application of Bioproducts and Bioseparation Engineering. DESCRIBE and REPEAT the Bioseparation techniques/processes and RIPP (Recovery, Isolation, Purification and Polishing) scheme. DEVELOP techniques/processes and RIPP (Recovery, Isolation, Purification and Polishing) scheme for downstream processes. DESCRIBE and DISC USS about some mechanicalphysical separation technique like cell disruption, centrifugation and electrophoresis ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

BIOPROD UCT? Chemical substances / combination of chemical substances that are made by LIVING THING range from methanol to whole cells. Derived by EXTRACTION from whole plants and animals By synthesis in bioreactors containing cells / enzymes ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

BIOLOGICAL PROD UCTS (with different classification) Biological products - chemical classification Solvents, e. g. ethanol, acetone, butanol Cells, e. g. bakers yeast, brewers yeast, freeze dried lactobacillus Crude cellular extracts, e. g. yeast extract, soy extracts Organics acids, e. g. citric acid, lactic acid, butyric acid Vitamins, e. g. ascorbic acid, vitamin B 12 Amino acids e. g. lysine, phenylalanine, glycine Gums and polymers, e. g. xanthan, gellan, dextran Antibiotics, e. g. penicillins, rifanpicin, streptomycin Proteins, e. g. industrial enzymes, egg proteins, milk proteins, whey protein therapeutic enzymes, monoclonal antibodies, plasma proteins Sugars and carbohydrates, e. g. glucose, fructose, starch, dextran Lipids, e. g. glycerol, fatty acids, steroids Nucleic acids, e. g. plasmids, therapeutic DNA ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Biological products - applications Industrial chemicals, e. g. solvents, organic acids, industrial enzymes Agrochemicals, e. g. biofertilizers, biopesticides Pharmaceuticals, e. g. antibiotics, hormones, monoclonal antibodies, plasma proteins, vaccines Food and food additives, e. g. whey proteins, milk proteins, egg proteins, soy proteins Nutraceuticals, e. g. vitamins, amino acids, purified whey proteins Diagnostic products, e. g. glucose oxidase, peroxidase Commodity chemicals, e. g. detergent enzymes, insecticides Laboratory reagents, e. g. bovine serum albumin, ovalbumin, lysozyme Cosmetic products, e. g. plant extracts, animal tissue extracts ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

BIOLOGICAL PROD UCTS (with different bioseparation process) Product Nature of bioseparation required Alcoholic beverages: Beer, wine, spirits Clarification, distillation Organic acids: Acetic acid, citric acid Precipitation, filtration, adsorption, solvent extraction Vitamins: Vitamin C, vitamin B 12, riboflavin Precipitation, filtration, adsorption, solvent extraction Amino acids: Lysine, glycine, phenylalanine Precipitation, filtration, adsorption, solvent extraction Antibiotics: Penicillins, neomycin, bacitracin Precipitation, filtration, adsorption, solvent extraction Carbohydrates: Starch, sugars, dextrans Precipitation, filtration, adsorption Lipids: Glycerol, fats, fatty acids Precipitation, filtration, adsorption, solvent extraction ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
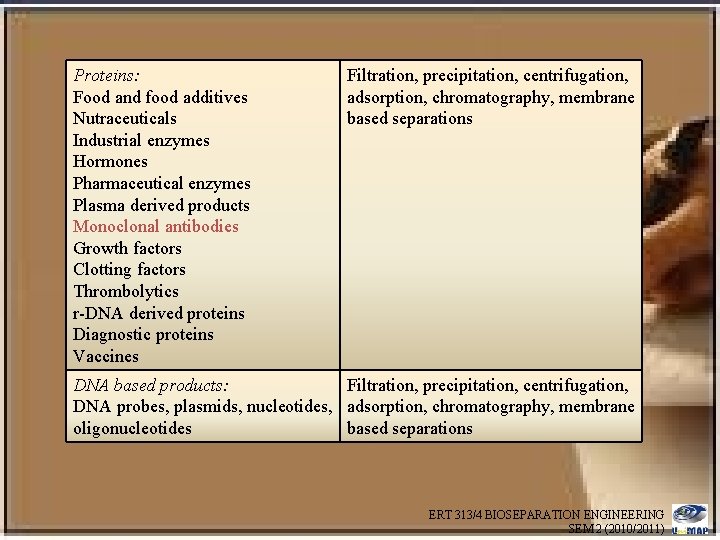
Proteins: Food and food additives Nutraceuticals Industrial enzymes Hormones Pharmaceutical enzymes Plasma derived products Monoclonal antibodies Growth factors Clotting factors Thrombolytics r-DNA derived proteins Diagnostic proteins Vaccines Filtration, precipitation, centrifugation, adsorption, chromatography, membrane based separations DNA based products: Filtration, precipitation, centrifugation, DNA probes, plasmids, nucleotides, adsorption, chromatography, membrane oligonucleotides based separations ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

