Equilibrium The Extent of Chemical Reactions 17 1

Equilibrium: The Extent of Chemical Reactions 17 -1

Equilibrium: The Extent of Chemical Reactions The Equilibrium State and the Equilibrium Constant The Reaction Quotient and the Equilibrium Constant Expressing Equilibria with Pressure Terms: Relation between Kc and Kp Comparing Q and K to Determine Reaction Direction How to Solve Equilibrium Problems Reaction Conditions and Equilibrium: Le Châtelier’s Principle 17 -2

Many Chemical reactions are Reversible Reactions • Photochromic sunglass: Ag. Br(clear) + hv ⇌Ag(dark) + Cl • Temperature dependent coloration: N 2 O 4 + heat ⇌ 2 NO 2 • Acid base indicators • Rechargeable battery 17 -3 3

Reversible Reaction All reactions are reversible. For example: Decomposition of dinitrogen tetroxide: N 2 O 4 2 NO 2 Starting from pure N 2 O 4, NO 2 will form as product form decomposition of dinitrogen tetroxide Starting from pure NO 2 , N 2 O 4 will form as product from combination of nitrogen dioxide 2 NO 2 N 2 O 4 17 -4
![The Equilibrium State Starting from pure reactant, as reaction progresses, [reactant] ↓, [product] ↑ The Equilibrium State Starting from pure reactant, as reaction progresses, [reactant] ↓, [product] ↑](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-5.jpg)
The Equilibrium State Starting from pure reactant, as reaction progresses, [reactant] ↓, [product] ↑ Therefore rateforward ↓ , ratereversed ↑ • When rateforward = ratereversed, [product] and [reactant] remain constant, the reaction reaches equilibrium Chemical equilibrium is a dynamic state, meaning: • Forward and reverse reactions continue to occur • Forward and reverse reaction at the same rate • No net change is observed on the macroscopic level 17 -5
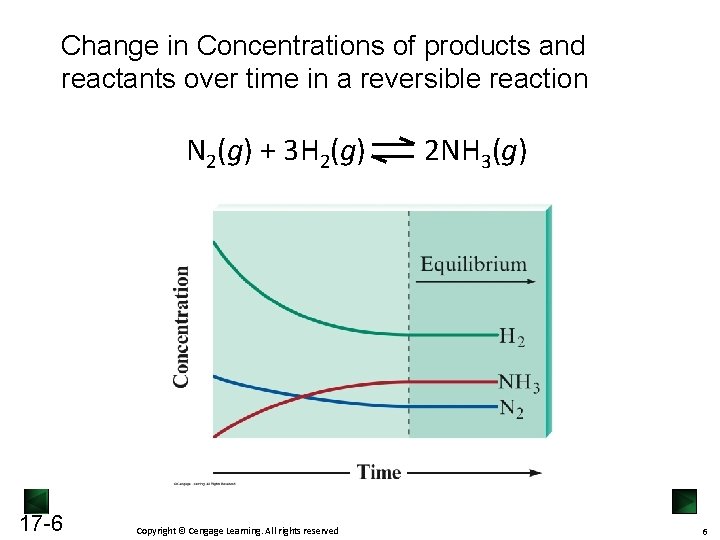
Change in Concentrations of products and reactants over time in a reversible reaction N 2(g) + 3 H 2(g) 17 -6 Copyright © Cengage Learning. All rights reserved 2 NH 3(g) 6
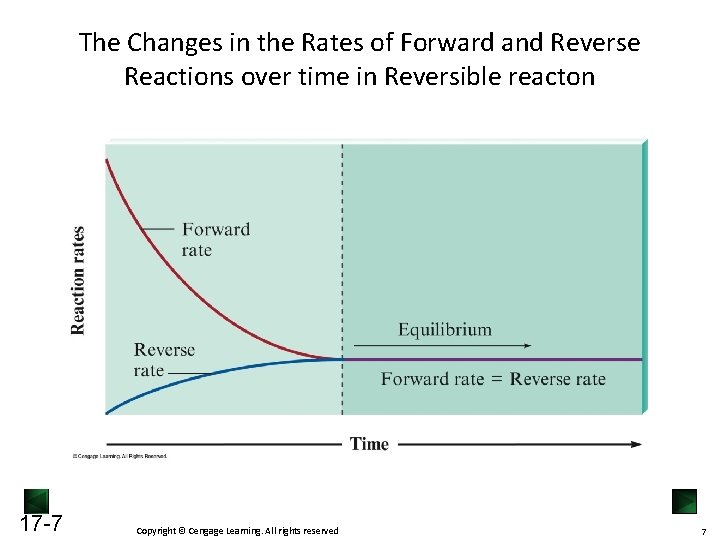
The Changes in the Rates of Forward and Reverse Reactions over time in Reversible reacton 17 -7 Copyright © Cengage Learning. All rights reserved 7
![At equilibrium: Both [Reactant] and [Products] remain constant; they co-exist 17 -8 At equilibrium: Both [Reactant] and [Products] remain constant; they co-exist 17 -8](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-8.jpg)
At equilibrium: Both [Reactant] and [Products] remain constant; they co-exist 17 -8
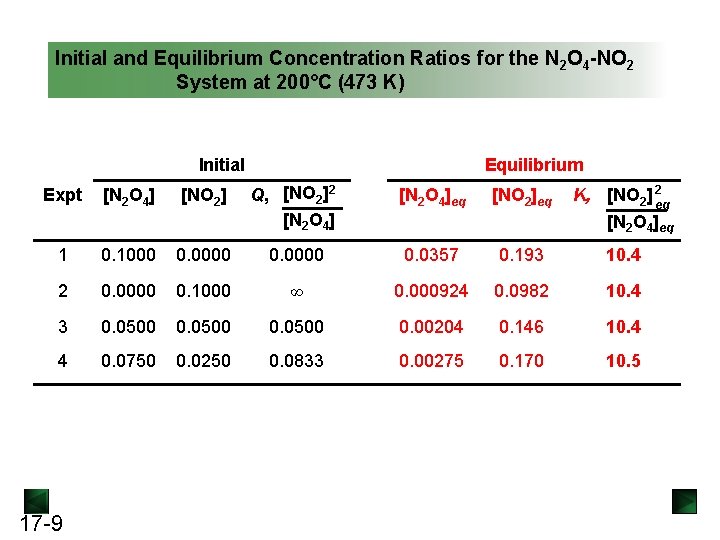
Initial and Equilibrium Concentration Ratios for the N 2 O 4 -NO 2 System at 200°C (473 K) Initial Equilibrium Expt [N 2 O 4] [NO 2] Q, [NO 2]2 [N 2 O 4]eq [NO 2]eq K, [NO 2] 2 eq [N 2 O 4]eq 1 0. 1000 0. 0000 0. 0357 0. 193 10. 4 2 0. 0000 0. 1000 ∞ 0. 000924 0. 0982 10. 4 3 0. 0500 0. 00204 0. 146 10. 4 4 0. 0750 0. 0250 0. 0833 0. 00275 0. 170 10. 5 17 -9
![Equilibrium Constant Equilibrium constant K : a particular ratio of [product] over [reactant] at Equilibrium Constant Equilibrium constant K : a particular ratio of [product] over [reactant] at](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-10.jpg)
Equilibrium Constant Equilibrium constant K : a particular ratio of [product] over [reactant] at equilibrium For the general reaction K depends only on temperature, not affected by concentrations of reactant or product 17 -10

