Equilibrium Systems 15 1 b Equilibrium Constant K

Equilibrium Systems 15. 1 b Equilibrium Constant, K c

Equilibrium Law created in 1864 by Guldberg and Waage (Norweigen) for a reaction: a. A + b. B ⇄ c. C + d. D equilibrium constant: Kc

Equilibrium Law con’t always use a balanced equation with whole number coefficients when using equilibrium law the greater the Kc value the more the products are favoured at equilibrium both temperature and concentration changes can effect the value of Kc Kc only provides a measure of equilibrium position and not info on reaction rates

Equilibrium Constant will always have the same value at a certain temperature no matter what amounts are added ratio at equilibrium will always be same Since equilibrium depends on concentrations, these substances must be expressed as they exist ions in solution are written as net ionic equations
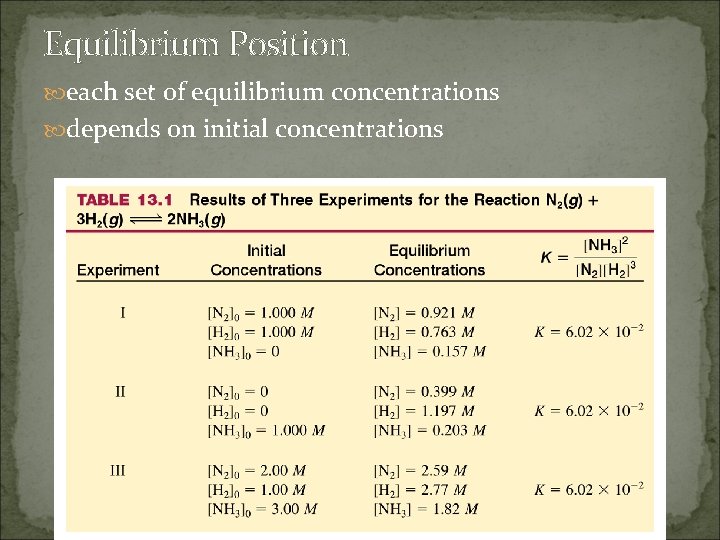
Equilibrium Position each set of equilibrium concentrations depends on initial concentrations

Heterogeneous Equilibrium involves more than one phase position of heterogeneous equilibrium does NOT depend on amounts of: pure solids pure liquids because their concentrations stay constant (since they are PURE)

Heterogeneous Equilibrium do not include liquids or solids in equilibrium expression only include gases and solutions (aq)

Expression does not include pure solids or liquidsonly gases and aqueous species
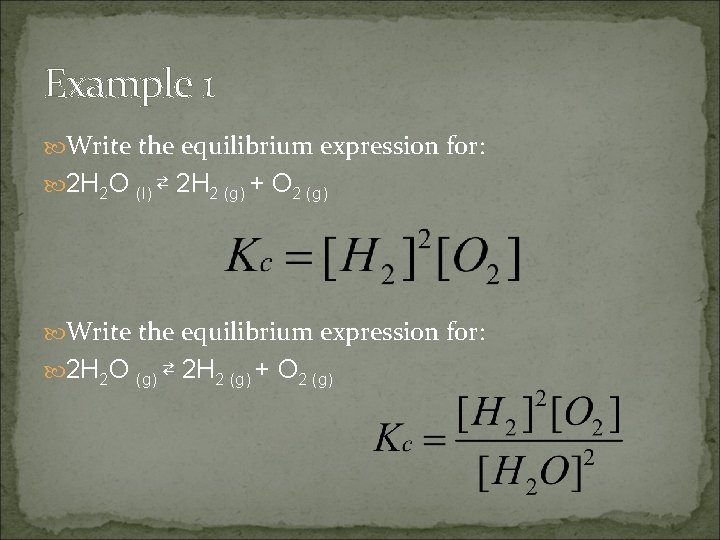
Example 1 Write the equilibrium expression for: 2 H 2 O (l) ⇄ 2 H 2 (g) + O 2 (g) Write the equilibrium expression for: 2 H 2 O (g) ⇄ 2 H 2 (g) + O 2 (g)
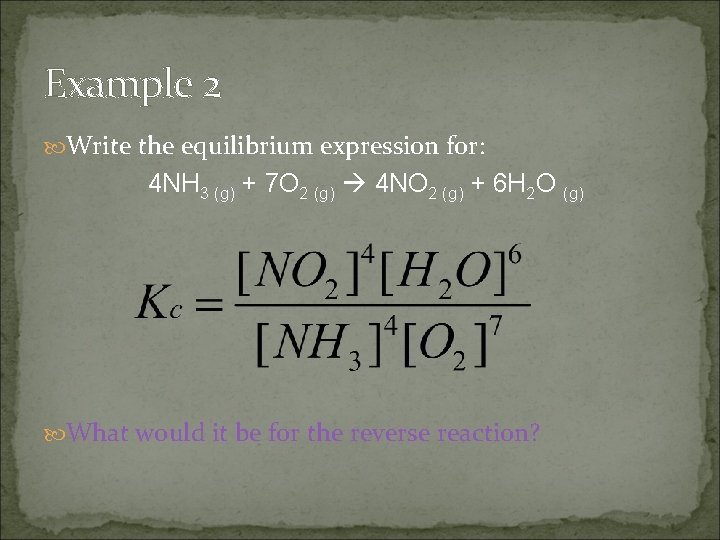
Example 2 Write the equilibrium expression for: 4 NH 3 (g) + 7 O 2 (g) 4 NO 2 (g) + 6 H 2 O (g) What would it be for the reverse reaction?

Homework Text p 688 #1 -4 LSM 15. 1 C summary
- Slides: 11