OVERVIEW OF BIOPROCESSING OF BIOCHEMICAL & PHARMACE UTICAL PRODUCTS q One of the major segments within biotechnology where R&D is bioprocessing which deals with the manufacture of biochemicals, food, neutraceuticals and agrochemicals. q New biologically derived product have been developed, approved and licensed. q Eg: Monoclonal antibodies (used for treatment of canser) ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

OVERVIEW OF BIOPROCESSING OF BIOCHEMICAL & PHARMACE UTICAL PROD UCTS q all biochemicals & pharmaceutical product MUST be extensively P U RIFIED before used in respective application. q. Bioprocessing / downstream processing of biochemical & pharmaceuticals products refer to the SYSTEMATIC study of the scientific and engineering principle utilized for the large scale purification of biological products. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

OVERVIEW OF BIOPROCESSING OF BIOCHEMICAL & PHARMACE UTICAL PROD UCTS 2 categories of bioprocessing : i) reactive bioprocessing -bio-separation process follows some form of biological reactions ii)extractive bioprocessing -almost entirely involves bioseparation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
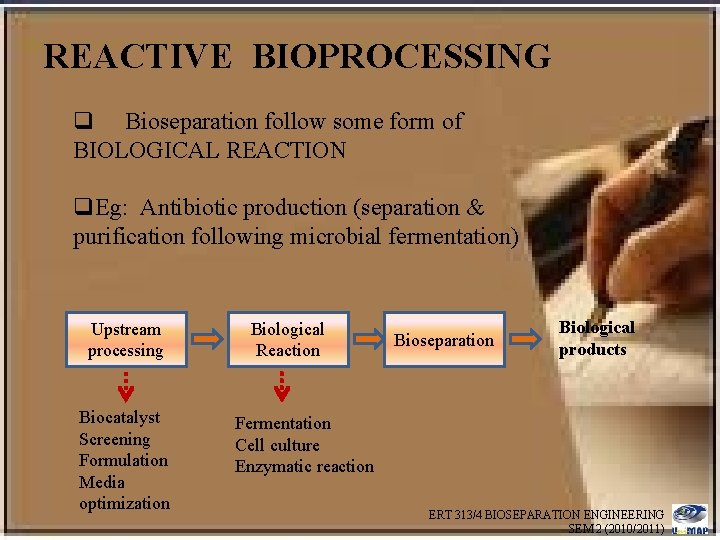
REACTIVE BIOPROCESSING q Bioseparation follow some form of BIOLOGICAL REACTION q. Eg: Antibiotic production (separation & purification following microbial fermentation) Upstream processing Biocatalyst Screening Formulation Media optimization Biological Reaction Bioseparation Biological products Fermentation Cell culture Enzymatic reaction ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
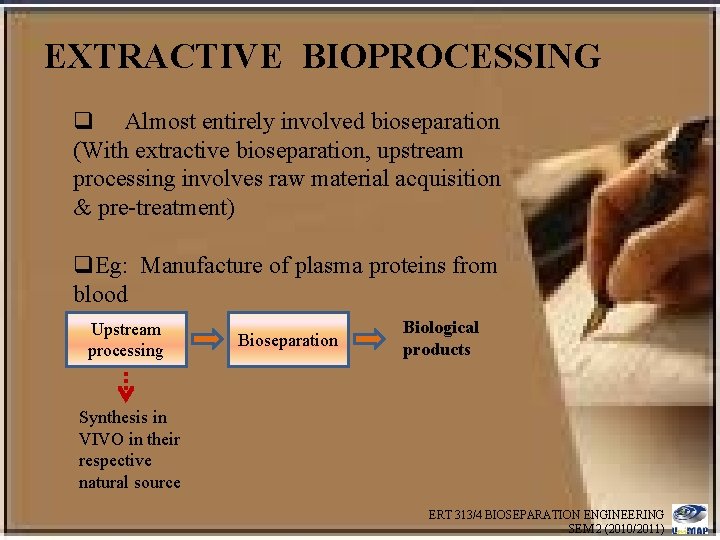
EXTRACTIVE BIOPROCESSING q Almost entirely involved bioseparation (With extractive bioseparation, upstream processing involves raw material acquisition & pre-treatment) q. Eg: Manufacture of plasma proteins from blood Upstream processing Bioseparation Biological products Synthesis in VIVO in their respective natural source ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Bioseparations engineering • Definition: Recovery, isolation, purification and polishing of products synthesized by biotechnological processes • Extended definition: Final polishing steps of processes such as biotechnology based effluent treatment and water purification Bioproduct/s Upstream processing Bioreaction Downstream processing Impurities ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Why do we need bioseparation? • • Enrichment of target product Reduction in bulk Removal of specific impurities Enhancement of product stability Achievement of product specifications Prevention of product degradation Prevention of catalysis other than the type desired Prevention of catalyst poisoning ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Challenges in Bioseparations Engineering • Low product concentration • Large number of impurities, • Thermolabile bioproducts. • Narrow operating p. H and ionic strength window • Shear sensitivity of bioproducts • Low solubility of bioproducts in organic solvents • Instability of bioproducts in organic solvents • Stringent quality requirements • Percentage purity • Absence of specific impurities An ideal bioseparation process should combine high throughput with high selectivity, and should ensure stability of product. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