Example: Determine K from equilibrium concentrations Formation of ammonia is a reversible reaction: N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g) At equilibrium, [N 2] = 0. 12 M, [H 2] = 0. 25 M and [NH 3] = 0. 21 M. Find the equilibrium constant. 17 -11 24

Magnitude of K: the extent of reaction K reflects the extent of a reaction, i. e. , how far a reaction proceeds towards the products at a given temperature. Small K (<< 1): the reaction yields ______ product when reaching equilibrium. The reaction favors the reactants. K ~10 -4 for equilibrium HF(aq) ⇌ H+(aq) + F-(aq) Large K (>> 1): the reaction reaches equilibrium with very ______ reactant remaining. The reaction favors the products. K ~106 for equilibrium HCl(aq) ⇌ H+(aq) + Cl-(aq) 17 -12
![Reaction Quotient Q For the general reaction [C]c[D]d the reaction quotient Q = [A]a[B]b Reaction Quotient Q For the general reaction [C]c[D]d the reaction quotient Q = [A]a[B]b](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-13.jpg)
Reaction Quotient Q For the general reaction [C]c[D]d the reaction quotient Q = [A]a[B]b Meaning of Q : the ratio of product concentrations to reactant concentrations at any point in a reaction. At equilibrium: Q = K For a particular system and temperature, the same equilibrium state is attained regardless of starting concentrations. 17 -13

Practice: Writing the Reaction Quotient from the Balanced Equation PROBLEM: Write the reaction quotient, Q, for each of the following reactions: (a) N 2 + 3 H 2 ⇌ 2 NH 3 17 -14
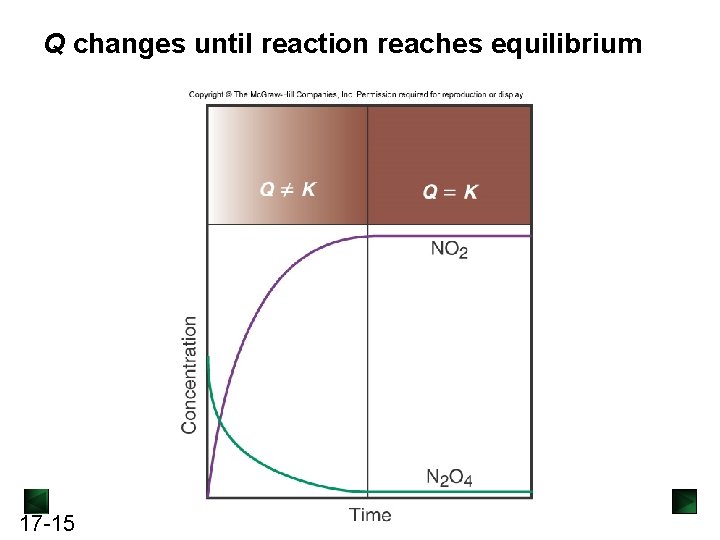
Q changes until reaction reaches equilibrium 17 -15
![Q vs. K: Determining the Direction of Reaction Q = [Product]m/[Reactant]n, if Q K, Q vs. K: Determining the Direction of Reaction Q = [Product]m/[Reactant]n, if Q K,](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-16.jpg)
Q vs. K: Determining the Direction of Reaction Q = [Product]m/[Reactant]n, if Q K, the reaction must proceed to reach equilibrium. Q < K: [products] /[reactants], “too much _____”, reaction shift to products to reach equilibrium. Shift to R. Q > K: [products]/[reactants] , too much _____”, reaction shift to reactant to reach equilibrium. Shift to L. Q = K: the system is at equilibrium and no further net change takes place. 17 -16

Example: Using reaction quotient to Determine Reaction Direction For the reaction N 2(g) + 3 H 2(g) ⇌ 2 NH 3(g) Kc = 9. 60 at 573 K. At a point during the reaction, [N 2] = 0. 12 M, [H 2] = 2. 1 M and [NH 3] = 0. 21 M. Is the reaction at equilibrium? If not, in which direction is it progressing? 17 -17 0. 040 , to R