A Good Bioseparation Process: • • • Ensures desired purity of product Ensures stability of product Keeps cost low Is reproducible Is scalable Meets regulatory guidelines ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Economic Importance of Bioseparation Engineering The purification of biological product from their respective starting material. Eg: cell culture media : technically difficult and expensive The critical limiting factor in the commercialization of biological product Many cases, bioseparation cost can be a substantial component of the total cost of bioprocessing ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
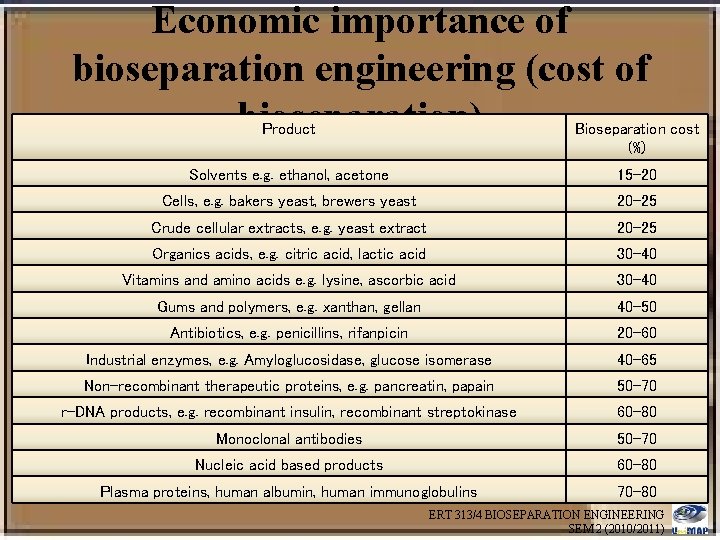
Economic importance of bioseparation engineering (cost of bioseparation) Product Bioseparation cost (%) Solvents e. g. ethanol, acetone 15 -20 Cells, e. g. bakers yeast, brewers yeast 20 -25 Crude cellular extracts, e. g. yeast extract 20 -25 Organics acids, e. g. citric acid, lactic acid 30 -40 Vitamins and amino acids e. g. lysine, ascorbic acid 30 -40 Gums and polymers, e. g. xanthan, gellan 40 -50 Antibiotics, e. g. penicillins, rifanpicin 20 -60 Industrial enzymes, e. g. Amyloglucosidase, glucose isomerase 40 -65 Non-recombinant therapeutic proteins, e. g. pancreatin, papain 50 -70 r-DNA products, e. g. recombinant insulin, recombinant streptokinase 60 -80 Monoclonal antibodies 50 -70 Nucleic acid based products 60 -80 Plasma proteins, human albumin, human immunoglobulins 70 -80 ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Strategies for Bioseparation A large number of bioseparation methods are available The strategy is based on how best these can be utilized for a given separation The following need to be taken into account: • The volume of process stream • The relative abundance of the product in this process stream • The intended use of the product, i. e. purity requirements • The cost of the product • Stability requirements ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Conventional strategy: The RIPP scheme • Recovery, isolation, purification and polishing • Based on a logical arrangement of bioseparation methods • Low-resolution (less selectivity), high-throughput (product) techniques (e. g. precipitation, filtration, centrifugation, crystallization) are first used for recovery and isolation • High-resolution techniques (e. g. adsorption, chromatography, electrophoresis) are then used for purification and polishing ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Conventional strategy: The RIPP scheme Biotechnological processes usually yield products at very low concentrations in the product stream. The product of interest also needs to be separated from a large number of impurities, some of which have physical and chemical properties not too different from the product. If such a product stream is sent directly to a highresolution separation device, it will soon be overwhelmed and fail to function properly. Therefore it makes sense to use high throughput-low resolution techniques first to significantly reduce the volume/concentration of process stream. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Conventional strategy: The RIPP scheme These high throughput-low resolution steps are referred to as recovery and isolation steps. The processed product stream is then further processed by high resolution-low throughput steps to get pure finished products. With the advent of membrane separation processes it is now possible to replace the conventional RIPP scheme. Membrane processes give high throughput and can be fine-tuned/optimized to give very high resolution. The use of membrane technology reduces the number of bioseparation steps and hence contributes towards high product recovery. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

The RIPP Scheme • Multi-technique separation • Process design should take into consideration the following: • The nature of starting material • The initial location of the target product • The volume of process stream • The relative abundance of the product in the starting material • The susceptibility to degradation of the product • The desired physical form of the final product • The quality requirements • Costing ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Commonly Used Bioseparation Processes Low resolution-high throughput • • • Cell disruption Precipitation Centrifugation Liquid-liquid extraction Leaching Filtration Supercritical fluid extraction Microfiltration Dialysis High resolution-low throughput • • • Ultracentrifugation Adsorption Packed bed chromatography Affinity separation Electrophoresis High resolution-high throughput • Ultrafiltration • Fluidized bed chromatography • Membrane chromatography ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Objective & Typical Unit Operations of The 4 Stages in Bioseparation Stage Objectives Typical unit operation Recovery (separation of insoluble) Remove/ collect cells, cells debris /other particulate. Reduce volume Filtration, sedimentation, extraction, adsorption Isolation of product Remove material have properties widely different from those desired in product. Reduce volume Extraction, adsorption, ultrafiltration, precipitation Purification Remove remaining impurities which typically similar to desired product in chemical functionality & physical properties. Chromatography, affinity method, functional precipitation Polishing Remove liquid. Convert product to crystallized form Drying, crystallization ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Nature of Bioseparation • Largely based on chemical separation techniques • Chemical separation techniques are modified based on specific requirements • Novel separations may be necessary in some cases • High throughput/productivity • High selectivity • Need to satisfy stringent quality requirements • Need to take into account degradable material • Low temperature operations ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Different Attributes of Bioseparation • Dilute biological products • Impurities & by-product also present • Stringent require for product (quality requirement) • Susceptible to denaturation & degradation • Thermo bile • Multi technique separation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Basis of Separation Biological products are separated based on one or several of the following in combination: • Size, e. g. filtration, membrane separation, gelfiltration, centrifugation • Density, e. g. centrifugation, sedimentation, flotation • Diffusivity, e. g. membrane separation, supercritical fluid extraction • Shape, e. g. centrifugation, filtration, sedimentation • Polarity, e. g. extraction, chromatography, adsorption • Solubility, e. g. extraction, membrane separation, precipitation, crystallization • Electrostatic charge, e. g. adsorption, membrane separation • Mobility, e. g. electrophoresis, membrane separation • Volatility e. g. distillation, membrane distillation, pervaporation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
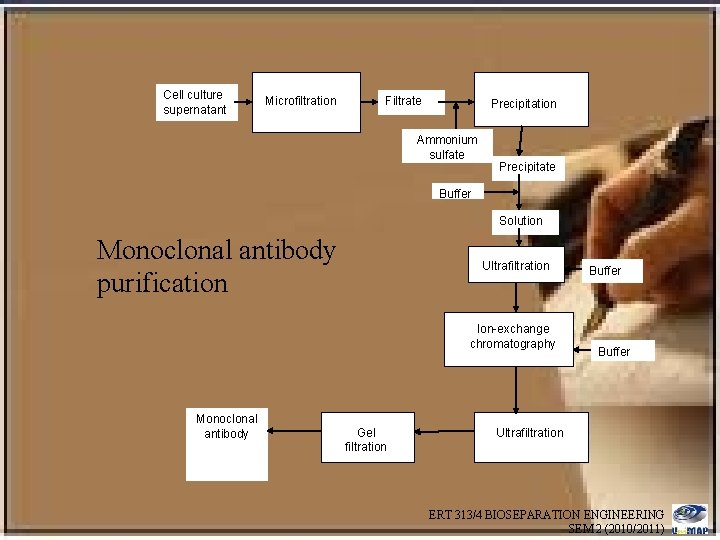
Cell culture supernatant Microfiltration Filtrate Precipitation Ammonium sulfate Precipitate Buffer Solution Monoclonal antibody purification Ultrafiltration Ion-exchange chromatography Monoclonal antibody Gel filtration Buffer Ultrafiltration ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