K or Q: Combined Reaction, Reversed Reaction Combined reaction: For an overall reaction that is the sum of two or more individual reactions (#1, 2, etc. ): Qoverall = Q 1 x Q 2 x Q 3 x …. . and Koverall = K 1 x K 2 x K 3 x …… Reversed reaction: The form of Q and K depend on the direction in which the balanced equation is written: Qc(rev) = 17 -18 1 Qc(fwd) Kc(rev) = 1 Kc(fwd)

K and Q: Coefficient Change If the coicients of a balanced equation are multiplied by a common factor n, c[D]d [C] Q' = Qn = [A]a[B]b (a) N 2 + 3 H 2 ↔ 2 NH 3 (a) ½ N 2 + 3/2 H 2 ↔ NH 3 17 -19 n and K' = Kn

Example Finding K for an Overall Reaction Nitrogen dioxide is formed is through the following sequence: Given that both reactions occur at the same temperature, find Kc for reaction N 2(g) + 2 O 2(g) ↔ 2 NO 2(g) 17 -20 2. 8 E-15

Example: Finding the Equilibrium Constant for an Equation Multiplied by a Common Factor For the ammonia formation reaction, the reference equation is Kc is 2. 4 x 10 -3 at 1000 K. Find Kc for the following equations: (a) (b) 0. 13 20 17 -21

Practice Finding K Given equilibrium constants for the following reaction: a. 2 H 2 O(l) ↔ OH-(aq) + H 3 O+(aq) K = 1. 0 E-14 b. HF(aq) + H 2 O(l) ↔ F-(aq) + H 3 O+(aq) K = 6. 6 E-4 Find K for reaction F-(aq) + H 2 O(l) ↔ OH-(aq) + HF(aq) 17 -22 1. 5 E-11

K and Q for hetereogeneous equilibrium A hetereogeneous equilibrium involves reactants and/or products in different phases. A pure solid or liquid always has the same “concentration”, i. e. , the same number of moles per liter of solid or liquid. The expressions for Q and K include only species whose concentrations change as the reaction approaches equilibrium. Pure solids and liquids are omitted (treated as 1) from the expression for Q or K. . 17 -23

Example: Write the expression of reaction quotient Q for following reaction 2 H+(aq) + Ca. CO 3(s) ↔ Ca 2+(aq) + H 2 O(l) + CO 2(g) 17 -24
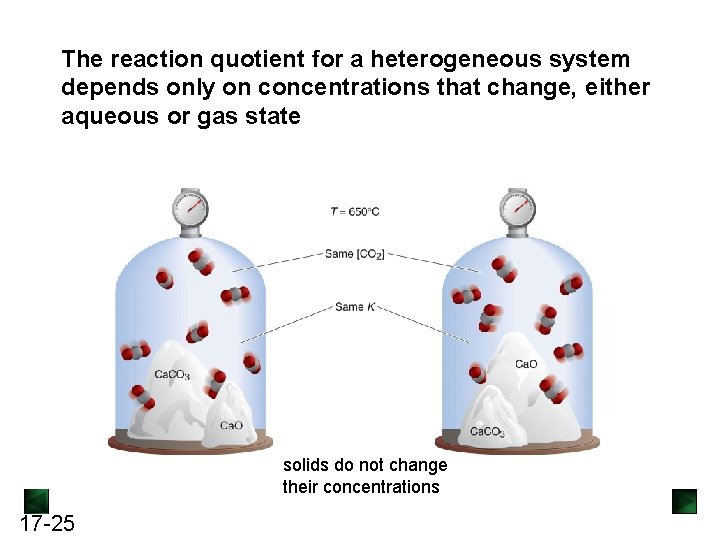
The reaction quotient for a heterogeneous system depends only on concentrations that change, either aqueous or gas state solids do not change their concentrations 17 -25
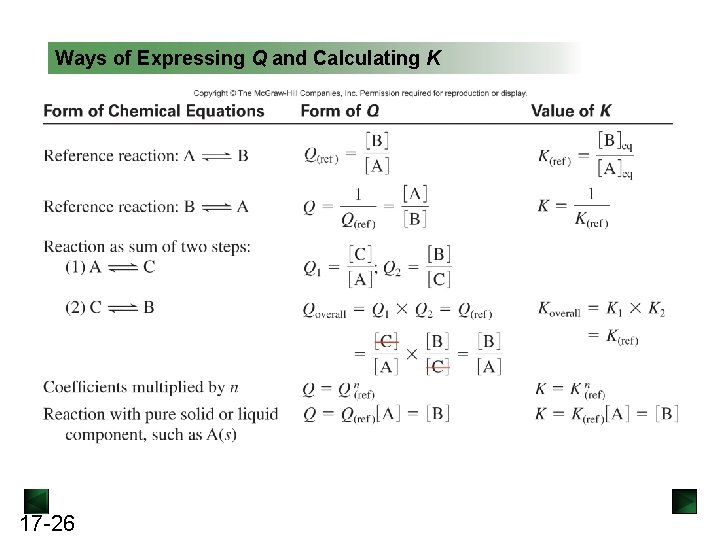
Ways of Expressing Q and Calculating K 17 -26

Expressing Equilibria with Pressure: Kp Kp : K for a reaction involving gas expressed using partial pressures of gaseous reactants instead of molarity. Partial pressure in atmosphere (atm). The partial pressure of each gas is directly proportional to its molarity. Qp = 17 -27 2 P NO 2 P 2 NO x PO Qc = 2 [NO 2]2 [NO]2 [O 2]

Correlation between Kc and Kp ____________ R= Purring Cat gets the nut For reaction a. A(g) + b. B(l) + c. C(s) → d. D(aq) + e. E(g) Dngas = coefficients of gas only Dngas = 2 -(2+1) = -1 If the amount (mol) of gas does not change in the reaction, Dngas = 0 and Kp = Kc. 17 -28