HOW TO CHOOSE SEPARATION METHOD 1. What is the product? 2. What is the value of product? 3. What is the acceptable product quality for the proposed end use? 4. Where is the product in the complex mixture? 5. What are the physical and chemical properties of the product and the impurities? 6. Is the product stable? 7. What are the economic of the various isolation procedure? 8. Are they any contamination / health risk? 9. Can the isolation procedure be scaled up? ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

RULES OF THUMB FOR BIOSEPARATIONS 1. Keep the number of step to a minimum. 2. Select the component that is easiest to remove first 3. Leave the most difficulty isolation step for last ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Current Paradigm in The Bioseparation Replacement of the conventional RIPP scheme by using new techniques which can significantly cut down the number of steps needed to bioseparation Some of these new and emerging techniques are: • Membrane chromatography • Expanded-bed chromatography • High-resolution ultrafiltration ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

CELL DISR UPTION ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Cell disruption / lysis is a method or process for releasing biological molecules from inside a cell (breaking / lysing cells and tissues) Biotechnological products produced by different types of cells can be intracellular or extracellular. If these are intracellular (inside the cell), the cells have to be disrupted to release these products before further separation can take place. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
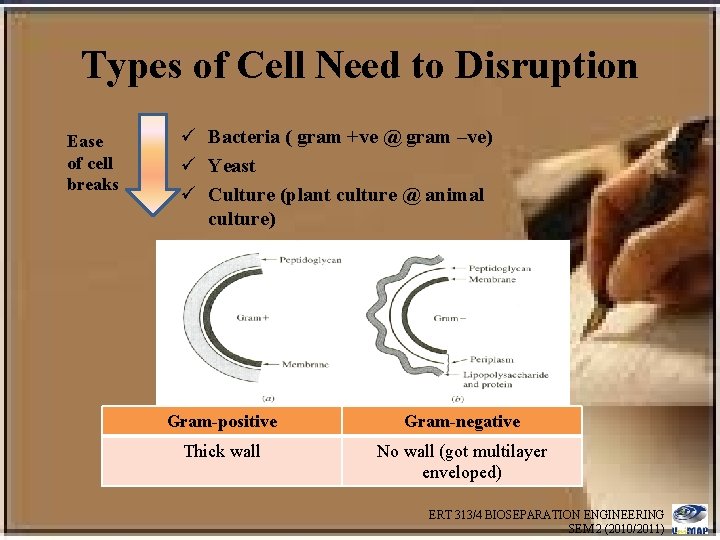
Types of Cell Need to Disruption Ease of cell breaks ü Bacteria ( gram +ve @ gram –ve) ü Yeast ü Culture (plant culture @ animal culture) Gram-positive Gram-negative Thick wall No wall (got multilayer enveloped) ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Some Elements of Cell Structure Prokaryotic Cells • Cells that do not contain a membrane-enclosed nucleus. • The bacteria cell envelope consists of an inner plasma membrane that separates all contents of the cell from the outside world, a peptidoglycan cell wall, and outer membrane • Bacteria cells with a very thick cell wall stain with crystal violet (Gram stain) and are called “Gram positive”, while those with thin cell wall stain very weakly – “Gram negative” ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Some Elements of Cell Structure Eukaryotic cells • Eukaryotic cells (cells