Practice: Converting Between Kc and Kp Find Kc for the following reaction, if CO 2 pressure is in atmospheres. 17 -29 2. 6 x 10 -6

Solving Equilibrium Problems If equilibrium quantities are given, we simply substitute these into the expression for Kc to calculate its value. If only some equilibrium quantities are given, we use a reaction table (or ICE table) to calculate them and find Kc. A reaction table shows • the balanced equation, • the initial quantities of reactants and products, • the changes in these quantities during the reaction, and • the equilibrium quantities. • Change = Equilibrium - Initial 17 -30
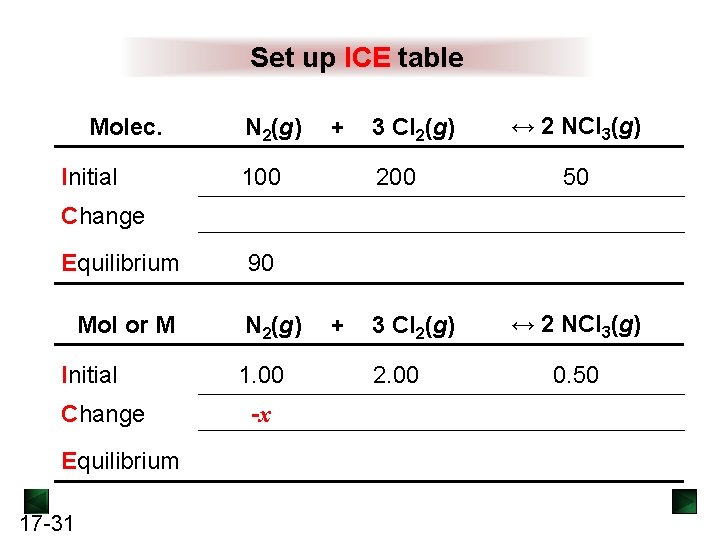
Set up ICE table Molec. Initial N 2(g) + 3 Cl 2(g) 100 200 ↔ 2 NCl 3(g) 50 Change Equilibrium 90 Mol or M N 2(g) + 3 Cl 2(g) Initial Change Equilibrium 17 -31 1. 00 -x 2. 00 ↔ 2 NCl 3(g) 0. 50

Key Features in ICE table For reaction a. A + b. B c. C + d. D The amount of Change depends on the coefficients: Change = -ax ____ Also Change = Equilibrium – Initial So there is 17 -32 E = ____

ICE table: Calculating Kc from Concentration To an evacuated 2. 00 -L flask were filled with 0. 200 mol HI gas and 0. 020 mol H 2 gas and allows the reaction to proceed at certain temperature. 2 HI(g) H 2(g) + I 2(g) At equilibrium, [HI] = 0. 078 M. Calculate Kc. 17 -33 [H 2] = 0. 021 M, [I 2] = 0. 011 M Kc= 0. 038

Basic Algebra plus ICE table to solve Equilibrium Problem Solve quadratic equation ax 2 + bx + c = 0, (x + a)(x + b) = x 2 + ____x + ____ • Set up the ICE table (molarity, Changes with unknown x) • Determine the formula for the equilibrium concentrations (E) of all reactant and product, E = _______ • Establish the equation between K and unknown x • If a quadratic equation is solved and TWO solutions are found, the valid solution x is the one that makes the ALL equilibrium concentrations ______. 17 -34

Example: Given K, find the equilibrium concentrations of reactant and/or products For equilibrium: At certain temperature, Kc = 0. 20. Calculate [N 2 O 4] and [NO 2], when 3. 0 mole NO 2 is allowed to react and reach equilibrium in a 10. 0 -L flask. [N 2 O 4] = 0. 08 M, [NO 2] = 0. 13 M 17 -35

Solving equilibrium concentrations using Quadratic Equation The decomposition of phosgene: COCl 2(g) ⇌ CO(g) + Cl 2(g) Practice: Kc = 0. 83 at certain temperature. Calculate [CO], [Cl 2], and [COCl 2] when 5. 00 mol COCl 2 decompose and reach equilibrium in a 10. 0 -L flask. 17 -36

Example: Solving Equilibrium when K >> 1 or K << 1 The decomposition of phosgene: COCl 2(g) ⇌ CO(g) + Cl 2(g) Kc = 8. 3 x 10 -4 at 360°C. Calculate [CO], [Cl 2], and [COCl 2] when initial [COCl 2 ] = 0. 50 M. 17 -37 [COCl 2] = 0. 480 M, [Cl 2] = [CO] = 2. 0 x 10 -2 M
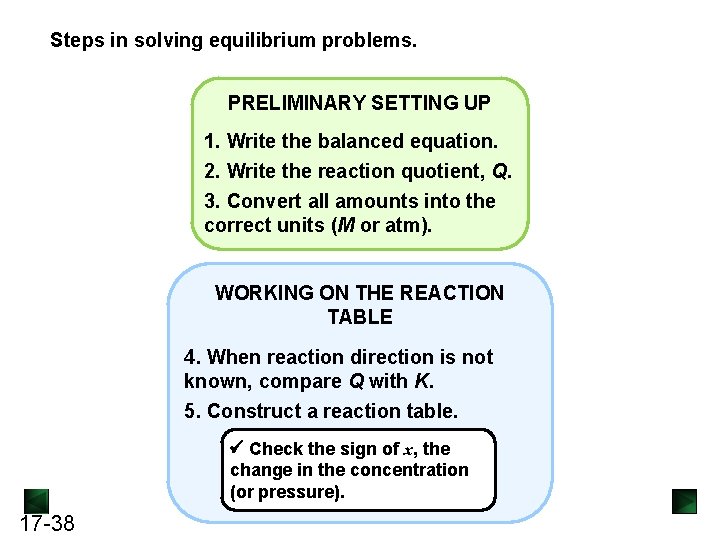
Steps in solving equilibrium problems. PRELIMINARY SETTING UP 1. Write the balanced equation. 2. Write the reaction quotient, Q. 3. Convert all amounts into the correct units (M or atm). WORKING ON THE REACTION TABLE 4. When reaction direction is not known, compare Q with K. 5. Construct a reaction table. Check the sign of x, the change in the concentration (or pressure). 17 -38