with nuclei and internal organelles) are considerably more complicated than prokaryotic cells, and bioproducts may have to released from intracellular particles that are themselves coated with membranes and/or consist of large macromolecular aggregates • The eukraryotes includes fungi, and, of course, the higher plants and animals • The cell membrane of animal cells is easily broken, whereas the cell wall of plants is strong and relatively difficult to break ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
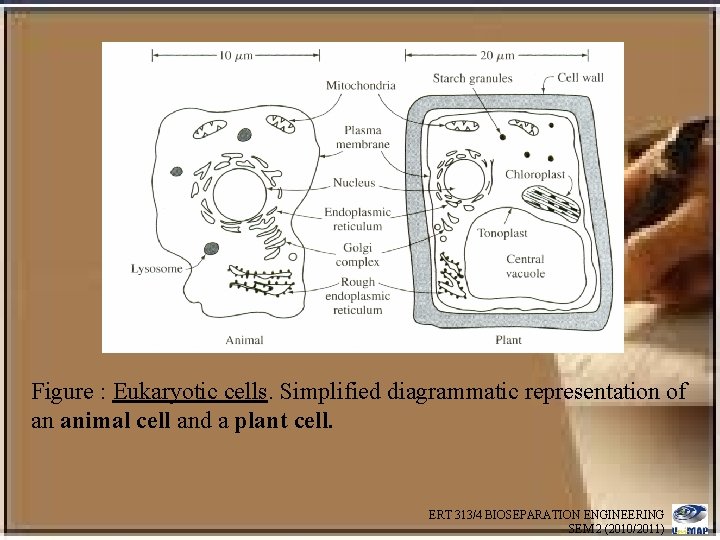
Figure : Eukaryotic cells. Simplified diagrammatic representation of an animal cell and a plant cell. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Different cell disruption techniques are used. These include: Physical methods • Disruption in ball mill or pebble mill • Disruption using a colloid mill • Disruption using French press • Disruption using ultrasonic vibrations Chemical methods • Disruption using detergents • Disruption using enzymes e. g. lysozyme • Combination of detergent and enzyme • Disruption using solvents ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
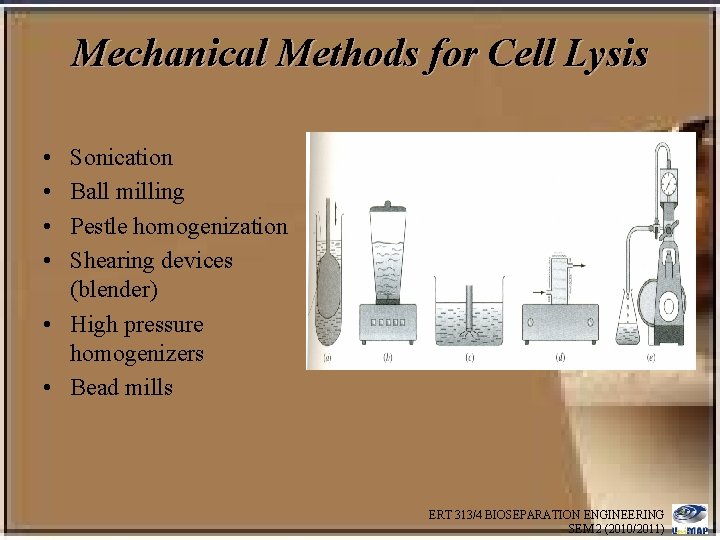
Mechanical Methods for Cell Lysis • • Sonication Ball milling Pestle homogenization Shearing devices (blender) • High pressure homogenizers • Bead mills ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Bead mill Cascading beads Rolling beads Cells being disrupted • Disruption takes place due to the grinding action of the rolling beads and the impact resulting from the cascading ones • Bead milling can generate substantial heat • Application: Yeast, animal and plant tissue • Small scale: Few kilograms of yeast cells per hour • Large scale: Hundreds of kilograms per hour. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Colloid mill Rotor Disrupted cells Cell suspension Stator • Typical rotation speeds: 10, 000 to 50, 000 rpm • Mechanism of cell disruption: High shear and turbulence • Application: Tissue based material • Single or multi-pass operation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
Separation of cells and medium • Recovery of cells and/or medium (clarification) – For intracellular enzyme, the cell fraction is required – For extracellular enzymes, the culture medium is required • On an industrial scale, cell/medium separation is almost always performed by centrifugation – Industrial scale centrifuges may be batch, continuous, or continuous with ERT 313/4 BIOSEPARATION ENGINEERING desludging SEM 2 (2010/2011)