(continued) SOLVING FOR x AND EQUILIBRIUM QUANTITIES 6. Substitute the quantities into Q. 7. To simplify the math, assume that x is negligible: ([A]init – x = [A]eq ≈ [A]init) 8. Solve for x. Check that assumption is justified (<5% error). If not, solve quadratic equation for x. 9. Find the equilibrium quantities. Check to see that calculated values give the known K. 17 -39

Le Châtelier’s Principle “When a chemical system at equilibrium is disturbed, it reattains equilibrium by undergoing a net reaction that reduces the effect of the disturbance. ” A system is disturbed when a change in conditions forces it temporarily out of equilibrium. The system responds to a disturbance by a shift in the equilibrium position, until a new equilibrium is reached. Water tank analogy: An Everyday Experience http: //employees. oneonta. edu/viningwj/modules/CI_lechateliers_principle_water_tank_analogy_16_11. html 17 -40

What can affect a chemical equilibrium? Starting from a reaction mixture at equilibrium, Q = K. If reaction quotient Q changes or equilibrium constant K changes, Q ≠ K, the reaction mixture will change to reach a new equilibrium. Recall: Q < K: [products] /[reactants]. To regain equilibrium, [reactants]↓ and [products]↑ Reaction proceeds to the right. Q > K: reaction proceeds to the left to reach equilibrium. 17 -41
![Change in Concentration: Change in Q If [A] increases, the system reacts to consume Change in Concentration: Change in Q If [A] increases, the system reacts to consume](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-42.jpg)
Change in Concentration: Change in Q If [A] increases, the system reacts to consume some of it. • If a reactant is added, the equilibrium position shifts to _____. • If a product is added, the equilibrium position shifts to the _____. If [B] decreases, the system reacts to recover some of it. • If reactant is removed, the equilibrium position shifts to ____. • If product is removed, the equilibrium position shifts to ____. Pure solid or liquid has _______ effect on equilibrium. A change in concentration will NOT affect the value of K. 17 -42

The effect of a change in concentration. 17 -43

Practice: Predicting the Effect of a Change in Concentration on the Equilibrium Position To improve air quality and obtain a useful product, chemists often remove sulfur from coal and natural gas by treating the contaminant hydrogen sulfide with O 2: 2 H 2 S(g) + O 2(g) 2 S(s) + 2 H 2 O(g) What happens to the equilibrium if (a) [H 2 O] if O 2 is added? (c) [O 2] if H 2 S is removed? 17 -44 (b) [H 2 S] if O 2 is added? (d) [H 2 S] if sulfur is added?

Equilibrium that involves Gases • Changes in P or V have no effect on the value of K. • Boyle’s law: at constant temperature, P causes V , vice versa. • Ideal gas law p. V = n. RT: If the moles of gas (n) do NOT equal between reactant and product (Dngas ≠ 0), the volumes of reactants do NOT equal that of products. • More moles of gas corresponds to _____ (larger/smaller) volume. 17 -45

Change in Pressure (Volume) Affect Equilibrium Changes in P or V affect equilibrium systems containing gaseous components. • Increase in P (or decrease in V) causes the shift toward the side with less volume between reactant and product. P or V↑ favor larger ngas. vice versa • Adding an inert gas has no effect on the equilibrium position, as long as the volume does not change. – This is because all concentrations and partial pressures remain unchanged. 17 -46

Change in pressure (volume) affects equilibrium 17 -47

Example: Change in Volume (Pressure) on the Equilibrium Position How would you change the volume of each of the following reactions to increase the yield of the products? Hint: Find Δng (a) Ca. CO 3(s) 17 -48 Ca. O(s) + CO 2(g) (b) S(s) + 3 F 2(g) SF 6(g) (c) Cl 2(g) + I 2(g) 2 ICl(g)

Temperature Effect on Equilibrium Heat as either Reactant or Product of the system. Heat is a _____ in an exothermic reaction (DH°rxn < 0). Example: N 2+ 3 H 2 Û 2 NH 3 + heat Heat is a _____ in an endothermic reaction (DH°rxn > 0). Example: N 2 O 4 + heat Û 2 NO 2 Increase in temperature ____ heat, favoring ____thermic reaction. Decrease in temperature ____ heat, favoring _thermic reaction. 17 -49

Temperature affects Equilibrium in Solution Co. Cl 64 - + 6 H 2 O Û Co(H 2 O)62+ + 6 Cl- : exothermic Lower temperature favors Co(H 2 O)62+ Higher temperature favors Co. Cl 6417 -50 50

Practice: Predicting the Effect of a Change in Temperature on the Equilibrium Position How does an increase in temperature affect the equilibrium concentration of the underlined substance for each of the following reactions? (a) Ca. O(s) + H 2 O(l) Ca(OH)2(aq) DH° = -82 k. J (b) SO 2(g) 17 -51 S(s) + O 2(g) DH° = 297 k. J

Catalysts and Equilibrium A catalyst speeds up a reaction by lowering its activation energy. A catalyst therefore speeds up the forward and reverse reactions to the same extent. A catalyst causes a reaction to reach equilibrium more quickly, but has no effect on the equilibrium position. 17 -52
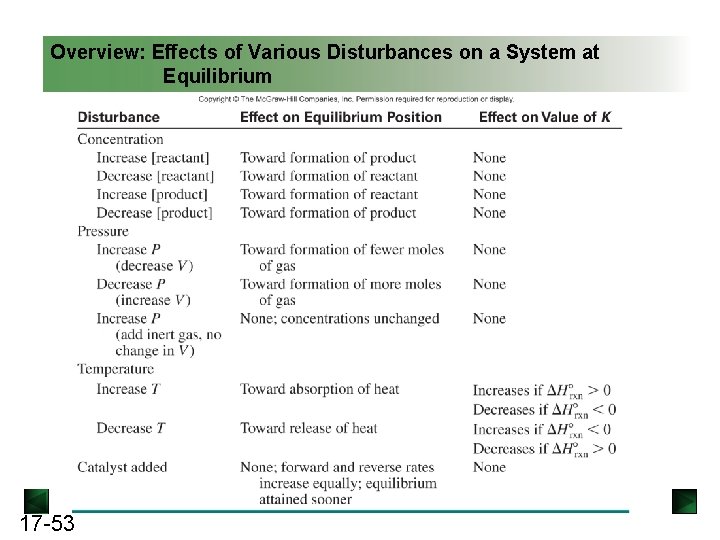
Overview: Effects of Various Disturbances on a System at Equilibrium 17 -53

The Synthesis of Ammonia is synthesized industrially via the Haber process: N 2(g) + 3 H 2(g) 2 NH 3(g) DH°rxn = -91. 8 k. J There are three ways to maximize the yield of NH 3: • Decrease [NH 3] by removing NH 3 as it forms. • Decrease the volume (increase the pressure). • Decrease the temperature. 17 -54

Effect of Temperature on Kc for Ammonia Synthesis N 2(g) + 3 H 2(g) 2 NH 3(g) DH°rxn = -91. 8 k. J T (K) Kc 200. 7. 17 x 1015 300. 2. 69 x 108 400. 3. 94 x 104 500. 1. 72 x 102 600. 4. 53 x 100 700. 2. 96 x 10 -1 800. 3. 96 x 10 -2 An increase in temperature causes the equilibrium to shift towards the reactants, since the reaction is exothermic. 17 -55
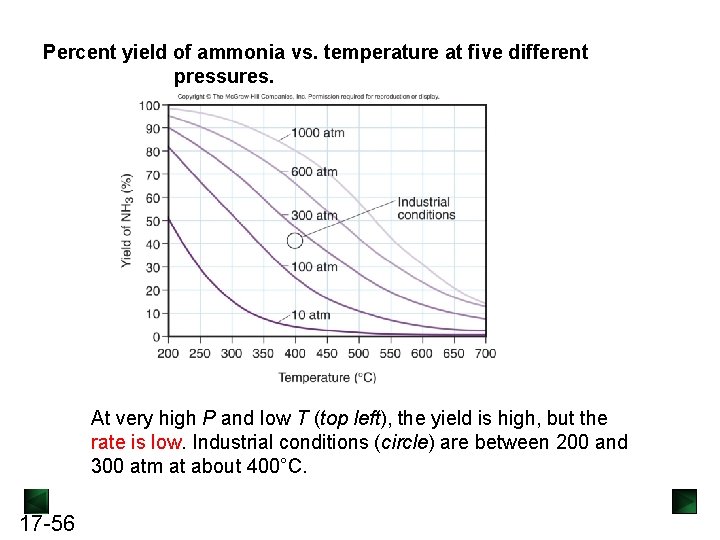
Percent yield of ammonia vs. temperature at five different pressures. At very high P and low T (top left), the yield is high, but the rate is low. Industrial conditions (circle) are between 200 and 300 atm at about 400°C. 17 -56
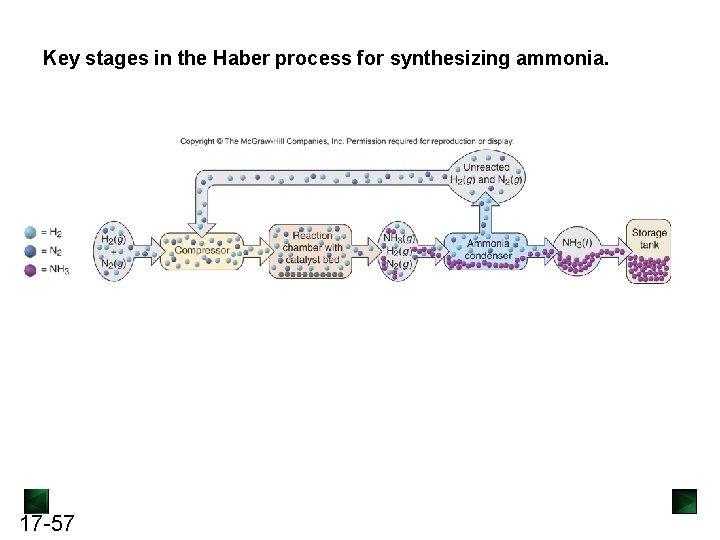
Key stages in the Haber process for synthesizing ammonia. 17 -57
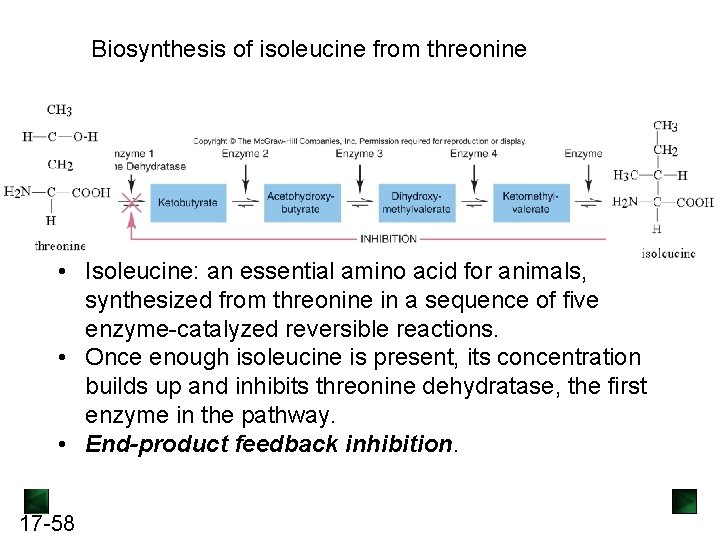
Biosynthesis of isoleucine from threonine • Isoleucine: an essential amino acid for animals, synthesized from threonine in a sequence of five enzyme-catalyzed reversible reactions. • Once enough isoleucine is present, its concentration builds up and inhibits threonine dehydratase, the first enzyme in the pathway. • End-product feedback inhibition. 17 -58
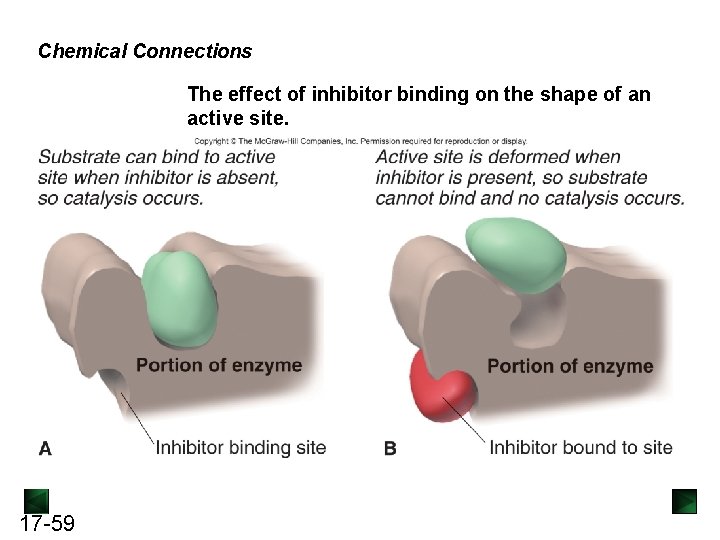
Chemical Connections The effect of inhibitor binding on the shape of an active site. 17 -59