CENTRIF UGATION ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

A centrifuge is used for separating particles from a solution according to their size, shape, density and viscosity of the medium by the application of an artificially induced gravitational field. In bioprocesses, these particles could be cells, sub cellular components, viruses and precipitated forms of proteins and nucleic acids. Centrifugation can be used to separate cells from a culture liquid, cell debris from a broth, and a group of precipitates. Centrifugation may be classified into two types: • Analytical centrifugation • Preparative Centrifugation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
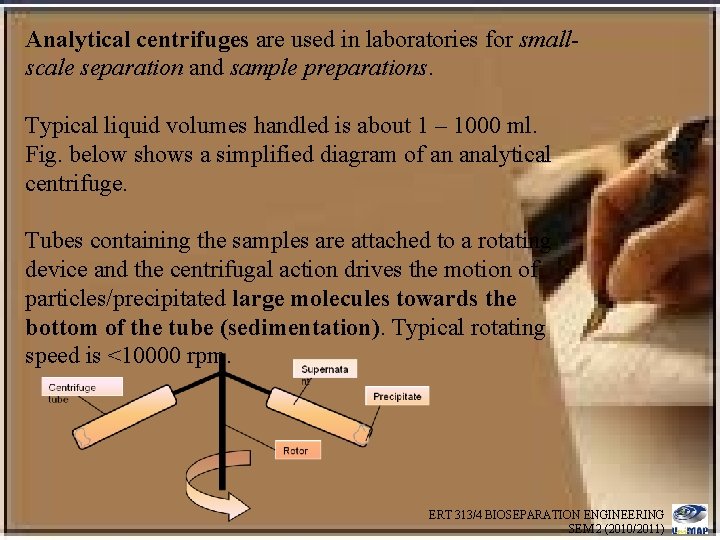
Analytical centrifuges are used in laboratories for smallscale separation and sample preparations. Typical liquid volumes handled is about 1 – 1000 ml. Fig. below shows a simplified diagram of an analytical centrifuge. Tubes containing the samples are attached to a rotating device and the centrifugal action drives the motion of particles/precipitated large molecules towards the bottom of the tube (sedimentation). Typical rotating speed is <10000 rpm. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Industrial centrifuges Tubular Bowl Centrifuge • Most useful for solid-liquid separation with enzymatic isolation • Can achieve excellent separation of microbial cells and animal, plant, and most microbial cell debris in solution Disc Bowl Centrifuge • Widely used for removing cells and animal debris • Can partially recover microbial cell debris and protein precipitates ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Perforate Bowl Basket Centrifuge • Exception at separation of adsorbents, such as cellulose and agarose Zonal Ultracentrifuge • Applied in the vaccine industry because it can easily remove cell debris from viruses • Can collect fine protein precipitates • Has been used experimentally to purify RNA polymerase and very fine debris in enzymes ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Properties of industrial centrifuges • Tube – High centrifugal force – Limited solids capacity – Good dewatering – Difficult to recover protein – Easy to clean • Chamber – Large solids capacity – No solids discharge – Cleaning difficult – Good dewatering – Bowl cooling possible – Solids recovery difficult • Disc type – Poor dewatering – Solids discharge – Difficult to clean – No foaming – Bowl cooling possible ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
Centrifugation properties of different cell types • Bacteria – Small cell size – Resilient – High speed required – Low cell damage • Yeast cells – Large cells – Resilient – Lower speed required – Low cell damage • Filamentous fungi – Mycelial – Resilient – Lower speed required – High water retention in pellet • Cultured animal cells – Large cells – Very fragile – Very susceptible to damage ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Forced Developed in Centrifugal Separation 1. Introductions • Centrifugal separators use the common principal that an object whirled about an axis or center point a constant radial distance from the point is acted on by a force • The object is constantly changing direction and is thus accelerating, even though the rotational speed is constant • This centripetal force acts in a direction toward the center of rotation ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Forced Developed in Centrifugal Separation • In cylindrical container, the contents of fluid and solids exert an equal and opposite force, called centrifugal force, outward to the walls of the container • This cause the settling or sedimentation of particles through a layer of liquid or filtration of a liquid through a bed of filter cake held inside a perforated rotating chamber ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
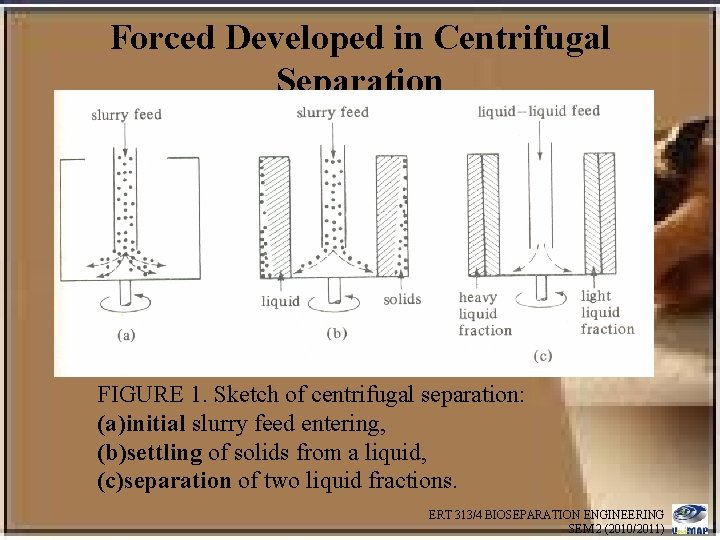
Forced Developed in Centrifugal Separation FIGURE 1. Sketch of centrifugal separation: (a)initial slurry feed entering, (b)settling of solids from a liquid, (c)separation of two liquid fractions. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