In-depth reading: Temperature affects K The only factor that affects the value of K for a given equilibrium system is temperature. For a reaction with DH°rxn > 0, an increase in temperature will cause K to increase. For a reaction with DH°rxn < 0, an increase in temperature will cause K to decrease. The van’t Hoff equation shows this relationship: ln 17 -60 K 2 K 1 o DH rxn = R 1 T 2 - 1 T 1 R = 8. 314 J/mol·K K 1 is the equilibrium constant at T 1

Online tutorial: Part 1: https: //www. youtube. com/watch? v=7 zu. UV 455 z. Fs (4 min) Temperature effect: https: //www. youtube. com/watch? v=Xh. Q 02 eg. Us 5 Y& (3. 5 min) 17 -61

Le Chatlier: For equilibrium: 2 H 2 S(g) + O 2(g) 2 S(s) + 2 H 2 O(g) What happens to the equilibrium if (a) [H 2 O] if O 2 is added? (b) [H 2 S] if O 2 is added? (c) [O 2] if H 2 S is removed? (d) [O 2] if solid sulfur is added? 2 Qc = [H 2 O] [H 2 S]2[O 2] (a) When O 2 is added, Q decreases and the reaction proceeds to the right until Qc = Kc again, so [H 2 O] increases. (b) When O 2 is added, Q decreases and the reaction proceeds to the right until Qc = Kc again, so [H 2 S] decreases. (c) When H 2 S is removed, Q increases and the reaction proceeds to the left until Qc = Kc again, so [O 2] increases. (d) The concentration of solid S is unchanged and does not appear in the reaction quotient. Adding more S has no effect, so [H 2 S] is unchanged. 17 -62
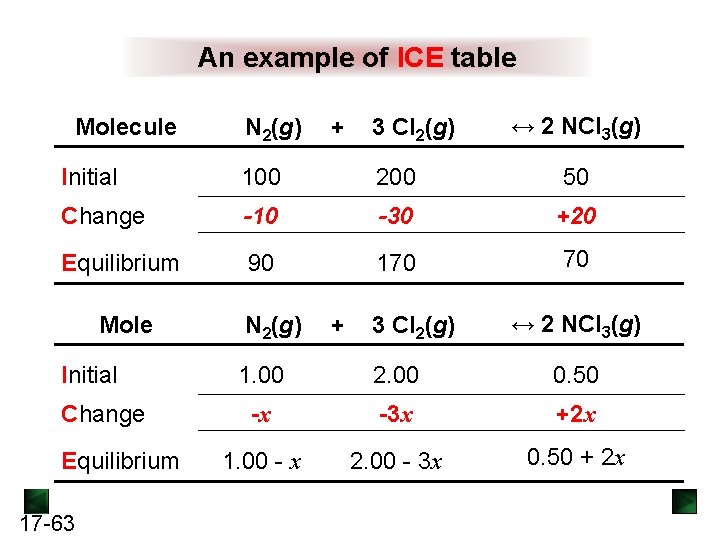
An example of ICE table Molecule N 2(g) + 3 Cl 2(g) ↔ 2 NCl 3(g) Initial 100 200 50 Change -10 -30 +20 Equilibrium 90 170 70 Mole Initial Change Equilibrium 17 -63 N 2(g) + 3 Cl 2(g) ↔ 2 NCl 3(g) 1. 00 2. 00 0. 50 -x -3 x +2 x 1. 00 - x 2. 00 - 3 x 0. 50 + 2 x