• In Fig. 1 a a cylindrical bowl is shown rotating, with a slurry feed of solid particles and liquid being admitted at the center. • The feed enters and is immediately thrown outward to the walls of the container as in Fig. 1 b • The liquid and solids are now acted upon by the vertical and the horizontal centrifugal forces • The liquid layer then assumes the equilibrium position, with the surface almost vertical • The particles settle horizontally outward and press against the vertical bowl wall ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
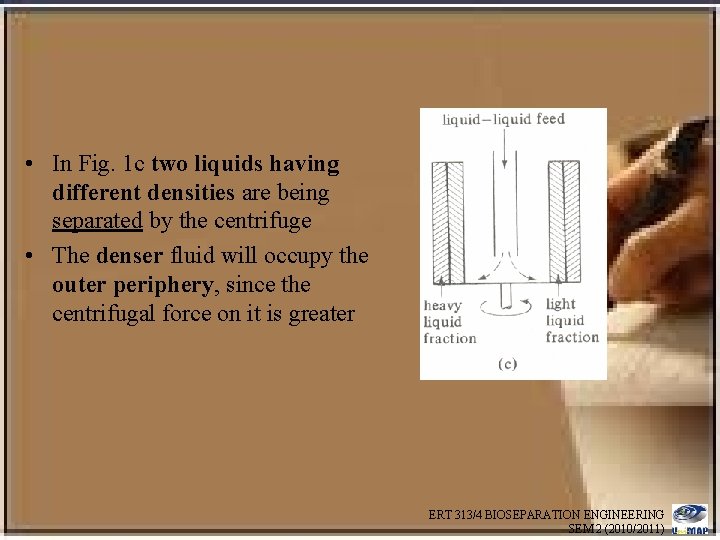
• In Fig. 1 c two liquids having different densities are being separated by the centrifuge • The denser fluid will occupy the outer periphery, since the centrifugal force on it is greater ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Equations for centrifugal force • In circular motion the acceleration due to the centrifugal force is (1) • The centrifugal force Fc in N (lbf) acting on the particle is given by (2) where gc = 32. 174 lbm·ft /lbf • s 2 • Since ω= v/r, where v is the tangential velocity of the particle in m/s (ft/s) (3) ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

• Often rotational speeds are given as N (rev/min) (4) (5) • Substituting Eq. (4) into Eq. (2), (6) • The gravitational force on a particle is (a) • In terms of gravitational force, the centrifugal force is: (7) ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Example 1 A centrifuge having a radius of the bowl of 0. 1016 m (0. 333 ft) is rotating at N = 1000 rev/min. a) Calculate the centrifugal force developed in terms of gravity forces. (b) Compare this force to that for a bowl with a radius of 0. 2032 m rotating at the same rev/mm. ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

Solution: For part (a), r = 0. 1016 m and N = 1000. Substituting into Eq. (7), For part (b), r = 0. 2032 m. Substituting into Eq. (7), ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)

ERT 313/4 BIOSEPARATION ENGINEERING SEM 2 (2010/2011)
- Slides: 60