Challenges: At 1080 K, gaseous CO 2 is added to a pressure of 0. 458 atm and CO forms. At equilibrium, the total pressure is 0. 757 atm. Calculate Kp. CO 2(g) + C(graphite) At equilibrium PCO 2(eq) = 0. 159 atm PCO = 0. 598 atm 17 -64 2 CO(g) Kp = 2 PCO 2(eq) PCO(eq) 2 = 0. 598 0. 159 = 2. 25

Example: Sometimes, a Simplifying Assumption for calculation helps PROBLEM: The decomposition of phosgene: COCl 2(g) CO(g) + Cl 2(g); Kc = 8. 3 x 10 -4 at 360°C. Calculate [CO], [Cl 2], and [COCl 2] when the following amounts of phosgene decompose and reach equilibrium in a 10. 0 -L flask. (a) 5. 00 mol COCl 2 (b) 0. 100 mol COCl 2 17 -65
![Analysis (a): Kc = [CO][Cl 2] [COCl 2] x 2 = 8. 3 x Analysis (a): Kc = [CO][Cl 2] [COCl 2] x 2 = 8. 3 x](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-66.jpg)
Analysis (a): Kc = [CO][Cl 2] [COCl 2] x 2 = 8. 3 x 10 -4 = 0. 500 - x Since Kc is very small, the reaction does not proceed very far to the right, so let's assume that x can be neglected when we calculate [COCl 2]eqm: Kc = 8. 3 x 10 -4 x 2 ≈ 0. 500 x 2 ≈ (8. 3 x 10 -4)(0. 500) so x ≈ 2. 0 x 10 -2 At equilibrium, [COCl 2] = 0. 500 – 2. 0 x 10 -2 = 0. 480 M and [Cl 2] = [CO] = 2. 0 x 10 -2 M Check that the assumption is justified: x 2. 0 x 10 -2 x 100 = 4%, which is < 5%, so the x 100 = [COCl 2]init 0. 500 assumption is justified. 17 -66
![Analysis (b): 0. 100 mol (b) Calculating initial concentrations, [COCl 2] = = 0. Analysis (b): 0. 100 mol (b) Calculating initial concentrations, [COCl 2] = = 0.](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-67.jpg)
Analysis (b): 0. 100 mol (b) Calculating initial concentrations, [COCl 2] = = 0. 0100 M 10. 0 L [CO][Cl 2] x 2 Kc = = = 8. 3 x 10 -4 0. 0100 - x [COCl 2] If we assume that 0. 0100 – x ≈ 0. 100 x 2 -4 and x ≈ 2. 9 x 10 -3 then Kc = 8. 3 x 10 ≈ 0. 0100 Checking the assumption: 2. 9 x 10 -3 x 100 = 29%, which is > 5%, so the assumption is not 0. 0100 justified. 17 -67
![How to solve part (b): Kc = [CO][Cl 2] [COCl 2] x 2 = How to solve part (b): Kc = [CO][Cl 2] [COCl 2] x 2 =](http://slidetodoc.com/presentation_image_h/d305313e4da94c3231d50ecb556c5672/image-68.jpg)
How to solve part (b): Kc = [CO][Cl 2] [COCl 2] x 2 = = 8. 3 x 10 -4 0. 0100 - x Rearrange the above equation we get x 2 + (8. 3 x 10 -4)x – (8. 3 x 10 -6) = 0 Solve quadratic equation ax 2 + bx + c = 0, Solve the above quadratic equation, we get x = 2. 5 x 10 -3 and 0. 0100 - x = 7. 5 x 10 -3 M [CO] = [Cl 2] = 2. 5 x 10 -3 M, [COCl 2] = 7. 5 x 10 -3 M 17 -68

Example: Predicting the Effect of a Change in Volume (Pressure) on the Equilibrium Position PROBLEM: How would you change the volume of each of the following reactions to increase the yield of the products? (a) Ca. CO 3(s) 17 -69 Ca. O(s) + CO 2(g) (b) S(s) + 3 F 2(g) SF 6(g) (c) Cl 2(g) + I 2(g) 2 ICl(g)

Analysis: SOLUTION: (a) CO 2 is the only gas present. To increase its yield, we should increase the volume (decrease the pressure). (b) There are moles of gaseous reactants than products, so we should decrease the volume (increase the pressure) to shift the equilibrium to the right. (c) The number of moles of gas is the same in both the reactants and the products; therefore a change in volume will have no effect. 17 -70

Practice: Predicting the Effect of a Change in Temperature on the Equilibrium Position PROBLEM: How does an increase in temperature affect the equilibrium concentration of the underlined substance for each of the following reactions? (a) Ca. O(s) + H 2 O(l) (b) Ca. CO 3(s) (c) SO 2(g) 17 -71 Ca(OH)2(aq) DH° = -82 k. J Ca. O(s) + CO 2(g) DH° = 178 k. J S(s) + O 2(g) DH° = 297 k. J

Analysis: SOLUTION: (a) Ca. O(s) + H 2 O(l) Ca(OH)2(aq) + heat An increase in temperature will shift the reaction to the left, so [Ca(OH)2] will decrease. (b) Ca. CO 3(s) + heat Ca. O(s) + CO 2(g) An increase in temperature will shift the reaction to the right, so [CO 2] will increase. (c) SO 2(g) + heat S(s) + O 2(g) An increase in temperature will shift the reaction to the right, so [SO 2] will decrease. 17 -72

Practice: Determining Equilibrium Concentrations and Kc At a certain temperature, 1. 00 mol of N 2 and 2. 00 mol H 2 were placed in a 2. 00 L container where the following reaction occurs N 2(g) + 3 H 2(g) ⇌ 2 NH 3 (g) If a reaction mixture at equilibrium contains 0. 200 M N 2, calculate: a. The concentration of hydrogen gas and ammonia at equilibrium; b. The equilibrium constant Kc at this temperature. 17 -73
- Slides: 73